Apr 4, 2026

Experienced researchers know something beginners do not about tirzepatide purity. They know the number on the label tells only part of the story. They know that "research grade" and "pharmaceutical grade" are not interchangeable terms. They know that a Certificate of Analysis can be fabricated, a purity percentage can be real but still misleading, and that two vials from two different vendors, both claiming 99% purity, can produce wildly different results in practice. Purity is not a simple checkbox. It is a spectrum, a set of standards, and a discipline that separates serious researchers from those who are gambling with their work.
Tirzepatide is not a simple peptide. It is a 39-amino acid dual agonist that targets both GIP and GLP-1 receptors simultaneously, and it carries a C20 fatty diacid lipid modification attached to its backbone.
That lipid tail, which extends the half-life and enables subcutaneous delivery, also makes tirzepatide one of the most technically demanding peptides to synthesize cleanly. Every additional step in synthesis introduces another opportunity for impurities to form. Every modification increases the chances that something in the final product is not what it claims to be.
When researchers talk about pure tirzepatide, they are really asking a layered question. Is the active peptide present at high concentration relative to everything else in the vial? Are known impurities, like diketopiperazine and oxidation products, below acceptable thresholds? Has the vial been tested by an independent laboratory using validated methods? And critically, does the quantity in the vial actually match what the label says? That last question, as you will discover, trips up even experienced buyers who focus exclusively on purity percentage.
This guide covers everything. The chemistry of purity. The testing methods that actually matter. The impurities you need to know by name. The difference between purity and quantity. How to read a CoA without being fooled. And what independent data from real vendor testing reveals about the research peptide marketplace. If you are working with tirzepatide, this is the foundation you need.
What "pure" tirzepatide actually means
The word "pure" gets used loosely in the peptide research space. Vendors put it in product descriptions. Buyers use it to mean "good quality." But in analytical chemistry, purity has a specific and measurable definition, and understanding it changes how you evaluate everything.
Purity, when applied to a peptide like tirzepatide, refers to the percentage of the desired compound present in a sample relative to all other substances. A tirzepatide vial with 98% purity contains 98% tirzepatide and 2% other things. Those other things might be closely related molecular variants, synthesis byproducts, residual solvents, degradation products, or in worst-case scenarios, bacterial contaminants and endotoxins.
The key phrase is "relative to all other substances." Purity is measured by mass or by peak area on an HPLC chromatogram, and the method matters enormously. Two different HPLC methods can produce different purity numbers from the same sample, which is why validated, standardized methods are essential for meaningful comparison across vendors.
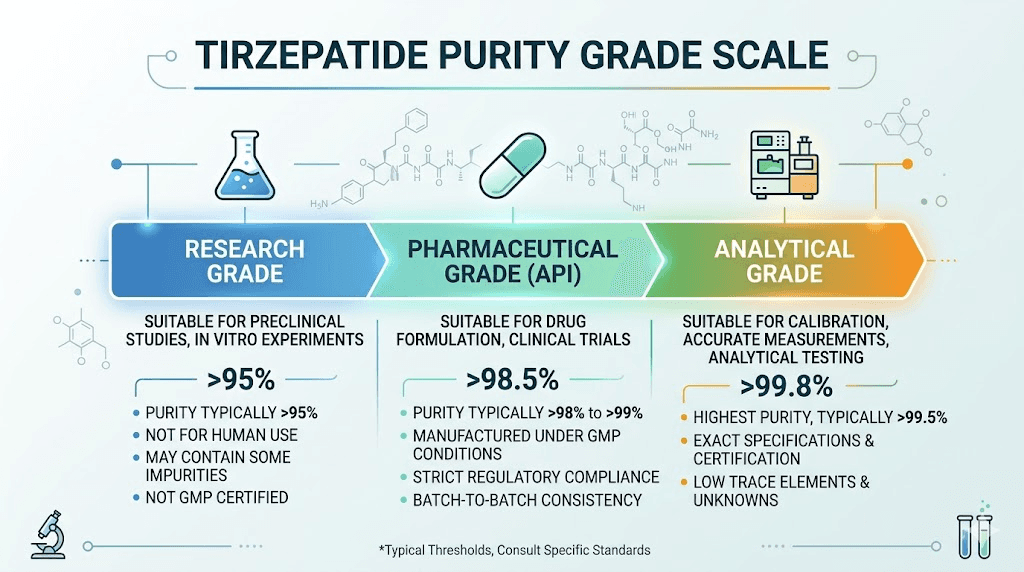
For research applications, two purity tiers are generally recognized. Research grade tirzepatide is typically defined as 95% purity or higher. This is the minimum standard for serious research use, and anything below this threshold introduces too many variables to produce reliable data. Pharmaceutical grade tirzepatide is defined as 98% purity or higher. This is the standard that applies to compounded medications and anything used in clinical settings.
Most legitimate research peptide vendors operate at or near the pharmaceutical grade threshold. According to testing data compiled by Finnrick Analytics from 1,576 samples across 123 vendors, the 5th-to-95th percentile purity range for tirzepatide products runs from approximately 98.74% to 99.95%. That range tells a meaningful story. The best products on the market cluster above 99%. Products at the lower end of that range are still technically pharmaceutical grade, but they sit at the floor rather than the ceiling.
Pure tirzepatide, in the strictest sense, means tirzepatide with a purity value above 99%, tested by reversed-phase HPLC using validated methods, with a CoA issued by an accredited third-party laboratory, and with quantity values that match the label within an acceptable margin. All four of those conditions need to be true simultaneously. Meeting three out of four is not enough.
Understanding this full picture is what separates researchers who get consistent results from those who wonder why their data does not replicate across experiments. Before you look at any other aspect of tirzepatide quality, internalize this definition. Purity is a percentage of active compound. It is verified by specific analytical methods. And it is only part of the complete quality picture.
For context on how tirzepatide compares structurally to other peptides in the GLP-1 space, the semaglutide vs tirzepatide comparison breaks down the structural differences that affect synthesis complexity and, by extension, purity challenges. Tirzepatide is harder to make cleanly than semaglutide, and that difficulty is reflected in vendor pricing and testing requirements.
Understanding purity percentages and grades
Numbers without context are misleading. A purity of 97% sounds impressive until you realize that 3% impurity in a peptide vial can represent multiple distinct contaminants, each with potentially different effects on research outcomes.
Understanding what purity percentages actually mean, and how the grading system works, prevents costly misinterpretations.
Start with the baseline. Any tirzepatide product below 95% purity should be considered unsuitable for serious research use. At purity levels below 95%, the concentration of impurities is high enough to introduce confounding variables that undermine experimental validity. Researchers who prioritize cost over purity at this threshold are not saving money. They are compromising data quality.
The 95-98% range covers most products marketed as "research grade." These products are adequate for preliminary screening and exploratory work, but they fall short of pharmaceutical grade standards. The 2-5% impurity fraction in this range often contains a mixture of deletion sequences, oxidation products, and diketopiperazine formation byproducts. These impurities are not inert. They have their own molecular activity, and at sufficient concentrations, they can shift experimental results in measurable ways.
Above 98% is where pharmaceutical grade begins. Products in the 98-99% range meet the minimum standard for compounded medication formulations and rigorous research protocols. The 1-2% impurity fraction at this level is typically dominated by a single major impurity class rather than a mixture, which makes it more predictable and controllable.
Above 99% is where the best research products operate. At this purity level, the combined impurity fraction is less than 1%, and individual impurities are typically below 0.1% each. This is the range where the active compound dominates so completely that impurity contributions to experimental outcomes become negligible for most research applications. The Finnrick Analytics data showing that the top-performing vendors achieve 99.95% purity represents the practical ceiling for commercially synthesized tirzepatide using solid-phase peptide synthesis methods.
One important nuance: purity percentages from HPLC analysis represent relative purity within the detectable fraction of the sample. Contaminants that do not absorb UV light at the measurement wavelength, including certain endotoxins and residual solvents, may not appear in the HPLC purity value. A comprehensive quality assessment requires supplementary testing beyond HPLC alone. Mass spectrometry confirms molecular identity. Endotoxin testing (LAL assay) detects bacterial contamination. Residual solvent analysis identifies carry-over from synthesis reagents. High purity on HPLC alone is necessary but not fully sufficient.
When evaluating vendor claims, pay attention to how purity is reported. A vendor that reports "purity: 98%" without specifying the testing method, the instrument, or the laboratory is providing information that cannot be independently validated. Legitimate vendors report HPLC purity with method details, CoA issue date, and third-party laboratory identification. The peptide calculator tools help researchers plan experiments based on actual peptide concentration, and accurate purity data is an essential input for those calculations.
How tirzepatide purity is tested
Understanding purity testing is one of the most critical skills for any serious researcher. SeekPeptides provides comprehensive guides on analytical methods, helping members navigate the complexities of peptide quality verification with confidence.
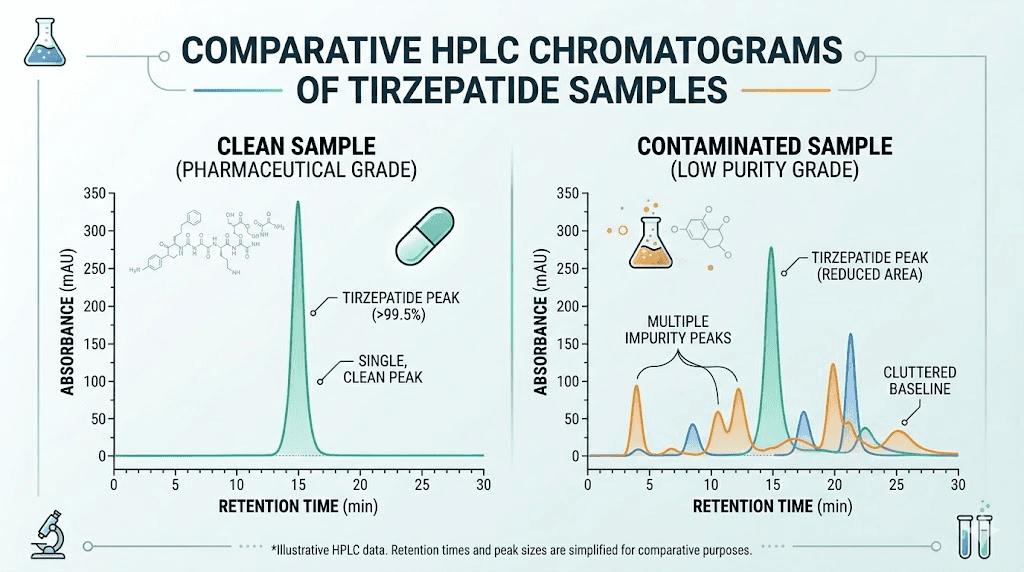
The gold standard for tirzepatide purity testing is reversed-phase high-performance liquid chromatography, or RP-HPLC. This analytical technique separates the components of a sample based on their relative affinity for a stationary phase column and a mobile phase solvent system. The result is a chromatogram where each component appears as a distinct peak, and purity is calculated as the percentage of the target compound peak area relative to all detected peak areas.
For tirzepatide specifically, the HPLC configuration requires particular attention. The lipid modification, the C20 fatty diacid chain attached to the peptide backbone, significantly affects chromatographic behavior. This modification makes tirzepatide more hydrophobic than most research peptides, and the method must account for this. Reversed-phase columns with C18 or C4 stationary phases and 300 angstrom pore sizes provide the appropriate separation characteristics. Smaller pore sizes used for smaller peptides can produce inadequate resolution for a modified peptide of this molecular weight.
The mobile phase system uses a shallow acetonitrile gradient combined with 0.1% trifluoroacetic acid (TFA) as an ion-pairing agent. The TFA improves peak shape and resolution by suppressing silanophilic interactions between the peptide and the silica-based column material. The gradient must be carefully optimized because tirzepatide elutes in a region where closely related impurities, including single amino acid deletion sequences, can co-elute if the gradient is too steep.
Mass spectrometry is the second critical analytical tool for tirzepatide characterization. While HPLC quantifies purity by relative peak area, mass spectrometry confirms molecular identity by measuring the mass-to-charge ratio of detected ions. The molecular weight of tirzepatide is approximately 4813.5 daltons, and the mass spectrum must match this expected value within acceptable tolerance. Mass spectrometry also detects molecular variants, including oxidized forms and deletion sequences, that may partially co-elute with the main tirzepatide peak on HPLC and would therefore be missed or underestimated by HPLC analysis alone.
The combination of RP-HPLC and LC-MS (liquid chromatography-mass spectrometry) provides the most complete purity picture. Laboratories that use both methods can characterize not just the overall purity percentage but also the identity and relative abundance of each individual impurity. This level of detail matters because different impurities have different research implications, and knowing which impurities are present (and at what levels) enables more precise experimental design.
Additional testing that reputable vendors perform includes:
Limulus amebocyte lysate (LAL) assay for endotoxin quantification
Sterility testing to confirm absence of bacterial contamination
Residual solvent analysis by GC or NMR
Karl Fischer titration for water content in lyophilized products
Amino acid analysis to confirm sequence accuracy
Not every vendor performs all of these tests, and not every CoA includes all of this data. But understanding what a complete analytical package looks like helps researchers evaluate vendor CoAs intelligently and identify gaps in quality documentation. The peptide reconstitution calculator requires accurate concentration data, which depends on knowing both purity percentage and actual peptide content per vial, topics explored in detail later in this guide.
Understanding testing methodology is also important for evaluating third-party testing services. Independent laboratories use the same RP-HPLC and LC-MS methods, but the specific column, gradient, and detector settings can influence results. Researchers commissioning their own independent testing should specify that the laboratory uses validated methods for lipid-modified peptides specifically, not general peptide protocols, because the lipid modification on tirzepatide requires method adaptation that not all contract laboratories have implemented.
Common impurities and contaminants in tirzepatide
Impurities in synthetic peptides fall into two broad categories: process-related impurities that arise during synthesis, and degradation-related impurities that form during storage or handling. Tirzepatide has specific vulnerability profiles in both categories due to its structural complexity. Knowing the names and origins of common tirzepatide impurities is not academic trivia. It is practical knowledge that helps researchers interpret CoA data and understand why purity standards matter.
The most significant synthesis-related impurity class in tirzepatide is deletion sequences. These arise when Fmoc-protected amino acid coupling steps during solid-phase peptide synthesis are incomplete. An incomplete coupling step means that one amino acid in the 39-residue sequence is missing in some fraction of the synthesized chains. The result is a collection of peptide molecules that are almost identical to tirzepatide but are missing one or more amino acids at various positions in the chain. These deletion sequences are biologically active, but their receptor binding profile differs from intact tirzepatide. At purity levels below 98%, deletion sequences can constitute a meaningful fraction of the total peptide content.
Insertion sequences represent the complementary problem. If an Fmoc protecting group is not fully removed during deprotection steps, the coupling reaction can proceed but with the wrong chemistry, occasionally resulting in amino acid insertions or modifications. These are less common than deletion sequences but contribute to the impurity profile at lower synthesis efficiency.
Diketopiperazine (DKP) formation is a particularly important impurity in tirzepatide and other N-terminally unprotected peptides. DKP forms when the first two amino acids at the N-terminus of the peptide cyclize spontaneously under certain conditions during synthesis or storage. The reaction is pH-dependent and temperature-dependent. At neutral to mildly acidic pH and elevated temperatures, DKP formation accelerates. In tirzepatide, this impurity can appear during synthesis if conditions are not tightly controlled, and it can also form during improper storage of the reconstituted peptide. DKP is a distinct molecular entity with no GIP or GLP-1 receptor activity, and its presence dilutes the effective concentration of the active compound.
Oxidation products represent another significant impurity class. Tirzepatide contains methionine residues that are susceptible to oxidation, particularly at pH 5, which is near the optimal stability pH for many peptides. Oxidized methionine (methionine sulfoxide) changes the local structure of the peptide and alters receptor binding affinity.
Oxidation occurs both during synthesis (if reagents or conditions introduce reactive oxygen species) and during storage (particularly in improperly sealed vials with residual oxygen). This is one reason why lyophilized tirzepatide stored under inert atmosphere conditions maintains purity longer than liquid preparations. The lyophilized tirzepatide guide covers storage and purity preservation in comprehensive detail.
Residual solvents from synthesis are another impurity category that HPLC does not detect unless supplementary testing is performed. Solid-phase peptide synthesis uses dimethylformamide (DMF), dichloromethane (DCM), and other organic solvents that must be removed during the purification and lyophilization process. If these solvents are not fully removed, they remain in the final product at potentially toxic concentrations. Reputable manufacturers test for residual solvents using gas chromatography and ensure levels fall within International Council for Harmonisation (ICH) guidelines.
Bacterial contamination and endotoxins are the most dangerous category of contaminants for researchers performing in vivo work. Endotoxins are lipopolysaccharide fragments from bacterial cell walls that trigger potent immune responses at very low concentrations. They do not affect HPLC purity values but can completely invalidate biological assays. Products from manufacturers without rigorous sterility testing and endotoxin testing are unsuitable for any application involving biological systems.
One chemically specific interaction unique to compounded tirzepatide formulations deserves special attention: the chemical reaction between tirzepatide and vitamin B12 (cyanocobalamin). Independent analytical testing has confirmed that tirzepatide and cyanocobalamin react when combined in solution, generating measurable impurities that are not present in either compound alone. This has significant implications for researchers working with compounded tirzepatide with B12 formulations. The tirzepatide with B12 complete guide provides detailed information about this interaction and its implications for purity and stability.
Other tirzepatide compound combinations warrant similar scrutiny from a purity standpoint. Tirzepatide combined with niacinamide, tirzepatide with glycine, and tirzepatide with methylcobalamin each present unique chemical compatibility questions that researchers need to evaluate before assuming that combined formulations maintain the same purity profile as single-compound tirzepatide.
Purity vs quantity: the critical distinction researchers miss
This is the section that changes how experienced researchers think about peptide quality. Purity and quantity are two different attributes. They measure different things. They can be independently accurate or independently wrong. And conflating them is one of the most common mistakes in research peptide evaluation.
Purity tells you what fraction of the material in the vial is tirzepatide versus other substances. Quantity tells you how much tirzepatide is actually present in absolute terms. A vial can have 99.5% purity and still contain dramatically less tirzepatide than the label claims. These two quality failures are completely independent of each other.
The data from Finnrick Analytics makes this concrete. In systematic testing of research tirzepatide products, vials labeled as containing 5 milligrams were found to actually contain between 3.89 and 4.29 milligrams, representing a shortfall of 14% to 22% relative to the labeled quantity. These are not outliers from disreputable vendors. This shortfall range reflects the 5th-to-95th percentile variation across the tested product pool. The same vendors who report 99%+ purity on their CoA may still be under-filling vials significantly.
The divergence between labeled and actual quantity can reach plus or minus 43% in extreme cases. A researcher planning a dosing protocol around a labeled 10mg vial who is actually working with 5.7mg of tirzepatide is not running the experiment they think they are running. The implications for research validity are serious.
Why does this happen? Several mechanisms contribute. First, lyophilization efficiency varies. The freeze-drying process that converts reconstituted peptide solutions into the dry powder in research vials is not perfectly precise. Peptide loss during lyophilization is a known phenomenon, and batch-to-batch consistency in fill volume before lyophilization affects final content. Second, analytical weighing errors during vial filling accumulate across production batches. Third, some vendors use purity-corrected versus nominal weight reporting differently, and the labeling conventions are not standardized across the industry.
For researchers who use peptide reconstitution calculators and dosage calculators, this quantity discrepancy has direct practical implications. If you reconstitute what you believe is 10mg of tirzepatide in 2mL of bacteriostatic water, you expect a concentration of 5mg/mL. If the vial actually contains 7.2mg due to the potential for either overfill or underfill, your actual concentration is different from your assumed concentration. Every dosage calculation downstream from that reconstitution step is affected.
How do you account for this? The most rigorous approach is to use vendors with independently verified quantity data, not just purity data. CoAs that include only HPLC purity percentage do not address quantity. Look for CoAs that include a quantitative assay result, expressed as milligrams per vial or as percentage of label claim. Some vendors include this data. Many do not. The vendors that do are demonstrating a higher level of analytical rigor.
The peptide cost calculator becomes more meaningful when quantity accuracy is factored in. A vendor selling 10mg vials at a lower price may actually be delivering less active compound per dollar than a vendor selling the same vials at higher price with verified quantity. Price per milligram only makes sense when milligrams are accurately labeled. Understanding this distinction is fundamental to evaluating value in the research peptide market. The affordable tirzepatide guide explores cost considerations in this broader context.
How tirzepatide structure affects purity challenges
To understand why tirzepatide is harder to synthesize at high purity than most research peptides, you need to understand its molecular architecture. Tirzepatide is not a simple linear peptide. It is a 39-amino acid sequence with a C20 fatty diacid modification, and that modification fundamentally changes both synthesis chemistry and analytical testing requirements.
The 39-amino acid chain is long by solid-phase peptide synthesis standards. Most research peptides targeted for high-purity synthesis contain fewer than 20 amino acids. Longer chains require more coupling and deprotection cycles, and each cycle introduces a small but nonzero probability of incomplete reaction. These error probabilities multiply across 39 steps, meaning the theoretical maximum yield of full-length, fully intact tirzepatide from any single synthesis batch is lower than for shorter peptides. Achieving 99%+ purity with a 39-residue sequence requires exceptional reagent quality, optimized coupling conditions, and rigorous purification by preparative HPLC after synthesis.
The C20 fatty diacid lipid modification adds another layer of complexity. This modification is attached to a lysine residue (at position 26 via a gamma-glutamic acid linker) and consists of a C20 fatty diacid chain that mimics the lipid modifications used in the pharmaceutical tirzepatide (Mounjaro/Zepbound) to extend half-life through albumin binding. The chemistry for attaching this lipid modification during solid-phase synthesis is more complex than standard amide bond formation. It requires orthogonal protecting group strategies and additional coupling steps. Incomplete lipid modification produces a tirzepatide-like peptide without the lipid chain, which is a distinct impurity with different pharmacokinetic behavior.
The lipid modification also affects HPLC analysis, as mentioned in the testing section. The increased hydrophobicity of lipid-modified tirzepatide changes its elution behavior on reversed-phase columns and requires method adaptation. Methods developed for standard peptides may not adequately resolve lipid-modified tirzepatide from all its impurities, potentially causing purity values to be overestimated if the method is not properly validated for this specific compound.
The structural complexity also influences degradation pathways. The lipid chain is not immune to chemical modification during storage. Oxidation and hydrolysis can affect the lipid moiety as well as the peptide backbone, generating degradation products that are specific to tirzepatide and would not be encountered with standard peptides. This is why tirzepatide storage requirements are more stringent than for many other research peptides, a topic covered in detail in the tirzepatide shelf life guide and the tirzepatide expiration guide.
Understanding the structural basis for purity challenges explains why vendor pricing variation exists. A synthesis facility capable of consistently producing 99%+ pure tirzepatide with verified quantity has invested significantly in process optimization, analytical equipment, and quality control infrastructure. This investment is reflected in product pricing. Vendors offering tirzepatide at dramatically below-market prices are, in most cases, either compromising on synthesis quality, skipping analytical testing, or falsifying CoA data. The structural complexity of tirzepatide makes cheap, high-purity synthesis physically improbable.
For researchers comparing tirzepatide to structurally simpler peptides in their work, the injectable vs oral peptides comparison provides context on how molecular complexity affects formulation options. Tirzepatide structural complexity also limits oral bioavailability, which is why tirzepatide oral vs injection research is complicated by the same molecular characteristics that complicate synthesis purity.
Reading and interpreting a Certificate of Analysis
A Certificate of Analysis is the primary quality documentation for research peptides. Every legitimate vendor provides one. But not all CoAs are equal, and knowing how to read one critically is a skill that separates researchers who make informed procurement decisions from those who are easily misled by impressive-looking documents.
The first thing to check is the issuing laboratory. A CoA issued by the vendor themselves, sometimes labeled as "in-house testing," is not independent verification. It is the peptide equivalent of grading your own exam. Legitimate CoAs are issued by accredited third-party analytical laboratories, and the laboratory name, accreditation number, and contact information should be clearly visible on the document. If you cannot find the issuing laboratory or verify its existence and accreditation, the CoA cannot be trusted regardless of what the purity number says.
Next, check the testing date. CoAs are specific to a batch tested at a specific point in time. A CoA from more than a year ago does not tell you anything about the purity of product you receive today, because purity degrades during storage and because vendors may switch synthesis partners or reformulate without updating their documentation. A fresh CoA dated within the last six months for the specific batch you are purchasing is the minimum standard.
The purity reporting format matters. Look for HPLC purity reported as a percentage with the testing method identified. Phrases like "HPLC purity: 99.2% (Method: RP-HPLC, C18 column, UV detection at 220nm)" are informative. A bare number without method identification is not adequate. If mass spectrometry is included (it should be for any serious tirzepatide CoA), the reported molecular weight should match the expected value of approximately 4813.5 Da within 1-2 Da tolerance.
Look for individual impurity peaks listed by relative retention time and percentage area. A comprehensive CoA does not just report total purity. It identifies the major impurities and their relative concentrations. The presence of a single impurity at 0.5% is very different from five impurities at 0.1% each, even though both yield the same 99.5% purity headline number. The identity of the major impurity, if determinable from mass spectrometry, tells you whether you are dealing with a synthesis-related issue (deletion sequences) or a stability-related issue (oxidation products).
Endotoxin results should be present on the CoA for any peptide used in biological systems. Acceptable endotoxin levels for research use are typically below 1 endotoxin unit (EU) per milligram. Values above this threshold indicate inadequate sterility control during synthesis or handling.
The quantity per vial, as discussed, is often missing from CoAs that only report HPLC purity. If the CoA includes a quantitative assay result showing milligrams of tirzepatide per vial, check whether it matches the labeled amount and note the deviation. A CoA that reports 4.7mg in a labeled 5mg vial is being transparent about a known shortfall. A CoA that simply states "purity: 99%" without addressing quantity is leaving half the quality picture unaddressed.
Finally, look for authenticity indicators. QR codes linking to database-verified CoA records, batch-specific serial numbers, and laboratory signatures or stamps are features that legitimate high-quality laboratories include. Some vendors also participate in third-party verification programs where CoA databases can be queried by batch number to confirm document authenticity. The increasing sophistication of CoA forgery in the research peptide space makes these verification mechanisms increasingly important.
The tirzepatide blood work guide touches on related quality verification concepts from a different angle. Researchers who want to understand what verified tirzepatide activity looks like in practice, rather than just on paper, can find useful context there.
Vendor purity ratings and independent testing data
Independent testing data from Finnrick Analytics provides the most systematic public view of tirzepatide purity across the research vendor landscape. The organization has tested 1,576 tirzepatide samples from 123 different vendors, making it the largest independent dataset currently available for this compound. The findings are informative, occasionally alarming, and essential context for any researcher evaluating vendor options.
The overall purity distribution across tested vendors centers well above 98%. The 5th-to-95th percentile range running from 98.74% to 99.95% indicates that the research peptide market, at least among the vendors that Finnrick has tested, is generally producing pharmaceutical grade material. This is a more optimistic picture than the peptide research community sometimes expects, reflecting the maturation of synthesis standards among established vendors over the past several years.
The top-rated vendors in the Finnrick Analytics system, receiving A ratings based on purity, quantity accuracy, and analytical documentation quality, include Polaris Peptides, Aavant Research, Paradigm Peptide, Nuscience Peptides, and Skye Peptides. These vendors consistently demonstrate purity values at or near the top of the distribution and, importantly, show quantity values that more closely match labeled amounts than the market average.
The existence of vendor-specific ratings matters because purity performance is not randomly distributed. There is significant clustering. Top-tier vendors consistently outperform the median. Lower-tier vendors cluster below 99% and show higher variance across batches. Batch-to-batch consistency is itself a quality indicator. A vendor who hits 99.2% one batch and 97.8% the next is not demonstrating the same level of process control as a vendor who consistently achieves 99.1-99.4% across multiple batches.
One significant finding from the Finnrick dataset involves documented vendor fraud. Testing of products from Nantong Guangyuan Chemical revealed sufficient irregularities that all test results for this vendor were retracted from the public database. Retraction of test results typically occurs when evidence emerges that samples submitted for testing were different from products shipped to customers, a practice sometimes called "cherry-picking" or sending "golden samples." This finding underscores that even independent testing has limitations when vendor cooperation with the testing process is fraudulent.
For researchers specifically evaluating compounded tirzepatide from pharmacy sources, the vendor landscape differs from the research chemical market. Compounding pharmacies operate under 503A or 503B designations with specific FDA oversight requirements. The 503B compounding pharmacy tirzepatide guide covers the regulatory framework that governs purity standards in that sector. Specific compounding pharmacy reviews, including Polaris tirzepatide, Empower tirzepatide, and BPI Labs tirzepatide, provide product-specific purity and quality information.
Other compounding sources covered in depth include Red Rock Pharmacy tirzepatide, Vitastir tirzepatide, Strive tirzepatide, and Lavender Sky tirzepatide. Each source has different analytical documentation practices and purity track records that researchers and clinicians evaluate when making sourcing decisions. Sources like Simple Peptide tirzepatide, Trava tirzepatide, and IVIM tirzepatide each have dedicated guides covering their specific product offerings and quality standards.
Telehealth platforms that provide tirzepatide access, including ProRx tirzepatide, Peter MD tirzepatide, Medvi tirzepatide, and Zealthy tirzepatide, operate as prescription pathways rather than direct research chemical suppliers. Their purity standards reflect pharmacy compounding regulations rather than research chemical market conventions, which generally provides a higher baseline of oversight and documentation.
For budget-conscious researchers, the cheap compounded tirzepatide guide addresses where cost savings are actually achievable without sacrificing purity standards, and where price cutting correlates with quality cuts that undermine research validity.
How storage and handling affect purity over time
Even the purest tirzepatide degrades without proper storage. SeekPeptides members access detailed storage protocols and stability timelines for every major peptide, including temperature-specific guidance that most vendors never provide.
Purity at the time of manufacture is not purity at the time of use. Every tirzepatide vial is on a slow journey toward lower purity from the moment it is sealed. The rate of that journey depends entirely on storage and handling conditions. Researchers who understand degradation kinetics can slow purity loss dramatically. Those who do not can render a 99.5% pure vial functionally unusable within weeks.
Lyophilized tirzepatide is the most stable form. In its dry, freeze-dried state, the molecular mobility required for most degradation reactions is severely limited. Water content in lyophilized peptides is typically below 5%, and at this moisture level, hydrolysis (the primary degradation pathway for peptides) proceeds at a negligible rate. Lyophilized tirzepatide stored properly, meaning refrigerated at 2-8 degrees Celsius and sealed under inert atmosphere, maintains pharmaceutical grade purity for 12 months or more without significant purity loss. Some manufacturers provide two-year stability data for properly stored lyophilized tirzepatide.
Temperature is the dominant variable for lyophilized storage. Every 10-degree increase in storage temperature roughly doubles the rate of degradation reactions (a general approximation of the Arrhenius equation as applied to peptide stability). Room temperature storage of lyophilized tirzepatide accelerates purity loss by a factor of roughly 4-8 compared to refrigerated storage. A vial that would maintain 99%+ purity for 12 months under refrigeration might fall to 95% purity within a few months at room temperature. The tirzepatide refrigeration guide covers this in practical terms.
Reconstitution fundamentally changes the stability picture. When lyophilized tirzepatide is dissolved in bacteriostatic water, the molecular mobility required for degradation is restored. The peptide is now in an aqueous environment where hydrolysis, oxidation, and DKP formation can all proceed at meaningful rates. Reconstituted tirzepatide should be used within 28 days when stored refrigerated at 2-8 degrees Celsius. Beyond this window, purity degradation is measurable and clinically relevant. The tirzepatide reconstitution guide covers proper technique for minimizing purity damage during this process.
The pH of the reconstitution solution matters. Bacteriostatic water has a pH near neutral (6.5-7.0), which is acceptable but not optimal for tirzepatide stability. Some formulations use slightly acidic buffers (pH 5-6) because many peptides show improved stability in mildly acidic conditions. However, tirzepatide has specific vulnerabilities near pH 5, including accelerated oxidation at the methionine residue. The reconstitution solution choice affects not just immediate dissolution but downstream stability over the storage period.
Freezing reconstituted tirzepatide causes purity damage. Ice crystal formation during freezing physically disrupts the peptide structure and can cause aggregation, which reduces effective concentration and changes the purity profile. This is a critical point that contradicts intuition. Freezing something to preserve it is not universally valid for peptides. The tirzepatide freezing guide explains this counterintuitive finding in detail. Only lyophilized tirzepatide should be frozen. Reconstituted tirzepatide should never be frozen.
Light exposure is another degradation factor that is often overlooked. Tryptophan residues in peptides are photosensitive and can undergo photodegradation under UV exposure. While tirzepatide does not contain tryptophan, other aromatic amino acids and the lipid modification can be affected by extended light exposure. Standard practice is to store peptide vials in opaque containers or in the dark to eliminate this variable.
Multiple freeze-thaw cycles for lyophilized powder are acceptable because the dry state limits ice crystal damage. However, frequent temperature cycling even within the refrigerated range introduces thermal stress that can accumulate over time. Best practice is to maintain consistent refrigeration without repeated temperature excursions. The guide on tirzepatide temperature exposure addresses accidental temperature excursions and provides practical guidance on whether a warmed vial can still be used.
For researchers planning longer-term storage of tirzepatide, understanding degradation rates under different conditions is essential for experimental planning. The tirzepatide fridge shelf life guide, the tirzepatide shelf life overview, and the comprehensive shelf life guide provide condition-specific stability data. If you use multiple vials across an extended study, standardizing storage conditions across all vials ensures that purity differences between vials do not become confounding variables in your data.
Compounded tirzepatide purity considerations
Compounded tirzepatide exists in a distinct regulatory and quality context compared to research-grade peptides. Compounding pharmacies, operating under 503A or 503B designations, are subject to state pharmacy board oversight and, for 503B facilities, additional FDA current Good Manufacturing Practice (cGMP) requirements. These regulatory frameworks impose quality standards that are both more formal and, in practice, more variable than the voluntary quality systems used by better research peptide vendors.
The 503A versus 503B distinction matters for purity. A 503A pharmacy compounds medications for individual patients on a prescription basis. These pharmacies are not required to perform release testing on every batch using validated analytical methods. They operate under USP standards but with less FDA oversight of actual testing practices. A 503B outsourcing facility operates more like a pharmaceutical manufacturer. They must follow cGMP guidelines, perform lot-release testing, and maintain analytical documentation that approaches pharmaceutical industry standards. 503B compounded tirzepatide generally offers higher and more consistent purity assurance than 503A compounded product.
The compounding process introduces purity variables that do not exist for pure tirzepatide powder. Compounded formulations often combine tirzepatide with excipients and adjuncts including niacinamide, glycine, B vitamins, and carnitine derivatives. Each additive represents a potential source of chemical interaction with the peptide. The B12 interaction mentioned earlier, where tirzepatide reacts with cyanocobalamin to form detectable impurities, is the most well-documented example. The tirzepatide glycine and B12 compound guide addresses the purity implications of this specific combination.
Other additive combinations have less characterization data. The tirzepatide with levocarnitine combination and tirzepatide levocarnitine blends are prescribed by some compounding pharmacies with varying analytical support for stability and purity. The tirzepatide with hydroxocobalamin formulation represents a different cobalamin variant than cyanocobalamin, and its reactivity profile with tirzepatide may differ. The tirzepatide with B6 combination adds another B vitamin interaction variable. Researchers and clinicians evaluating these combination products should specifically ask compounding pharmacies for purity data on the finished combination product, not just on the tirzepatide bulk ingredient.
Compounded tirzepatide expiration and beyond-use dating adds another dimension to purity management. Unlike pharmaceutical products with FDA-validated expiration dates based on accelerated stability studies, compounded products carry beyond-use dates (BUDs) that are set by pharmacy policy rather than empirical stability data in most 503A settings. The compounded tirzepatide expiration guide covers BUD standards and what they mean for purity. The guide on compounded tirzepatide shelf life and temperature excursion guidance for compounded products provide practical parameters for maintaining purity through the product lifecycle.
The lyophilized vs liquid peptides comparison is directly relevant here. Most research-grade tirzepatide is sold as lyophilized powder, while compounded tirzepatide is often dispensed as a liquid solution (which has inherently shorter shelf life and lower purity stability). Researchers choosing between lyophilized research material and liquid compounded formulations should factor this stability difference into their quality calculations.
Red flags that indicate impure tirzepatide
Knowing what warning signs to look for prevents costly mistakes. Some red flags are visible before purchase. Others emerge after receiving the product. All of them are meaningful signals that purity may be compromised.
Before purchase, pricing is a primary signal. Tirzepatide synthesis at 99%+ purity using validated manufacturing processes has real, substantial costs. Solid-phase peptide synthesizer time, HPLC purification capacity, analytical testing, and sterile lyophilization equipment are all capital-intensive. A vendor pricing tirzepatide at 50-70% below the market average for equivalent quantities is not passing along a special discount. They are signaling that one or more quality steps has been skipped or compromised. The grey market tirzepatide guide addresses the specific risks of below-market pricing in detail.
Absent or suspicious CoA documentation is another pre-purchase red flag. A vendor who cannot provide a current, third-party CoA with full method identification is not meeting minimum documentation standards. A vendor whose CoA lists a laboratory that cannot be independently verified via accreditation databases is potentially providing fabricated documentation. A CoA with perfect round numbers (purity: exactly 99.00%, exactly 5.000mg per vial) is statistically implausible from real analytical measurement. Real analytical data shows decimal variation reflecting measurement uncertainty.
Post-receipt red flags require more careful observation. Tirzepatide in its lyophilized form should appear as a white to off-white powder. Significant color deviation, yellowing, browning, or visible particles in reconstituted solution are indicators of degradation or contamination. The tirzepatide color guide provides a detailed visual reference for normal and abnormal appearance across different tirzepatide formulations and combinations. Comparison guides like the color guide for tirzepatide with B12 are useful for specific combination formulations.
Unusual reconstitution behavior is another post-receipt signal. Pure tirzepatide dissolves readily in bacteriostatic water at room temperature with gentle agitation. Aggregation visible as cloudiness or particulate matter, failure to fully dissolve, or immediate precipitation after reconstitution suggests either degradation or the presence of contaminants that affect solubility. Proper reconstitution technique matters, but even perfect technique cannot rescue severely degraded or contaminated product.
Inconsistent biological activity (for in vivo research applications) is the downstream indicator of purity problems. If experimental results are unexpectedly variable across animals receiving consistent labeled doses, or if the magnitude of observed effects is significantly lower than literature-reported values, purity and quantity inaccuracy are both plausible explanations. Ruling out dosing and administration errors before concluding that the product is impure is essential, but recurring inconsistency that does not resolve with technique refinement warrants independent testing of the product batch.
Unusually long storage before shipping is another concern. Some vendors maintain large stockpiles of product and ship from old inventory. A vial manufactured 18 months ago and stored under unknown warehouse conditions may have significantly lower purity than a freshly manufactured batch, even if the CoA shows high purity at time of manufacture. Asking vendors for manufacture date (not just expiration date) before purchasing provides useful information about product age.
How to verify purity yourself through third-party testing
Researchers who need absolute confidence in their tirzepatide purity can commission independent third-party testing. This is not a routine step for most research applications, but it is appropriate for high-stakes work where purity variance would meaningfully affect outcomes, or when a vendor documentation history raises concerns.
The process starts with selecting an appropriate analytical laboratory. Look for ISO 17025-accredited contract laboratories that explicitly list peptide analysis, and specifically lipid-modified peptide analysis, among their capabilities. General analytical laboratories that do not have experience with modified peptides may use standard peptide HPLC methods that are not optimized for tirzepatide, producing purity values that are not comparable to vendor-reported results.
When submitting a sample for testing, provide the vendor CoA to the laboratory so they can run the same methods and produce directly comparable results. Request RP-HPLC purity analysis using a C18 or C4 column with 300 angstrom pore size and shallow acetonitrile gradient. Request LC-MS identity confirmation with molecular weight verification. If your application requires it, request endotoxin testing and residual solvent analysis as well.
Sample size for testing is an important practical consideration. Most contract laboratories require at minimum 1-5mg of material for comprehensive analysis. For a 10mg research vial, allocating 2-3mg for testing, approximately 20-30% of the vial, is a significant investment. This cost must be weighed against the value of the verified purity data for the specific application. For high-volume research programs ordering from a consistent vendor with strong documentation, periodic batch testing (testing one vial from each new batch order) is a cost-effective verification strategy rather than testing every vial.
Results from independent testing that show significant deviation from vendor-reported purity values, greater than 1-2 percentage points, should trigger both replacement requests and skepticism about the vendor relationship going forward. If independent testing consistently confirms vendor CoA values, confidence in that vendor relationship is justified by empirical data.
Some research organizations that work with tirzepatide at scale have developed in-house HPLC capability for routine purity checks. This requires significant capital investment and method development time, but it eliminates the turnaround time and per-sample cost of contract laboratory testing. For academic or institutional research groups with access to existing analytical infrastructure, adapting existing HPLC methods for tirzepatide analysis is more straightforward than establishing capability from scratch.
For researchers who want to understand what verified pure tirzepatide looks like in biological context, reviewing the published literature on tirzepatide receptor binding affinity and metabolic effects provides benchmark data. Research using pharmaceutical-grade tirzepatide (the Eli Lilly reference standard) provides the clearest activity benchmarks against which research-grade material performance can be compared. Significant divergence from literature benchmark values can indicate purity or quantity issues in the research material being used.
SeekPeptides members have access to detailed guides on research verification strategies, including protocols for evaluating peptide quality and interpreting analytical documentation. The platform resources complement the technical information in this guide with practical application guidance that helps researchers make confident procurement and experimental design decisions.
Tirzepatide dosing and purity: practical research implications
Purity knowledge is only useful when it translates into better research practice. This section connects purity understanding to practical dosing and experimental design decisions that researchers face in day-to-day work with tirzepatide.
The starting point for any tirzepatide research protocol is understanding what you actually have in the vial. High purity percentage combined with accurate quantity information tells you the mass of active tirzepatide available. From that starting point, reconstitution volume and dosing calculations follow directly. The tirzepatide dosage in units guide and the tirzepatide dose chart provide the reference frameworks for translating milligrams to injection volumes. The compounded tirzepatide dosage calculator guide and the dosage calculator tool support the arithmetic.
When purity is below the 99% threshold, some researchers apply a purity correction factor to their dosing calculations. If a batch tests at 97% purity and you want to deliver the equivalent of 2.5mg of pure tirzepatide, you would use approximately 2.58mg of the 97% material to account for the 3% impurity fraction. This type of purity-adjusted dosing is standard practice in pharmaceutical manufacturing and is increasingly relevant in research settings as awareness of purity variation grows.
The compounded tirzepatide starting dose guide addresses dose selection for new protocols. The microdosing tirzepatide chart and microdosing tirzepatide guide cover lower-dose protocols where purity accuracy is especially important because the absolute quantity of active compound per injection is small and errors are proportionally larger.
Injection technique contributes to purity preservation post-reconstitution. Proper subcutaneous injection practices minimize exposure of the reconstituted peptide to contamination and ensure accurate dose delivery. The stomach injection guide, syringe injection guide, and thigh injection site guide provide technique reference. Appropriate needle selection from the needles for tirzepatide guide also affects injection quality and site reactions, covered in the injection site reaction treatment guide.
Timing of injections relative to storage conditions matters for purity maintenance between doses. The best time to take tirzepatide shots, best day to take tirzepatide, and best time of day to inject tirzepatide guides consider scheduling factors alongside storage considerations. Researchers who reconstitute entire vials at once and use them over multiple weeks must account for the ongoing purity degradation of the reconstituted solution.
For research comparing tirzepatide to other peptides in the GLP-1 class, the switching from tirzepatide to semaglutide guide, the semaglutide vs tirzepatide side effects comparison, and the semaglutide vs tirzepatide comparison page provide cross-compound context. Understanding how purity standards compare across these compounds helps researchers make consistent quality decisions when switching or comparing compounds in multi-arm studies.
Tirzepatide bulk powder and advanced procurement strategies
Research programs that use tirzepatide at scale, whether for multi-animal in vivo studies or extended protocol development, often evaluate bulk procurement options to manage cost and ensure batch consistency. Bulk tirzepatide powder purchasing introduces unique purity considerations compared to individual vial procurement.
Bulk tirzepatide is typically sold in gram quantities as crude or semi-purified synthesis product that is then purified and lyophilized into individual vials either by the buyer or by the seller. The purity of bulk material is highly variable, and the final vial purity depends on the purification steps applied to the bulk. Researchers purchasing bulk material must either perform their own preparative HPLC purification or work with a contract purification service. Skipping purification of bulk material and directly lyophilizing crude synthesis product produces materials with purity levels that are entirely unpredictable without independent testing.
For most academic and institutional research programs, individual pre-purified vials from reputable vendors represent better value than bulk material, even at higher per-milligram cost. The certainty of known purity from a CoA-backed vial is worth more than the cost savings from bulk procurement when research validity depends on consistent active compound concentration. The peptide cost calculator helps researchers model the true cost comparison when factoring in testing and purification costs for bulk material.
For researchers evaluating specific vendor options in the research peptide market, the Peptide Sciences tirzepatide review and Planet Peptide tirzepatide review provide vendor-specific analysis. These reviews evaluate not just purity claims but analytical documentation quality, batch consistency data, and customer-reported experience with product performance.
Purity in the context of tirzepatide research applications
The significance of purity varies somewhat depending on the research application. Understanding how purity requirements scale with application complexity helps researchers apply appropriate rigor to their quality standards.
In vitro assays, including cell-based receptor binding and signaling studies, are most sensitive to purity. Impurities at the 1-5% level can interfere with receptor activation measurements, particularly if any impurity has partial agonist or antagonist activity at GIP or GLP-1 receptors. Deletion sequences of tirzepatide are structurally similar enough to full-length tirzepatide that some may retain partial receptor affinity. For in vitro work, pharmaceutical grade purity at 98%+ is the minimum standard, and 99%+ is strongly preferred.
In vivo animal studies are somewhat less sensitive to purity at the population level, because biological variability between animals typically exceeds the variance introduced by 1-2% impurity differences. However, for precise dose-response studies where the relationship between quantity administered and magnitude of response is the primary endpoint, purity accuracy remains critical. Metabolic research examining tirzepatide metabolic effects, energy effects (tirzepatide energy effects), and weight loss trajectories (tirzepatide weight loss timeline) all depend on accurate dose-response relationships that require purity-corrected dosing.
Exploratory or observational research tracking general physiological responses to tirzepatide administration is least sensitive to exact purity values. If the research question is "does tirzepatide administration produce measurable changes in metabolic markers," a product at 97% purity is probably adequate. If the question is "what is the minimum effective dose of tirzepatide for a specific molecular endpoint," then purity precision becomes research-critical.
Comparative studies that examine tirzepatide in the context of broader peptide categories, such as peptides for fat loss, peptides for muscle growth, or peptides for anti-aging applications, require consistent purity standards across all compared compounds to ensure that purity differences do not confound cross-compound comparisons.
FAQ
What purity level should I require for research-grade tirzepatide?
The minimum acceptable purity for serious research applications is 95%, which defines the lower boundary of research grade. For rigorous experimental work, pharmaceutical grade at 98%+ is the appropriate standard. For highest-quality research with maximum data reliability, seek vendors whose products consistently test above 99% purity by RP-HPLC with third-party CoA documentation. The top-performing vendors in the Finnrick Analytics dataset consistently achieve 99%+ across multiple batch tests, and those vendors represent the quality ceiling currently available in the research market.
Can tirzepatide look pure but actually contain dangerous contaminants?
Yes. HPLC purity measures the relative proportion of the target compound versus UV-absorbing impurities. Bacterial endotoxins, which are the most dangerous contaminant class for biological research, do not absorb UV light at the wavelengths used for peptide detection. A product can report 99%+ HPLC purity while containing endotoxin levels that would invalidate any in vivo experiment and cause severe immune responses in biological systems. Endotoxin testing by LAL assay is separate from HPLC purity testing and should be present on the CoA for any tirzepatide used in biological applications. Do not assume that high HPLC purity means freedom from endotoxins.
How do I know if my tirzepatide has degraded during storage?
Visual inspection provides limited but useful information. Reconstituted tirzepatide should be clear and colorless. Cloudiness, particulate matter, or yellow-brown discoloration are all signs of significant degradation. However, early-stage degradation that meaningfully reduces purity is not always visually detectable. If you have concerns about a specific batch, the most reliable approach is to submit a sample for independent HPLC testing. As a practical baseline, any tirzepatide that has been stored outside of refrigerated conditions for extended periods, or reconstituted for more than 28 days, should be considered potentially degraded regardless of visual appearance.
Why does the quantity in my vial not match the label?
Quantity shortfalls in research tirzepatide vials are common and well-documented. Finnrick Analytics testing found that vials labeled as 5mg typically contain 3.89-4.29mg, a shortfall of 14-22%. This results from several factors including lyophilization yield variability, imprecision in filling volumes before lyophilization, and inconsistent reporting conventions for purity-corrected vs nominal weight. To compensate, use vendors whose CoAs include quantitative assay results (milligrams per vial, not just purity percentage), or apply a conservative overage assumption in your dosing calculations when using vendors without quantity verification data.
Does the lipid modification on tirzepatide affect how I should interpret its purity?
Yes, significantly. The C20 fatty diacid modification on tirzepatide changes its chromatographic behavior on HPLC, requiring column specifications (C18 or C4, 300 angstrom pore size) and mobile phase conditions (shallow acetonitrile gradient with TFA) that differ from standard peptide analysis methods. If a vendor CoA does not specify the HPLC method used, there is no way to know whether the method was appropriate for lipid-modified tirzepatide or was borrowed from a generic peptide protocol. Generic peptide methods may not resolve all lipid-modification-related impurities from the main tirzepatide peak, potentially causing purity to be overestimated. Always verify that the analytical method cited in the CoA is validated for lipid-modified peptides specifically.
How does compounded tirzepatide purity compare to research-grade tirzepatide?
The comparison is nuanced. 503B compounding pharmacies operating under cGMP guidelines can produce tirzepatide to pharmaceutical grade standards with rigorous lot-release testing. This offers purity assurance that is formally regulated and documented. However, compounded tirzepatide dispensed as liquid solution has inherently lower stability than lyophilized research-grade material, meaning that purity at dispensing may differ from purity at use if there is a significant storage period in between. The most important factor in either market is whether the manufacturer performs validated third-party analytical testing and makes the results transparently available. This standard applies equally to research vendors and compounding pharmacies.
Can I trust a CoA that comes directly from the vendor rather than a third party?
No. In-house CoAs, meaning purity data generated and reported by the vendor selling the product, have an inherent conflict of interest that eliminates them as independent quality verification. The research peptide market has documented cases of CoA fabrication, where claimed purity values do not match independent testing results. The minimum standard for trustworthy quality documentation is a CoA issued by an identifiable, accredited third-party laboratory that can be contacted to verify the reported results. QR codes linking to laboratory databases where CoA records can be independently confirmed represent a higher standard that some vendors have adopted to combat document fraud.
What is the connection between tirzepatide purity and its side effects in research?
Impurities in tirzepatide can contribute to side effects that are incorrectly attributed to tirzepatide itself. Endotoxin contamination causes inflammation and immune activation. Deletion sequences with partial receptor activity may produce off-target signaling. DKP formation byproducts have no GIP/GLP-1 activity but their presence means the actual tirzepatide dose delivered is lower than calculated. Understanding this connection is particularly relevant when evaluating reported side effects from research tirzepatide use. The tirzepatide side effects guides, including tirzepatide body aches, tirzepatide bloating, and tirzepatide constipation, discuss side effects attributed to the active compound, but researchers should maintain awareness that impure products may produce additional or atypical effects not seen with pure tirzepatide.
External resources
FDA cGMP Regulations for Pharmaceutical Quality, U.S. Food and Drug Administration
USP General Chapters for Compounding Standards, United States Pharmacopeia
Tirzepatide pharmacology and mechanism of action, National Library of Medicine
ICH Quality Guidelines for Pharmaceutical Development, International Council for Harmonisation
Solid-phase peptide synthesis: advances and applications, National Library of Medicine
For researchers serious about peptide purity and protocol optimization, SeekPeptides offers the most comprehensive resource available. Members get access to evidence-based guides, vendor comparison tools, detailed reconstitution protocols, and a community of thousands who have navigated these exact quality questions. When purity matters, having the right information matters even more.
SeekPeptides members access in-depth protocol guides, vendor evaluation frameworks, and research reference materials that go beyond what is covered in public articles. The platform aggregates analytical data, purity verification resources, and expert guidance specifically developed for peptide researchers who need reliable, actionable information rather than generic overviews. If you are conducting serious tirzepatide research and need to make confident quality decisions, membership provides the depth of documentation and analysis that published resources alone cannot match. Purity matters. Your data quality depends on it. The right resources make the difference between research that replicates and research that confuses.
This is what experienced tirzepatide researchers have always known. Purity is not one number. It is a system. It is the analytical method that generates the number. It is the laboratory that runs the test. It is the documentation that records the result. It is the storage conditions that preserve purity from manufacture to use. And it is the knowledge to distinguish between vendors who demonstrate this discipline and those who merely claim it. You now have that knowledge. Use it.
In case I do not see you, good afternoon, good evening, and good night. May your purity percentages stay accurate, your Certificates of Analysis stay verifiable, and your research outcomes stay reproducible.