Mar 1, 2026
You bought a vial of tirzepatide. Maybe it cost you a significant amount. Maybe you waited weeks for it to arrive. And now you are staring at it, wondering one thing: how long do I actually have before this goes bad?
It is not a simple question. The answer depends on what kind of vial you have, how it was made, whether it has been opened, how you store it, and what formulation sits inside that glass container. A single-dose FDA-approved pen follows completely different rules than a compounded multi-dose vial from a pharmacy. And a lyophilized research vial? That is its own category entirely. Get the storage wrong and you lose potency. Get it very wrong and you waste the entire vial, along with every dollar you spent on it.
This guide breaks down exactly how long each type of tirzepatide vial lasts, from the moment it leaves the manufacturer to the moment you draw that final dose. You will learn the specific timelines for refrigerated storage, room temperature windows, beyond-use dates for compounded formulations, and the signs that tell you a vial has degraded past the point of safe use. Whether you are managing a single pen from your doctor or planning weeks of doses from a 30mg compounded vial, the timelines in this guide will help you get every last effective dose without risking your health or wasting your investment.
SeekPeptides members regularly ask about vial longevity because it directly affects protocol planning, budgeting, and results. The answer is never one number. It is a set of timelines that depend on your specific situation.
Quick answer: how long each tirzepatide vial type lasts
Before diving into the details, here is the overview. Different tirzepatide vial types have dramatically different lifespans, and confusing one for another is one of the most common mistakes people make.
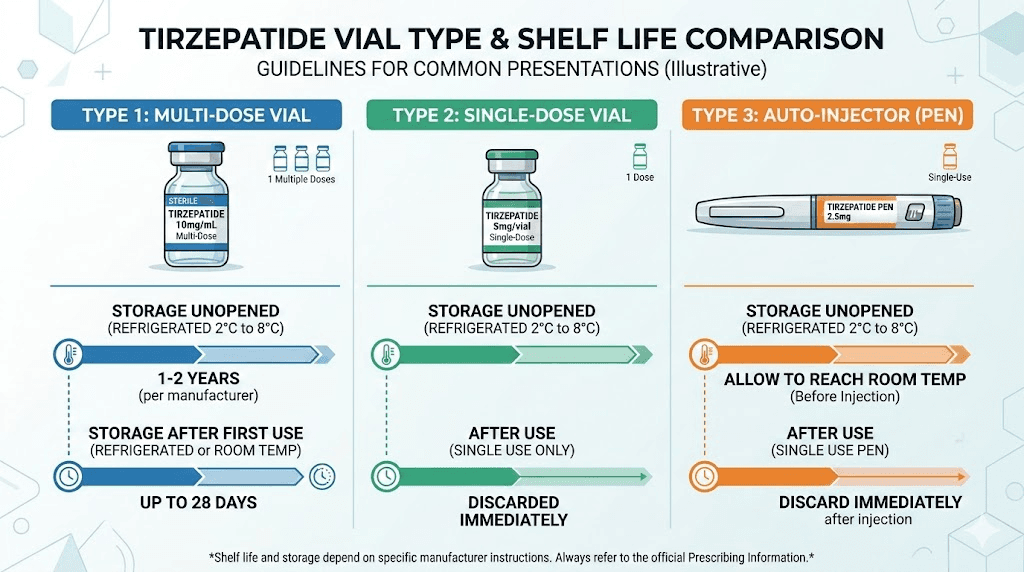
FDA-approved single-dose pens (Mounjaro, Zepbound): These contain one dose each. Use them within 21 days of removing from refrigeration. Unopened and refrigerated, they last until the printed expiration date, which is typically 12 to 18 months from manufacture.
Compounded multi-dose liquid vials: These typically last 28 to 90 days from the date of compounding, depending on the pharmacy. Once the rubber stopper is punctured, most pharmacies recommend using the vial within 28 days. The beyond-use date on your label is the definitive timeline to follow.
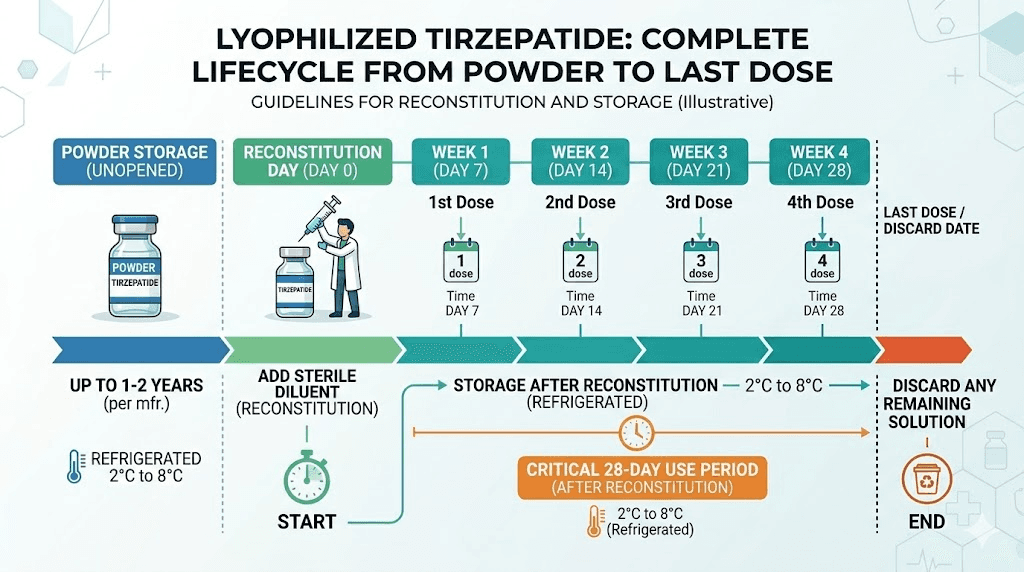
Lyophilized (freeze-dried) powder vials: Before reconstitution, these can remain stable for up to 24 months when stored properly. After reconstitution with bacteriostatic water, most protocols recommend use within 28 days when refrigerated.
Those are the quick numbers. But the real answer requires understanding what happens inside each vial type and why storage conditions can cut those timelines dramatically short.
FDA-approved single-dose pens and vials
The simplest tirzepatide vials to understand are the FDA-approved ones. Mounjaro and Zepbound, both manufactured by Eli Lilly, come as prefilled single-dose pens or single-dose vials. One pen or vial contains exactly one weekly dose in 0.5 mL of solution. You use it once and discard it.
That means a single FDA-approved vial technically lasts one week. You inject your weekly dose, throw away the pen, and open a new one next week. Simple.
But the storage timelines surrounding that single use are where things get important.
Unopened shelf life
An unopened Mounjaro or Zepbound pen stored in the refrigerator at 2 to 8 degrees Celsius (36 to 46 degrees Fahrenheit) remains effective until the expiration date printed on the packaging. This is typically 12 to 18 months from the manufacturing date, though some batches extend further depending on stability testing.
The key word is unopened. As long as that protective cap stays on and the pen remains refrigerated, the manufacturer guarantees full potency through the printed date. This gives you a comfortable window for stocking up or managing refills. If your prescription allows a 90-day supply, those extra pens sitting in the fridge are perfectly fine for months.
Check the expiration date on every pen when you receive it. Pharmacies generally dispense medication well within its shelf life, but it is worth verifying, especially if you receive multiple pens at once from a mail-order pharmacy. The last pen in your supply should still have weeks or months of shelf life remaining when you reach it.
The 21-day room temperature rule
Once a Mounjaro or Zepbound pen leaves the refrigerator, a 21-day clock starts. This applies whether you have used the pen or not. The medication can safely remain at room temperature, defined as below 30 degrees Celsius (86 degrees Fahrenheit), for up to 21 days.
After 21 days outside refrigeration, discard the pen. Even if the solution looks clear. Even if you have not used it yet. The stability data from Eli Lilly supports potency for 21 days at room temperature, and beyond that window, degradation accelerates in ways that may not be visible.
Here is an important detail many people miss: once a pen has been at room temperature, you cannot put it back in the refrigerator. Temperature cycling, moving the medication between cold and warm environments, can cause protein aggregation and degradation that compromises the medication. One-way trip only. Out of the fridge means out for good.
This 21-day rule matters most for travel situations. If you are taking a two-week vacation and cannot maintain refrigeration, your pen will remain effective. A three-week trip pushes right to the limit. Anything longer requires a cooling solution.
Available strengths and what they mean for vial duration
FDA-approved tirzepatide comes in six dose strengths: 2.5 mg, 5 mg, 7.5 mg, 10 mg, 12.5 mg, and 15 mg. Each is packaged as a 0.5 mL single-dose unit. Your prescribed dose determines which strength you receive, but the storage timelines are identical across all strengths.
The standard titration schedule starts at 2.5 mg weekly for the first four weeks, then increases to 5 mg. From there, your provider may increase the dose by 2.5 mg increments every four weeks, up to a maximum of 15 mg per week. Each dose increase means a new pen strength, but the vial longevity rules do not change.
A typical monthly prescription includes four pens (one per week). When your dose changes, the pharmacy dispenses the new strength. There is no situation where you would draw a partial dose from an FDA single-dose pen and save the rest for later. Use it or lose it, in one injection, once per week.
Compounded multi-dose tirzepatide vials
Compounded tirzepatide is a different story. These vials contain multiple doses in a single container, which means you draw from the same vial over several weeks. The question of how long a vial lasts becomes genuinely complex because it depends on the total medication in the vial, your weekly dose, the compounding pharmacy protocols, and your storage habits.
Compounded formulations have been a popular alternative for researchers and patients seeking more affordable tirzepatide options. Understanding vial longevity is critical for both safety and getting full value from each purchase.
Beyond-use dates explained
Every compounded tirzepatide vial carries a beyond-use date (BUD) set by the compounding pharmacy. This is not an expiration date in the traditional sense. It is the date after which the pharmacy cannot guarantee the medication meets its labeled potency and sterility standards.
Beyond-use dates for compounded tirzepatide typically fall between 28 and 90 days from the date of compounding. The variation depends on several factors:
USP guidelines: The United States Pharmacopeia sets standards for compounded sterile preparations. USP Chapter 797 defines beyond-use dating based on the sterility testing and stability data available to the pharmacy.
Preservatives used: Some compounded formulations include preservatives like benzyl alcohol or methylparaben that extend stability. Others do not, which shortens the usable window.
Pharmacy-specific stability testing: Larger compounding pharmacies sometimes conduct their own stability studies, which may support longer beyond-use dates than the default USP minimums.
Formulation type: Liquid ready-to-inject formulations and lyophilized powder formulations have different stability profiles.
The beyond-use date printed on your vial label is the date that matters. Not the date you opened it. Not a date you calculated yourself. Follow the pharmacy label. Period.
How many doses per compounded vial
This is where math becomes your best friend. Compounded tirzepatide vials come in various sizes and concentrations, and the number of doses you get depends on both.
Common compounded vial configurations include:
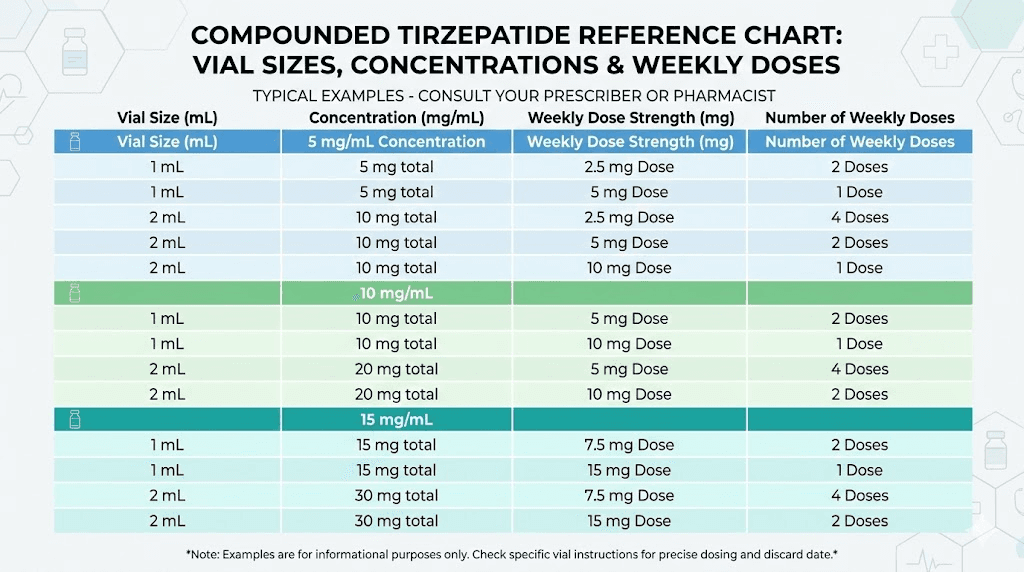
5 mg vial at 5 mg/mL (1 mL total): This contains enough for two doses at 2.5 mg or one dose at 5 mg. At the starting dose of 2.5 mg weekly, this vial covers two weeks.
10 mg vial at 10 mg/mL (1 mL total): This provides four doses at 2.5 mg or two doses at 5 mg. For someone starting out, this vial covers about four weeks, which aligns well with the typical 28-day beyond-use window.
30 mg vial at 10 mg/mL (3 mL total): A popular research vial size. At 5 mg per week, this provides six weeks of doses. At 2.5 mg per week, it covers twelve weeks. However, the 28-day post-puncture timeline often runs out before you use all the medication at lower doses, which creates waste. Use the compounded tirzepatide dosage calculator to plan your specific timeline.
30 mg vial at 20 mg/mL (1.5 mL total): Higher concentration means the same total medication in less liquid. At 10 mg per week, this covers three weeks. At 5 mg per week, it covers six weeks. The 20 mg/mL dosing chart helps with unit conversions at this concentration.
Here is the critical tension: a 30 mg vial at low doses provides enough medication for many weeks, but the 28-day post-puncture window may expire before you use it all. This is why vial size selection matters. Choosing a vial that matches your dose and usage timeline minimizes waste and maximizes value.
After first puncture: the 28-day timeline
The moment a needle pierces the rubber stopper on a multi-dose vial, a new clock starts. Most compounding pharmacies and USP guidelines recommend using the vial within 28 days of the first puncture, regardless of how much medication remains.
Why 28 days? Several things happen once sterility is breached:
Bacterial contamination risk increases. Even with proper technique, each needle insertion introduces a tiny risk of contamination. Bacteriostatic agents in the formulation help, but they have limits. Over time, the cumulative risk of microbial growth becomes unacceptable.
Oxidation accelerates. Oxygen enters the vial each time you draw a dose. Tirzepatide, like all peptides, is susceptible to oxidation. The more air exposure, the faster degradation occurs. This is why some researchers draw air out of the vial after each use, though this is not always practical.
Physical degradation accumulates. Temperature fluctuations during handling, brief exposure to warmth when drawing doses, and agitation all contribute to peptide degradation over time.
Some pharmacies set beyond-use dates longer than 28 days post-puncture, especially for formulations with preservatives. If your pharmacy says 42 days, trust their guidance over the generic 28-day rule. They have stability data supporting that timeline for their specific formulation.
But if no specific post-puncture timeline is given? Default to 28 days. Better to err on the side of potency than to risk injecting degraded medication.
Common compounding pharmacy vial timelines
Different compounding pharmacies use different formulations, preservatives, and testing protocols. This means vial longevity varies by source. Here is what to expect from some common scenarios:
Empower Pharmacy formulations have historically used beyond-use dates in the 42 to 60 day range for their tirzepatide preparations, supported by internal stability testing. Their formulations often include B12 or B6 as compounding agents, which can affect both the color of the solution and stability.
Smaller 503B compounding pharmacies may default to the USP minimum beyond-use dating of 28 days unless they have conducted their own stability studies. This is not a quality indicator. It simply reflects the level of testing performed. A 503B pharmacy following strict USP guidelines is being appropriately conservative.
Always check your vial label for two dates: the date of compounding and the beyond-use date. The gap between them tells you how long the pharmacy believes the formulation remains stable under proper storage conditions.
Lyophilized tirzepatide vials
Lyophilized, or freeze-dried, tirzepatide represents the most stable form available. The powder form dramatically extends shelf life compared to liquid formulations because the primary degradation pathways, hydrolysis and aggregation, require water to occur. Remove the water and you slow degradation to a crawl.
This is the form most commonly encountered in research settings. Understanding both the pre-reconstitution and post-reconstitution timelines is essential for planning protocols and avoiding waste.
Before reconstitution: the long shelf life advantage
Lyophilized tirzepatide stored properly can maintain stability for 12 to 24 months. Some manufacturers report even longer stability windows under optimal conditions. This makes lyophilized vials ideal for researchers who purchase in advance or need flexibility in their protocol timing.
Proper storage for lyophilized tirzepatide means:
Temperature: Refrigerated at 2 to 8 degrees Celsius is ideal. Some lyophilized peptides tolerate room temperature storage for shorter periods, but refrigeration maximizes shelf life.
Moisture: Keep vials sealed and dry. The entire point of lyophilization is removing water. Exposure to humidity reverses that protection.
Light: Store in a dark place or keep vials in their original packaging. UV light accelerates peptide degradation.
The powder should appear as a white to off-white cake or powder. If it looks yellow, brown, or has visible discoloration, degradation has likely occurred. Check the tirzepatide color guide for visual references on what normal and abnormal tirzepatide looks like.
One advantage of the lyophilized form that people often overlook is shipping stability. Liquid formulations can degrade during transit if temperatures are not controlled. Lyophilized powder is far more forgiving during shipping, which is why many research peptide suppliers offer it in this form.
After reconstitution: the clock starts ticking
Once you add bacteriostatic water to a lyophilized tirzepatide vial, the stability profile changes completely. The peptide is now in solution, and all the degradation pathways that lyophilization suppressed become active again.
The standard recommendation for reconstituted tirzepatide is 28 days when stored refrigerated at 2 to 8 degrees Celsius. Some sources extend this to 30 days. Very few go beyond that for peptides reconstituted with bacteriostatic water.
Here is why bacteriostatic water matters. It contains 0.9% benzyl alcohol, which inhibits bacterial growth. This is what allows you to draw from the same vial multiple times over weeks. If you reconstitute with sterile water instead (which contains no preservative), the usable window drops dramatically, often to just 24 to 48 hours. For multi-dose protocols, always use bacteriostatic water. The reconstitution guide covers the complete process.
The 28-day post-reconstitution window is a hard limit for most practical purposes. Even with perfect storage, peptide degradation in aqueous solution is progressive. By day 28, some potency loss has occurred. By day 42, the loss may be significant enough to affect results. By day 56, you are likely injecting substantially degraded material.
Reconstitution volumes and vial duration math
The amount of bacteriostatic water you add during reconstitution affects both your dose measurements and how you calculate vial duration. More water means a more dilute solution and easier measurement of small doses, but the total amount of tirzepatide in the vial remains the same.
For a 30 mg lyophilized vial reconstituted with 3 mL of bacteriostatic water, you get a concentration of 10 mg/mL. At a weekly dose of 5 mg, you draw 0.5 mL per week, giving you six weeks of doses from one vial. But the 28-day stability window means only four of those six weeks fall within the safe use period.
The same 30 mg vial reconstituted with 1.5 mL creates a 20 mg/mL concentration. Now 5 mg requires only 0.25 mL (25 units on an insulin syringe), and the same six weeks of medication is available. The stability window remains the same, but you use less bacteriostatic water per dose, which some researchers prefer. Check the reconstituting tirzepatide chart for detailed volume calculations.
For a 10 mg vial reconstituted with 1 mL (10 mg/mL concentration), at 2.5 mg per week you get four weeks of doses. This is the sweet spot because four weeks aligns perfectly with the 28-day stability window. No waste.
For a 5 mg vial reconstituted with 0.5 mL (also 10 mg/mL), at 2.5 mg per week you get two weeks. This is ideal for shorter trials or when you want to minimize any risk of degradation affecting later doses.
The key principle: match your vial size to your dosing schedule so that you use the entire vial within 28 days. The peptide reconstitution calculator can help you find the right volume for your specific vial and dose combination.
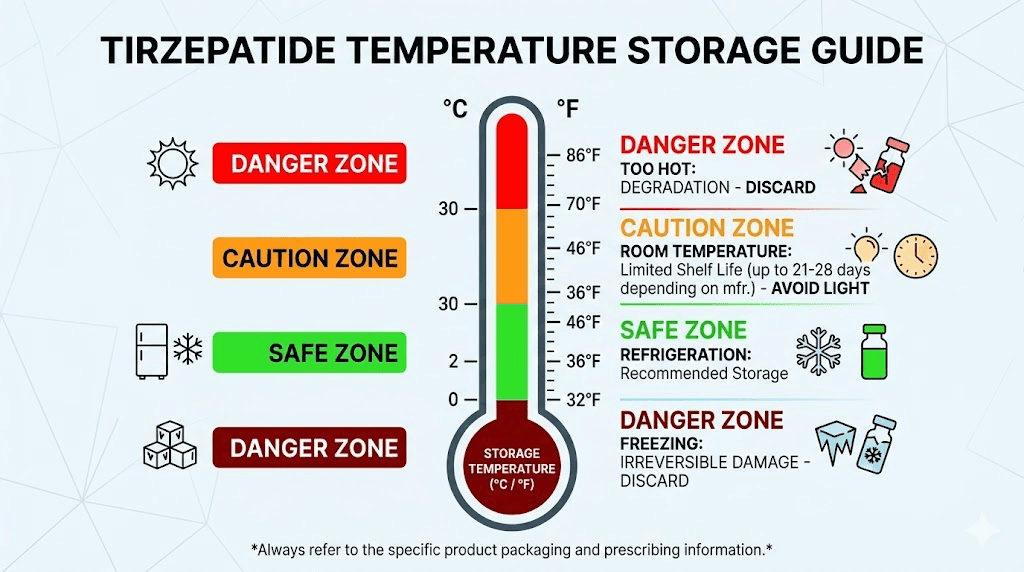
Storage conditions that determine how long your vial lasts
The timelines above assume proper storage. Deviate from the recommended conditions and those numbers shrink. Sometimes dramatically. Understanding exactly what degrades tirzepatide and how fast it happens gives you the knowledge to protect your investment.
Refrigerated storage: the gold standard
Every form of tirzepatide, whether FDA-approved, compounded, or lyophilized, performs best when stored at 2 to 8 degrees Celsius. This is standard refrigerator temperature. Not the door (too warm and variable). Not next to the freezer vent (too cold). The middle shelf of your refrigerator, ideally in the back where temperatures are most consistent.
At refrigerator temperatures, peptide degradation slows to its minimum rate. Chemical reactions that break peptide bonds, cause aggregation, or lead to oxidation all proceed more slowly at lower temperatures. This is basic chemistry, and it is why refrigerator storage is non-negotiable for maximizing vial life.
A dedicated medication refrigerator is ideal but not required. A regular kitchen refrigerator works fine as long as you follow a few rules. Keep the vial away from food that produces moisture. Store it in its original packaging or a sealed container. Do not place it where it might get knocked around or exposed to light every time the door opens.
Some researchers use a small thermometer inside the fridge to verify temperatures stay within range. This is especially useful for older refrigerators that may have inconsistent cooling. If your fridge dips below 2 degrees Celsius regularly, you risk partial freezing, which damages the medication.
Room temperature: the limited window
Room temperature storage is sometimes necessary. Maybe you are traveling. Maybe you need to let the vial warm slightly before injection to reduce discomfort. Whatever the reason, understand the limits.
For FDA-approved tirzepatide (Mounjaro, Zepbound), the room temperature window is 21 days at or below 30 degrees Celsius. After 21 days, discard the medication regardless of appearance.
For compounded liquid tirzepatide, room temperature tolerance varies by formulation. Most pharmacies recommend minimizing room temperature exposure to brief periods for dose preparation. Extended room temperature storage accelerates degradation significantly compared to refrigeration.
For reconstituted lyophilized tirzepatide, room temperature storage is not recommended at all for multi-dose vials. The 28-day refrigerated timeline assumes refrigeration. At room temperature, that window could shrink to days rather than weeks.
Brief room temperature exposure during dose preparation (20 to 30 minutes to let the vial warm slightly) is fine and even recommended by some protocols. Cold injections can be uncomfortable, and a brief warm-up does not significantly impact stability. Just do not leave the vial sitting on the counter for hours.
What happens when tirzepatide gets too warm
Heat is the enemy of peptide stability. Tirzepatide exposed to temperatures above 30 degrees Celsius begins to degrade at an accelerated rate. The higher the temperature and the longer the exposure, the greater the damage.
At 30 to 40 degrees Celsius (86 to 104 Fahrenheit), degradation accelerates noticeably. A few hours at this range may not destroy the medication but will reduce its effective shelf life. Think of it as burning through your stability window faster.
Above 40 degrees Celsius (104 Fahrenheit), degradation can become severe within hours. Leaving a vial in a hot car during summer, even for 30 minutes, can expose it to temperatures exceeding 60 degrees Celsius. At those temperatures, the peptide structure begins to unfold and aggregate irreversibly.
The specific degradation pathways that heat accelerates include:
Deamidation: Asparagine and glutamine residues in the peptide chain convert to aspartic acid and glutamic acid, changing the peptide structure and reducing biological activity.
Aggregation: Heat causes peptide molecules to unfold and stick together, forming inactive clumps that may appear as cloudiness or particles in solution.
Oxidation: Higher temperatures increase the rate of oxidative damage to methionine and other susceptible amino acid residues.
If your tirzepatide has been exposed to excessive heat, our guide on what happens when tirzepatide gets warm covers how to assess whether the medication is still usable.
Light exposure and its effects
Ultraviolet and visible light can degrade peptides through photodegradation. This is why most tirzepatide products come in amber vials or opaque packaging. The colored glass or packaging filters out the wavelengths most damaging to the peptide structure.
Keep your vial in its original packaging whenever possible. If the vial is clear glass (common with some compounded formulations), store it in a dark area of the refrigerator or wrap it in aluminum foil. Direct sunlight or prolonged exposure to fluorescent lighting can noticeably reduce potency over time.
Occasional brief light exposure during dose preparation is unavoidable and inconsequential. The concern is cumulative exposure over days and weeks, not the few minutes it takes to draw a dose.
Can you freeze tirzepatide?
No. Do not freeze tirzepatide in any form.
Freezing liquid tirzepatide causes ice crystals to form within the solution. These crystals physically damage the peptide molecules through a process called freeze-thaw stress. The peptide structure unfolds, aggregates, and cannot be restored by simply thawing the vial. The complete guide to freezing tirzepatide explains the science in detail.
Lyophilized (freeze-dried) tirzepatide is different. The lyophilization process itself involves controlled freezing and vacuum drying, but this is done under precise conditions that protect the peptide structure. Your home freezer does not replicate these conditions. Do not attempt to freeze lyophilized tirzepatide in hopes of extending its shelf life. It is already stable at refrigerator temperatures for months to years.
If your tirzepatide accidentally freezes, consider it compromised. Even if the solution looks normal after thawing, the molecular damage may be present but invisible. Degradation from freeze-thaw cycles does not always produce visible changes.
How to tell if your tirzepatide vial has gone bad
Knowing the timelines is important. Knowing how to visually and practically assess your vial is equally important. Sometimes a vial degrades before its labeled date due to a storage mishap. Other times a vial looks fine past its labeled date. But appearances can deceive. Here is how to evaluate your vial with confidence.
Visual signs of degradation
Start with your eyes. Properly formulated tirzepatide solution should be clear and colorless, or, in the case of formulations containing B12, a light yellow to light pink. Any deviation from this baseline is a red flag.
Cloudiness: A clear solution that has become cloudy indicates peptide aggregation. Microscopic clumps of degraded peptide scatter light, creating that hazy appearance. Do not inject cloudy tirzepatide. This is the most reliable visual indicator of significant degradation.
Particles: Visible particles floating in the solution or settled at the bottom of the vial indicate advanced degradation or contamination. Some particles may be peptide aggregates. Others could be microbial growth. Either way, discard the vial immediately.
Color changes: If your normally colorless tirzepatide has turned yellow, or if a B12-containing formulation has shifted from its expected color range, chemical degradation has occurred. The tirzepatide color guide provides detailed visual references for normal and abnormal appearances across different formulations.
Foaming: Excessive foaming or bubbles that do not dissipate after gentle mixing may indicate protein degradation. Some bubbling during gentle inversion is normal. Persistent foam is not.
Changes in effectiveness as a degradation signal
Sometimes a vial passes the visual test but has still lost significant potency. Degradation does not always produce visible changes, especially in the early stages. Instead, you might notice:
Appetite returning sooner than expected. If your appetite suppression normally lasts six or seven days between doses but suddenly fades by day four or five, reduced potency from degradation could be the reason.
Increased side effects. This seems counterintuitive, but degraded peptides can produce different side effect profiles. Aggregated peptides may trigger injection site reactions or immune responses that the intact peptide does not.
Diminished weight loss progress. If your weight loss trajectory stalls unexpectedly mid-vial, consider whether the medication may have degraded, especially if you are well past the first puncture date or have had any storage mishaps.
These subjective indicators are not definitive, as many factors influence tirzepatide response. But combined with any of the visual signs above, or with a known storage deviation, they strengthen the case for discarding the vial and starting fresh.
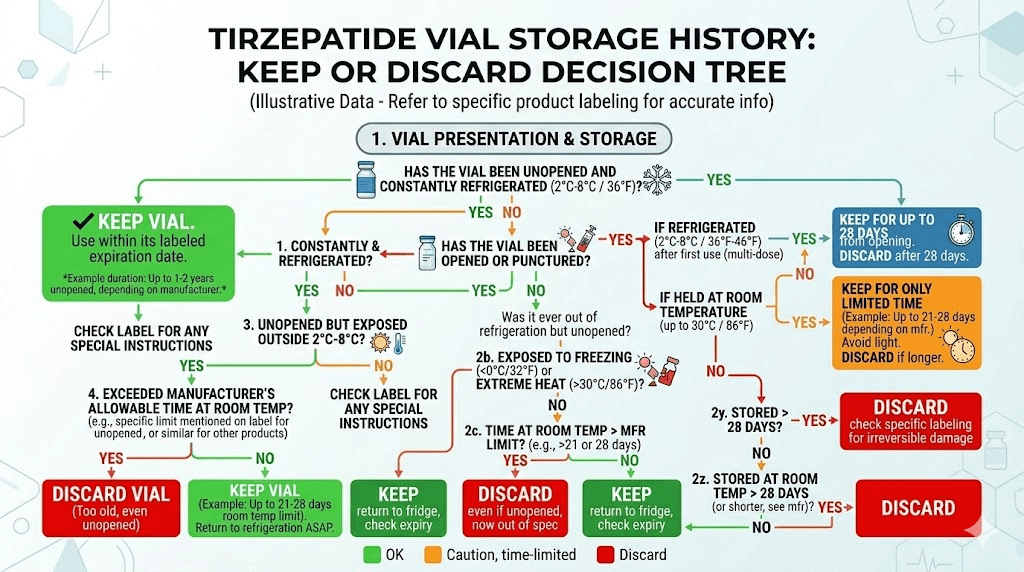
When to discard even if it looks fine
Looks are not everything. Discard your tirzepatide vial when:
The beyond-use date or expiration date has passed. No exceptions. The date on the label is based on stability testing, and extending past it is guessing.
More than 28 days have elapsed since first puncture (for multi-dose vials without a specific pharmacy-provided timeline).
More than 21 days at room temperature (for FDA-approved pens and vials).
Any known heat exposure above 30 degrees Celsius.
The vial was accidentally frozen.
You dropped the vial and the seal may be compromised.
You used the wrong type of water for reconstitution. Sterile water without preservative means the vial should be used within 24 hours, not days or weeks.
The cost of a wasted vial is always less than the cost of injecting degraded medication. Reduced effectiveness wastes your time and money over a longer period. Contaminated medication can cause infection. When in doubt, throw it out. This principle applies to all peptides, not just tirzepatide. Our guide on peptide expiration covers this in broader detail.
How many weeks of doses from one vial: the complete math
This is the section people actually want. How long will one vial sustain your protocol? The answer depends entirely on vial type, total medication, concentration, and your weekly dose. Let me break down every common scenario.
FDA single-dose pens: one week, period
Each Mounjaro or Zepbound pen contains exactly one dose. One pen equals one week. A monthly prescription includes four pens. There is no variability here, and there is no way to extend it. The single-dose design prevents multi-use and ensures accurate dosing every time.
At any dose level, from 2.5 mg starter pens to 15 mg maximum dose pens, one pen lasts one week.
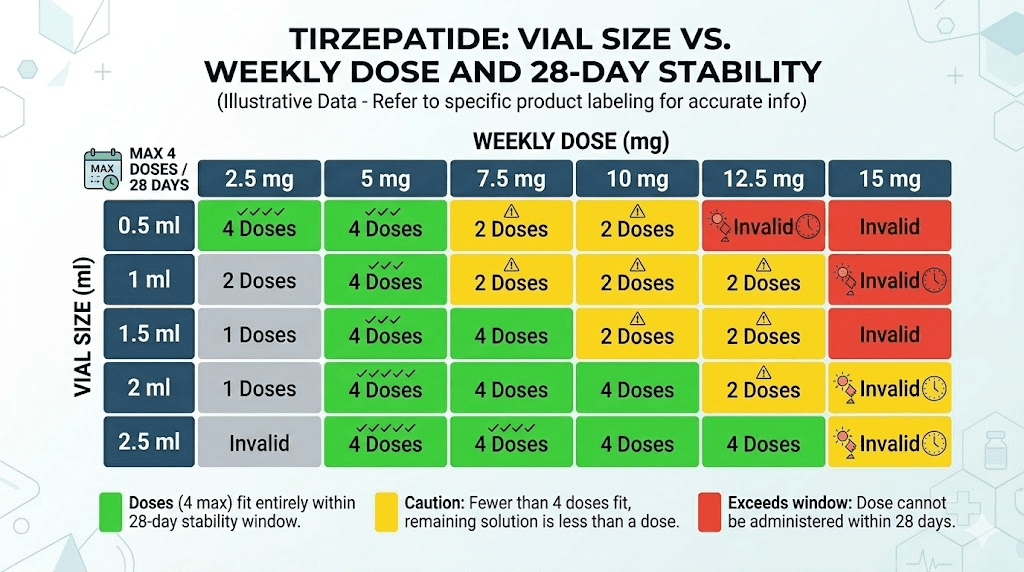
Compounded multi-dose vials: dose-dependent duration
Here is where it gets interesting. The table below shows how many weeks of doses common compounded vial configurations provide at different dose levels:
Vial size | Concentration | Total volume | Weeks at 2.5 mg | Weeks at 5 mg | Weeks at 7.5 mg | Weeks at 10 mg |
|---|---|---|---|---|---|---|
5 mg | 5 mg/mL | 1 mL | 2 weeks | 1 week | N/A | N/A |
10 mg | 10 mg/mL | 1 mL | 4 weeks | 2 weeks | 1.3 weeks | 1 week |
15 mg | 10 mg/mL | 1.5 mL | 6 weeks | 3 weeks | 2 weeks | 1.5 weeks |
30 mg | 10 mg/mL | 3 mL | 12 weeks | 6 weeks | 4 weeks | 3 weeks |
30 mg | 20 mg/mL | 1.5 mL | 12 weeks | 6 weeks | 4 weeks | 3 weeks |
60 mg | 20 mg/mL | 3 mL | 24 weeks | 12 weeks | 8 weeks | 6 weeks |
Now compare these dose-based durations against the 28-day (4-week) post-puncture stability limit. Notice the problem? A 30 mg vial at 2.5 mg per week provides 12 weeks of medication but only 4 weeks of stability after first puncture. That means eight weeks of medication goes to waste unless your pharmacy provides a longer beyond-use date.
The sweet spot varies by dose. At 5 mg per week, a 30 mg vial (6 weeks of medication) exceeds the 28-day window by two weeks. At 7.5 mg per week, the same vial provides 4 weeks of doses, which is a perfect match. At 10 mg per week, it lasts only 3 weeks, meaning you use the entire vial well within the safety window.
This is why choosing the right vial size for your specific dose matters so much. Buying a larger vial does not always save money if half the medication degrades before you can use it.
Lyophilized research vials: reconstitution determines everything
Lyophilized vials add another variable: the reconstitution volume. This affects concentration but not total medication amount. The number of weeks worth of doses remains the same regardless of how much water you add.
For a 10 mg lyophilized vial:
At 2.5 mg/week: 4 weeks of doses (perfectly matches the 28-day stability window)
At 5 mg/week: 2 weeks of doses (well within stability limits)
For a 30 mg lyophilized vial:
At 2.5 mg/week: 12 weeks of doses (far exceeds 28-day window, significant waste)
At 5 mg/week: 6 weeks (exceeds window by 2 weeks)
At 7.5 mg/week: 4 weeks (perfect alignment)
At 10 mg/week: 3 weeks (fully within stability limits)
The reconstitution volume you choose affects how easy it is to measure each dose but does not change these timelines. Adding 3 mL of bacteriostatic water to a 30 mg vial gives 10 mg/mL, where 5 mg equals 50 units on an insulin syringe. Adding 1.5 mL gives 20 mg/mL, where 5 mg equals 25 units. Same medication, different measurement convenience.
Maximizing the usable life of your tirzepatide vial
Understanding the timelines is step one. Actively protecting your vial to maximize those timelines is step two. Small habits make a big difference in how much effective medication you extract from each vial.
Proper storage habits that make a real difference
Store your vial in the same spot in your refrigerator every time. The middle shelf toward the back is ideal because temperatures are most stable there. Avoid the door shelves, which experience the greatest temperature fluctuations every time you open the fridge. Avoid the area directly below the freezer compartment, which can be colder than the target range.
Keep the vial in an opaque container or its original packaging. If your vial is clear glass, wrapping it in aluminum foil provides effective light protection at no cost.
Let the vial warm for 15 to 20 minutes before drawing your dose if cold injections bother you. Place it on the counter, draw your dose, and return it to the fridge immediately after. This brief temperature excursion is well within safe limits and can make the injection more comfortable. Our injection timing guide covers additional tips for comfortable injections.
Write the date you first punctured the vial directly on the label or attach a small sticker. Memory is unreliable, and guessing when you opened a vial is a recipe for using degraded medication. A simple date note eliminates the guesswork.
Drawing technique affects vial longevity
How you draw doses from a multi-dose vial affects how quickly it degrades. Good technique minimizes contamination risk and air exposure.
Swab the rubber stopper with an alcohol wipe before every puncture. This is non-negotiable. Skin bacteria transferred from fingers to the stopper can contaminate the vial over time, and contamination shortens usable life dramatically.
Use a new, sterile needle for every draw. Reusing needles introduces bacteria and can core the rubber stopper, creating channels where contaminants enter. The syringe injection guide covers proper drawing technique in detail.
Minimize the number of times you puncture the stopper. If your dose allows it, drawing multiple doses at once and storing pre-loaded syringes is sometimes recommended by pharmacies. However, the stability of tirzepatide in pre-loaded syringes is less studied, so check with your pharmacy before adopting this approach.
Do not shake the vial. Vigorous shaking causes frothing and can accelerate peptide aggregation. Gentle inversion (turning the vial upside down and back) is sufficient to mix the contents if settling has occurred. Five to ten gentle inversions should produce a uniform solution.
When to order your next vial
Do not wait until your current vial is empty to order the next one. Shipping delays, pharmacy processing times, and supply fluctuations can all create gaps in your protocol. Gaps in weekly dosing disrupt the steady-state medication levels that drive optimal results.
For FDA-approved pens, order your refill when you have two weeks of supply remaining.
For compounded vials, order when you estimate two weeks of doses remain based on your drawing schedule. Account for shipping time, which can be three to seven business days depending on the pharmacy and shipping method.
For lyophilized vials, you have more flexibility because the unreconstituted powder stores for months. Having an extra sealed vial in the fridge as backup eliminates any risk of protocol gaps. Reconstitute the new vial only when your current one is nearing its 28-day limit or running low on medication.
Travel considerations that affect vial life
Traveling with tirzepatide introduces unique vial longevity challenges. Temperature control becomes harder, and the 21-day room temperature window for FDA pens becomes a real constraint on longer trips.
For short trips (under a week), a simple insulated travel pouch with a cold pack keeps the medication within range. Most tirzepatide travel kits maintain appropriate temperatures for 12 to 24 hours.
For longer trips, portable medication coolers that maintain 2 to 8 degrees Celsius are available. These range from simple evaporative cooling cases to battery-powered miniature refrigerators. The investment is worthwhile if you travel frequently.
When flying, always carry tirzepatide in your carry-on luggage. Checked baggage compartments can reach temperatures well below freezing during flight, which would destroy the medication. Carry-on bags remain in the temperature-controlled cabin.
What happens if you use tirzepatide past its expiration
Using expired tirzepatide is not necessarily dangerous in the way that some expired medications are. Tirzepatide does not break down into toxic compounds. The primary risk is reduced effectiveness rather than acute harm.
However, reduced effectiveness is its own kind of waste. You inject the same volume, endure the same injection, and believe you are maintaining your protocol, but the actual dose of active medication may be substantially less than what you intended. This can lead to:
Reduced appetite suppression. The appetite-suppressing effects depend on adequate active tirzepatide reaching GLP-1 and GIP receptors. Degraded tirzepatide may not provide sufficient receptor activation.
Inconsistent blood sugar management. For those using tirzepatide for glycemic control, degraded medication means unpredictable glucose responses.
Immune reactions to aggregated peptides. Aggregated proteins can trigger immune responses that intact proteins do not. Injection site reactions, including redness, swelling, and itching, may be more common with degraded material.
Protocol disruption. If you do not realize the medication has degraded, you might attribute the loss of efficacy to tolerance, dosing issues, or other factors. This leads to unnecessary dose adjustments or protocol changes that complicate your treatment. Our guide on tirzepatide not working anymore covers this scenario and other reasons for efficacy loss.
The bottom line: using expired tirzepatide probably will not harm you directly, but it undermines the entire point of taking it. Check what happens when you use expired tirzepatide for a deeper dive into the specific degradation products and their effects.
Comparing vial longevity across GLP-1 medications
How does tirzepatide vial longevity compare to other GLP-1 receptor agonists? Understanding the differences helps you set appropriate expectations, especially if you are switching between medications or comparing options.
Tirzepatide vs semaglutide vial duration
Semaglutide (Ozempic, Wegovy, compounded forms) follows similar storage principles but with some notable differences:
FDA pens: Ozempic pens are multi-dose (unlike Mounjaro single-dose pens), containing four weekly doses per pen with a 56-day (8-week) in-use period after first use. Wegovy pens are single-dose, similar to Mounjaro. This means an Ozempic pen can last significantly longer than a Mounjaro pen because it contains multiple doses.
Compounded semaglutide: Similar beyond-use dates to compounded tirzepatide, typically 28 to 60 days depending on the pharmacy and formulation. The semaglutide expiration guide covers these timelines in detail.
Room temperature tolerance: Semaglutide can be stored at room temperature (below 30 degrees Celsius) for up to 56 days for Ozempic and 28 days for Wegovy. Compare this to tirzepatide at 21 days. If room temperature storage is a concern (frequent travel, unreliable refrigeration), semaglutide has a slight advantage. For a detailed comparison, see our semaglutide vs tirzepatide comparison page.
Lyophilized forms: Both tirzepatide and semaglutide are available in lyophilized form for research purposes, with similar 28-day post-reconstitution stability windows. The peptide storage after reconstitution guide applies equally to both.
How different tirzepatide formulations compare to each other
Within the tirzepatide family, formulations have different longevity profiles:
Formulation | Unopened shelf life | After opening/reconstitution | Room temp tolerance |
|---|---|---|---|
FDA pens (Mounjaro/Zepbound) | Until printed expiration (12-18 months) | Single use (one dose per pen) | 21 days below 30 C |
Compounded liquid | 28-90 days from compounding | 28 days after first puncture (varies) | Brief periods only |
Lyophilized powder | 12-24 months | 28 days after reconstitution | Not recommended |
Varies by pharmacy | Follow pharmacy BUD | Varies | |
Varies by pharmacy | Individual tablet packaging | Generally room temp stable |
The lyophilized form offers the longest total shelf life but requires reconstitution expertise. FDA pens offer the most convenience with no preparation required. Compounded liquid vials offer the best balance of multi-dose economy and reasonable shelf life. The oral vs injection comparison can help you evaluate which formulation fits your needs.
Protocol planning around vial timelines
Smart protocol planning means aligning your dose, vial size, and purchase schedule so that nothing goes to waste. This is especially important for researchers working with compounded or lyophilized formulations where waste from expired medication can be significant.
Starting a new protocol
If you are just beginning tirzepatide, you will likely start at 2.5 mg per week. At this dose, a 10 mg vial provides four weeks of medication, which is a perfect match for the 28-day stability window. Start here rather than with a larger vial.
After the first four weeks, you will typically increase to 5 mg per week. A 20 mg or 30 mg vial becomes appropriate at this stage. The tirzepatide dosing guide covers the complete titration schedule.
Mid-protocol dose changes
Dose increases can create timing mismatches with your vial. If you increase from 5 mg to 7.5 mg mid-vial, the number of remaining doses changes. Plan for this by:
Timing dose increases to coincide with starting a new vial when possible
Calculating remaining doses at the new higher dose before making the switch
Having a new vial ready if the dose change will exhaust your current vial sooner than expected
Some people choose to split their dose across two weekly injections for better tolerability. This does not change the total weekly medication used or the vial duration math, but it does mean more frequent punctures of the stopper, which slightly increases contamination risk over the vial life.
Microdosing protocols and vial economy
Microdosing tirzepatide at sub-standard doses (0.5 mg to 2 mg weekly) stretches vial contents further but creates a significant tension with the stability window. A 10 mg vial at 1 mg per week technically contains 10 weeks of doses, but the 28-day post-puncture window means you can only safely access four of those ten doses.
For microdosing protocols, smaller vials are essential. A 5 mg vial at 1 mg per week gives five weeks, which is only slightly beyond the 28-day window. A 2.5 mg vial (if available) at 1 mg per week gives 2.5 weeks, well within stability limits.
If only larger vials are available, some researchers reconstitute (for lyophilized) or draw multiple doses in advance and store pre-loaded syringes separately. Consult your pharmacist about the stability of pre-loaded tirzepatide syringes before adopting this approach.
Vial cost efficiency: getting the most value
Understanding vial longevity has direct financial implications. Wasting medication because a vial expired before you could use it all is throwing money away. Here are the cost-efficiency principles that SeekPeptides members use to optimize their protocols.
The peptide cost calculator can help you compare the per-dose cost across different vial sizes and concentrations.
Match vial size to your actual weekly usage. A 30 mg vial costs less per milligram than a 10 mg vial, but only if you use all 30 mg. At 2.5 mg weekly with a 28-day use window, you get 10 mg of usable medication from a 30 mg vial. The remaining 20 mg is waste. In that scenario, three 10 mg vials over twelve weeks costs more per milligram but wastes nothing.
Factor in shipping costs. Ordering fewer, larger vials reduces shipping frequency. But if larger vials create waste, the shipping savings may not offset the medication waste. Calculate both sides.
Consider titration plans. If you plan to increase your dose over the next few months, starting with smaller vials during the low-dose phase and moving to larger vials as your dose increases often provides the best overall economy.
Budget for proper storage. A $15 refrigerator thermometer or a $30 insulated travel case can prevent hundreds of dollars in wasted medication from storage mistakes.
Special situations that affect vial longevity
Compounded formulations with additives
Many compounded tirzepatide vials include additional ingredients beyond the base peptide. Common additions include vitamin B12, vitamin B6, glycine, and niacinamide.
These additives can affect vial longevity in several ways. Some, like glycine, may act as stabilizers that modestly extend peptide stability. Others may introduce additional degradation pathways. B12-containing formulations have a distinct pink or red color that makes visual degradation assessment different from clear formulations.
The beyond-use date set by your compounding pharmacy accounts for the specific additive profile in your formulation. Trust that date rather than trying to extrapolate from generic tirzepatide stability data. Different additives mean different stability profiles.
Combination compounds
Some compounding pharmacies produce tirzepatide combined with other active ingredients, such as levocarnitine or glycine and B12 together. These combination products may have different stability profiles than standalone tirzepatide.
The presence of multiple active ingredients means multiple degradation pathways interacting simultaneously. The beyond-use date for combination products is typically determined by whichever ingredient has the shortest stability, which may or may not be the tirzepatide component.
Power outages and refrigerator failures
A power outage does not immediately destroy your tirzepatide. The inside of a closed refrigerator maintains safe temperatures for approximately four hours. If the power is restored within that window, your medication is fine.
For outages lasting longer than four hours, the temperature inside the refrigerator gradually rises. Once it exceeds 8 degrees Celsius, the degradation rate increases, though not catastrophically at first. Most peptide formulations tolerate brief excursions to 15 or even 20 degrees Celsius without immediate destruction.
If a prolonged outage occurs (12+ hours in warm ambient conditions), consider the vial compromised. You do not know exactly what temperature it reached, and the conservative approach is to start a new vial. A refrigerator thermometer with a maximum temperature memory function can provide useful data in these situations.
Frequently asked questions
Can I use a tirzepatide vial past the beyond-use date if it still looks clear?
While a clear appearance is a good sign, it does not guarantee potency. Peptide degradation can occur at the molecular level without producing visible changes. The beyond-use date is based on stability testing that measures actual potency, not just appearance. Follow the labeled date. The tirzepatide expiration guide explains why visual assessment alone is insufficient.
How long does an unopened compounded tirzepatide vial last in the fridge?
Unopened compounded tirzepatide vials typically last until their beyond-use date, which ranges from 28 to 90 days from the date of compounding. This date appears on the vial label. Always check this date when you receive a new vial to ensure adequate time for your protocol. See our compounded tirzepatide shelf life guide for detailed information by pharmacy.
Does the size of the vial affect how long it stays potent?
The vial size itself does not change the stability timeline. A 5 mg vial and a 30 mg vial of the same formulation degrade at the same rate under the same conditions. The difference is practical: a larger vial takes longer to use up, which means the last doses drawn may have experienced more cumulative degradation than the first doses. Choosing a vial size that you can finish within the stability window eliminates this concern.
I accidentally left my tirzepatide out overnight. Is it still good?
Probably, but it depends on the ambient temperature. One night at room temperature (below 30 degrees Celsius) falls well within the tolerable range for all tirzepatide formulations. If your home was 20 to 25 degrees Celsius overnight, the impact is minimal. If it was a hot summer night exceeding 30 degrees, there is slightly more concern. Return it to the fridge and use it promptly. Check our heat exposure guide for specific temperature thresholds.
How many doses can I get from a 10 mg compounded vial?
At 2.5 mg per week, you get four doses (four weeks). At 5 mg per week, you get two doses (two weeks). At 10 mg, it is one dose. The 10 mg tirzepatide dosage chart provides unit-level conversion details for drawing the correct amount with an insulin syringe.
Is lyophilized tirzepatide better for long-term storage?
Yes, significantly. Lyophilized tirzepatide can remain stable for 12 to 24 months before reconstitution, compared to 28 to 90 days for compounded liquid formulations. If you need to store tirzepatide for extended periods, the lyophilized form is the clear winner. The tradeoff is that it requires reconstitution before use, which adds a preparation step. Learn the process with our reconstitution guide.
What is the difference between bacteriostatic water and sterile water for reconstitution?
Bacteriostatic water contains 0.9% benzyl alcohol as a preservative, which inhibits bacterial growth and allows multi-dose use over weeks. Sterile water has no preservative, meaning reconstituted solutions should be used within 24 hours. For multi-dose vials, always use bacteriostatic water. Sterile water is only appropriate for single-use reconstitution.
Does opening and closing the fridge frequently affect my stored tirzepatide?
Normal refrigerator use (opening the door several times per day for meals) does not create enough temperature fluctuation to affect stored tirzepatide. The concern would be a refrigerator that is opened so frequently it cannot maintain temperatures below 8 degrees Celsius. If your refrigerator runs efficiently and maintains proper temperature, standard daily use is not a concern. Store the vial toward the back of a shelf rather than on the door for the most stable temperature.
External resources
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions. Members access detailed storage guides, stability databases, and handling protocols that protect every dollar spent on peptide research.
In case I do not see you, good afternoon, good evening, and good night. May your vials stay potent, your storage stay cold, and your protocols stay consistent.