Apr 4, 2026

Retatrutide is the most talked-about weight loss drug that nobody can get yet. And for good reason. The Phase 3 data coming out of Eli Lilly's TRIUMPH program shows weight loss numbers that make semaglutide and tirzepatide look modest by comparison. Up to 28.7% body weight reduction in 68 weeks. An average of 71.2 pounds lost. Numbers that would have seemed impossible five years ago.
But here is the frustrating part for researchers tracking this compound. Retatrutide is not FDA approved. Not even close to a formal submission.
Eli Lilly has not filed a New Drug Application, and prediction markets currently sit at just 27% probability for approval happening this year. The realistic window? Late 2027 to early 2028 at the earliest.
So what is actually happening behind the scenes? The TRIUMPH clinical trial program includes multiple Phase 3 studies spanning weight loss, type 2 diabetes, metabolic conditions, liver disease, and even knee osteoarthritis. Results are rolling in throughout this year, and they will determine whether retatrutide reaches pharmacies or joins the long list of promising compounds that never made it. This guide covers everything researchers need to know: the current approval status, every TRIUMPH trial and what the data shows, the new safety signals that emerged, realistic timeline projections, and how retatrutide stacks up against the GLP-1 medications already on the market. Whether you are actively researching this triple agonist or simply tracking the next generation of GLP-1 based treatments, the information here reflects the most current data available from clinical trials and regulatory filings.
What is retatrutide and why does it matter
Before diving into approval timelines and trial data, understanding what makes retatrutide fundamentally different from existing medications explains why so many researchers are watching it closely.
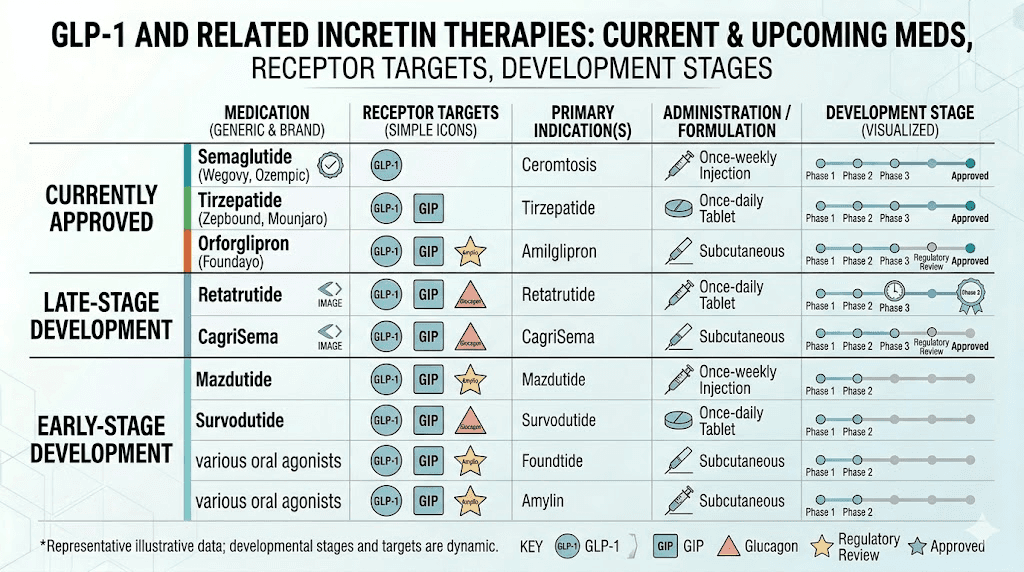
Retatrutide is a triple hormone receptor agonist. That means it activates three different receptors simultaneously: GLP-1 (glucagon-like peptide-1), GIP (glucose-dependent insulinotropic polypeptide), and glucagon. This triple mechanism sets it apart from every weight loss peptide currently available.
Semaglutide targets one receptor. GLP-1 only. Comparing retatrutide to semaglutide reveals a significant difference in approach. Tirzepatide targets two receptors, GLP-1 and GIP. Retatrutide goes further by adding glucagon receptor activation to the mix, creating a synergistic effect that produces greater metabolic changes than either dual or single agonists alone.
The three receptor targets explained
GLP-1 receptor activation reduces appetite, slows gastric emptying, and improves insulin secretion. This is the mechanism behind semaglutide (Ozempic/Wegovy) and the GLP-1 component of tirzepatide. It is well understood, extensively studied, and forms the backbone of modern GLP-1 therapy.
GIP receptor activation enhances the insulin response beyond what GLP-1 alone achieves. It also plays a role in fat metabolism and energy balance. This dual GLP-1/GIP approach is what makes tirzepatide so effective compared to single-agonist medications.
Glucagon receptor activation is the novel addition. Glucagon increases energy expenditure, promotes fat oxidation, and mobilizes glycogen stores from the liver. This is the component that makes retatrutide unique. Instead of just suppressing appetite and improving insulin function, it actively increases the body's energy output. Researchers describe this as the difference between turning down the input and also turning up the output.
The half-life of retatrutide is approximately 6 days, which allows for once-weekly administration. This matches the convenience profile of semaglutide and tirzepatide, which researchers already prefer for adherence reasons.
Why triple agonism produces superior results
The synergistic interaction among these three receptors creates metabolic effects that exceed what any single pathway can deliver. Think of it like this. GLP-1 reduces how much you want to eat. GIP improves how your body processes what you do eat. Glucagon increases how much energy your body burns at rest. Together, they attack the energy balance equation from three directions simultaneously.
This explains the clinical data. In head-to-head comparisons across trials, retatrutide consistently produces greater weight reduction than both semaglutide and tirzepatide at equivalent timepoints. The Phase 2 trial published in the New England Journal of Medicine showed up to 24.2% weight loss at 48 weeks. The Phase 3 TRIUMPH-4 data pushed that even higher, to 28.7% at 68 weeks.
For context, semaglutide (2.4mg) typically achieves about 14.9% weight loss over 68 weeks. Tirzepatide at its highest dose reaches approximately 22.5% over 72 weeks. Retatrutide at 12mg achieved 28.7% in 68 weeks, surpassing both.
Current FDA approval status
As of April 2026, retatrutide is not FDA approved. Eli Lilly has not submitted a New Drug Application (NDA) to the FDA. No international regulatory body has approved retatrutide for any indication.
This is important to state clearly because there is significant confusion online. Some vendors sell research-grade retatrutide peptide, and some websites imply FDA approval is imminent or already in progress. Neither is accurate. The compound remains firmly in the clinical trial phase, with several major studies still awaiting results.
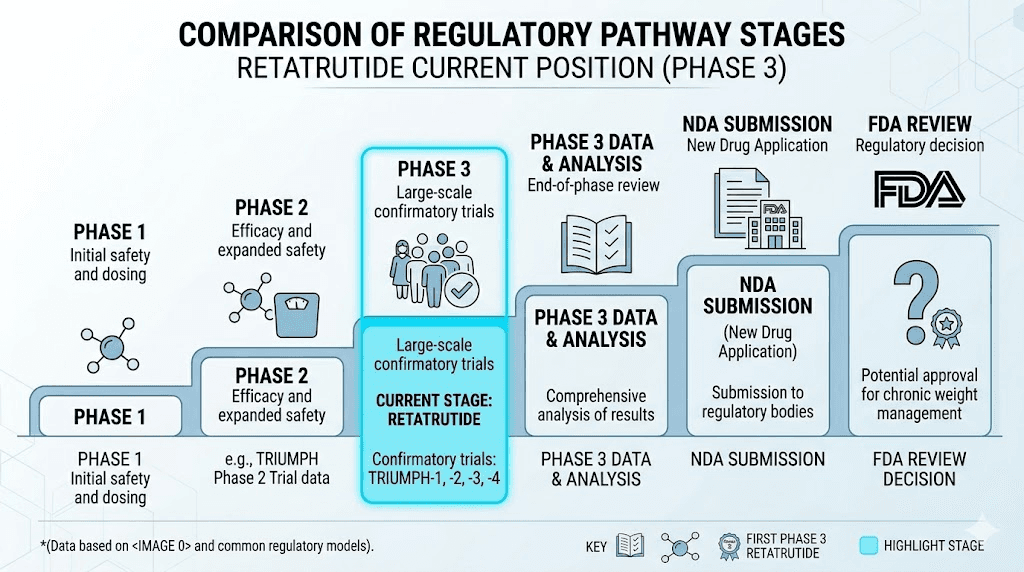
Where retatrutide stands in the regulatory pipeline
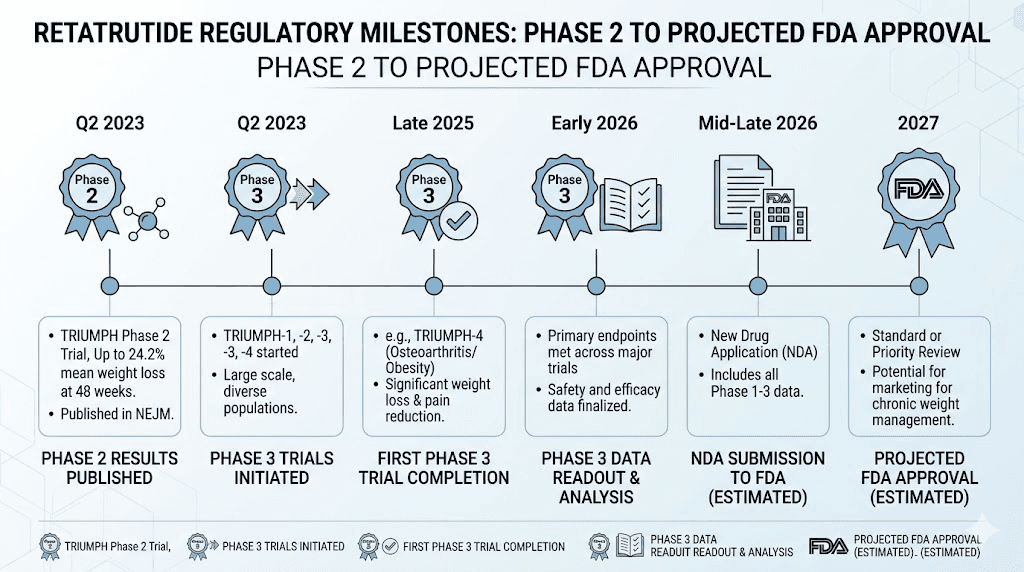
The regulatory pathway for a new drug follows a predictable sequence. Preclinical research comes first, followed by Phase 1 safety trials, Phase 2 efficacy trials, Phase 3 registration trials, NDA submission, FDA review, and finally approval or rejection. Retatrutide is currently in the Phase 3 stage, with one completed trial and several more expected to report throughout this year.
Specifically, here is the timeline so far:
Phase 1: Completed. Established basic safety and pharmacokinetic profile.
Phase 2: Published in the New England Journal of Medicine. Showed dose-dependent weight loss up to 24.2% at 48 weeks. This landmark study generated the initial excitement around the compound.
Phase 3 (TRIUMPH-4): Topline results announced December 2025. Showed 28.7% weight loss and significant osteoarthritis pain reduction.
Phase 3 (remaining TRIUMPH, TRANSCEND, SYNERGY trials): Results expected throughout 2026.
NDA submission: Not yet filed. Projected for late 2026 or early 2027, depending on trial results.
FDA review: Standard 10-month review period after NDA acceptance.
Potential approval: Realistic window is late 2027 to early 2028.
Eli Lilly cannot submit the NDA until enough Phase 3 data is available to demonstrate both efficacy and safety across the intended patient populations. One successful trial, TRIUMPH-4, is not sufficient. The FDA typically requires data from multiple Phase 3 studies covering different populations, doses, and endpoints before accepting a submission.
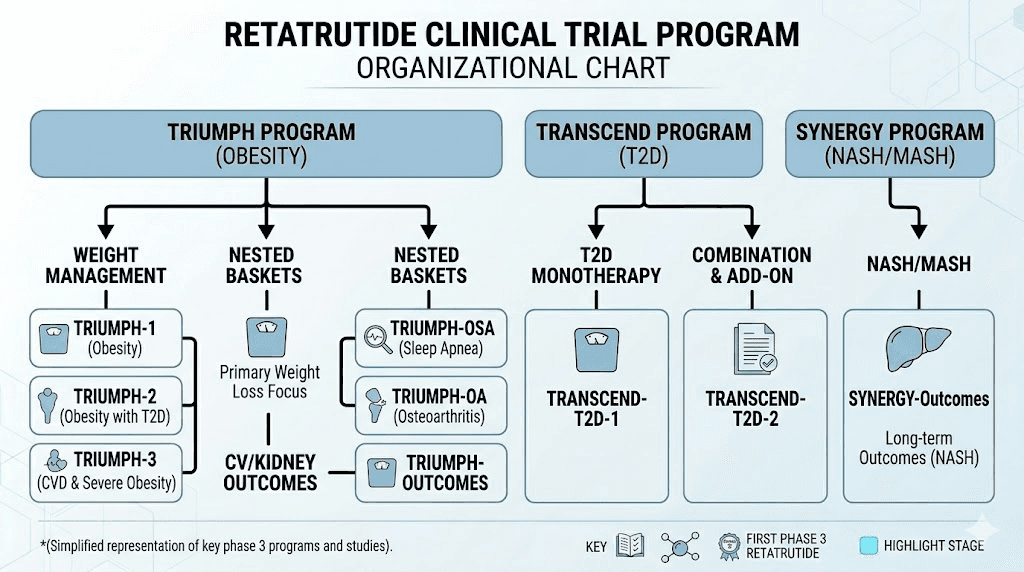
The TRIUMPH clinical trial program
The TRIUMPH program is the cornerstone of retatrutide's path to potential FDA approval. Understanding these trials, what they measure, and what the results mean, is essential for anyone tracking this compound.
TRIUMPH-1: obesity and overweight
TRIUMPH-1 (NCT05929066) is the main weight management trial. It evaluates retatrutide in adults with obesity or overweight, testing safety and efficacy at multiple dose levels. This trial also includes nested sub-studies evaluating participants with obstructive sleep apnea and knee osteoarthritis. The study enrolled participants across multiple sites and is expected to provide the primary efficacy data the FDA needs for the obesity indication.
Results from TRIUMPH-1 have not been publicly released as of April 2026. This is one of the most anticipated readouts, as it will provide the broadest picture of retatrutide's weight loss efficacy in a general obesity population.
TRIUMPH-2: type 2 diabetes with obesity
TRIUMPH-2 (NCT05929079) focuses on adults with both type 2 diabetes and obesity or overweight. This trial is critical because it evaluates retatrutide's dual benefits: weight reduction and glycemic control. A subset of participants with obstructive sleep apnea is also included.
The diabetes indication is significant for several reasons. First, the patient population is enormous. Second, existing GLP-1 medications like semaglutide already have approval for type 2 diabetes, so retatrutide would be entering a market with established competitors but potentially superior efficacy. Third, the combination of weight loss and blood sugar improvement addresses both conditions simultaneously, which is valuable for patients who qualify for treatment.
TRIUMPH-3: cardiovascular outcomes
TRIUMPH-3 (NCT05882045) evaluates retatrutide in people with obesity and existing cardiovascular disease. This trial is particularly important because cardiovascular outcome trials (CVOTs) are increasingly required by the FDA for metabolic medications. Semaglutide's SELECT trial demonstrated cardiovascular benefits, setting a new standard. Retatrutide needs similar data to compete.
The trial measures major adverse cardiovascular events (MACE), including heart attack, stroke, and cardiovascular death.
If retatrutide shows cardiovascular benefit similar to or better than semaglutide, it would significantly strengthen the case for approval and potentially expand the labeled indications.
TRIUMPH-4: knee osteoarthritis (completed)
TRIUMPH-4 is the first and, as of April 2026, the only Phase 3 trial to report results. The data announced in December 2025 was impressive:
Weight loss: Participants on the 12mg dose lost an average of 28.7% of their body weight (32.3 kg or 71.2 pounds) over 68 weeks. Starting from an average baseline of 112.7 kg (248.5 pounds, BMI 40.4), this represents transformative results.
Pain reduction: WOMAC pain scores dropped by up to 75.8% (4.5 points). More than 1 in 8 patients treated with retatrutide were completely free from knee pain by the end of the trial.
Cardiovascular markers: Reductions in non-HDL cholesterol and systolic blood pressure were observed, suggesting broader metabolic benefits beyond weight loss.
These results are particularly notable because they demonstrate that retatrutide's weight loss translates into meaningful clinical improvements, in this case, significant pain reduction for people with obesity-related joint problems.
Beyond TRIUMPH: the TRANSCEND and SYNERGY programs
Eli Lilly's retatrutide development extends beyond the TRIUMPH trials. Two additional Phase 3 programs target specific conditions:
TRANSCEND focuses on type 2 diabetes as the primary indication, separate from the obesity-focused TRIUMPH trials. While TRIUMPH-2 includes diabetes patients, TRANSCEND trials evaluate retatrutide specifically as a diabetes treatment.
SYNERGY targets metabolic dysfunction-associated steatotic liver disease (MASLD), previously known as non-alcoholic fatty liver disease (NAFLD). Phase 2 data showed retatrutide reduced liver fat by up to 82%, which is remarkable. The Phase 3 SYNERGY trial (NCT06859268) will evaluate whether retatrutide can resolve steatohepatitis (liver inflammation) and fibrosis, endpoints that would be required for regulatory approval specifically for liver disease treatment.
Together, the TRIUMPH, TRANSCEND, and SYNERGY programs represent one of the most comprehensive Phase 3 development programs in obesity pharmacotherapy. If successful across all indications, retatrutide could receive approval for weight management, type 2 diabetes, cardiovascular risk reduction, knee osteoarthritis, and liver disease.
How retatrutide compares to approved medications
Researchers naturally want to know how retatrutide measures against the GLP-1 medications already available. While direct head-to-head trials have not been conducted, cross-trial comparisons provide useful context.
Weight loss efficacy comparison
The numbers tell a clear story.
Semaglutide (2.4mg, Wegovy): Approximately 14.9% body weight reduction over 68 weeks in the STEP trials. This was considered groundbreaking when published. Semaglutide works reliably, but the ceiling appears to be around 15% for most patients.
Tirzepatide (15mg, Zepbound): Up to 22.5% body weight reduction over 72 weeks in the SURMOUNT trials. The dual GLP-1/GIP mechanism clearly outperformed single-agonist semaglutide. Tirzepatide weight loss results set a new benchmark.
Retatrutide (12mg): Up to 28.7% body weight reduction over 68 weeks in TRIUMPH-4. The triple-agonist approach produced the highest weight loss ever recorded in an obesity clinical trial. Network meta-analyses estimate approximately 23.77% average weight loss for retatrutide versus 16.79% for tirzepatide.
A simple comparison table puts this in perspective:
Medication | Receptors targeted | Max weight loss | Trial duration | FDA approved |
|---|---|---|---|---|
Semaglutide | GLP-1 | ~14.9% | 68 weeks | Yes |
Tirzepatide | GLP-1 + GIP | ~22.5% | 72 weeks | Yes |
Retatrutide | GLP-1 + GIP + Glucagon | ~28.7% | 68 weeks | No |
The progression is notable. Each additional receptor target correlates with greater efficacy. Whether this pattern continues with future compounds remains to be seen, but retatrutide represents the current peak of incretin-based weight loss pharmacotherapy.
Mechanism differences that matter
Beyond the weight loss numbers, the glucagon receptor component gives retatrutide properties that semaglutide and tirzepatide lack. Glucagon receptor activation increases energy expenditure, meaning the body burns more calories at rest. It also promotes hepatic fat oxidation, which explains the dramatic liver fat reduction seen in Phase 2 data (up to 82%).
This matters clinically. Many patients with obesity also have fatty liver disease. A medication that addresses both conditions simultaneously, rather than requiring separate treatments, has obvious advantages. Concerns about muscle loss with aggressive weight reduction are also relevant here. The increased energy expenditure from glucagon activation may help preserve lean mass compared to approaches that rely primarily on appetite suppression, though this hypothesis requires further study.
For researchers already familiar with retatrutide dosing protocols, the mechanism differences also explain why the titration schedule matters so much. Each receptor has its own dose-response curve, and finding the right balance between GLP-1, GIP, and glucagon activation requires careful escalation.
Safety profile and the dysesthesia signal
No discussion of retatrutide approval would be complete without addressing safety. The Phase 3 data revealed a new side effect that was not prominent in earlier trials, and it has become a focal point for both researchers and the FDA.
Common side effects from clinical trials
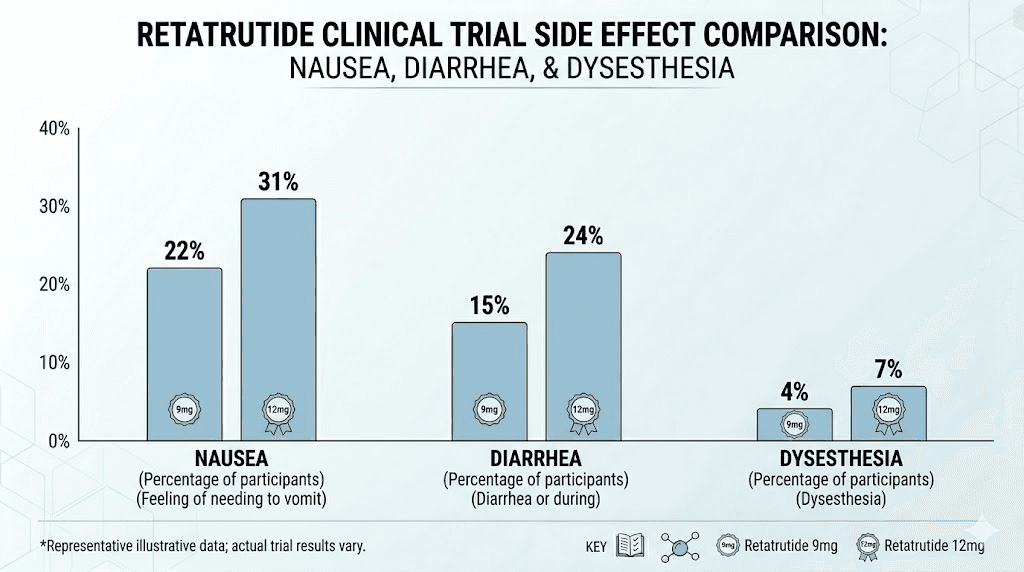
The most frequently reported adverse events with retatrutide mirror those of other GLP-1 based medications. Gastrointestinal symptoms dominate the side effect profile:
Nausea: The most commonly reported side effect, particularly during dose escalation. Consistent with the GLP-1 side effect profile seen across the class.
Diarrhea: Reported at higher rates with increasing doses. Usually self-limiting and manageable.
Vomiting: Less frequent than nausea but still notable, especially at higher doses.
Decreased appetite: Expected and, in the context of an obesity treatment, a therapeutic effect rather than a true side effect.
Constipation: Present but less prominent than with some other GLP-1 medications.
These gastrointestinal effects are generally dose-dependent, mild to moderate in severity, and tend to improve within 8 to 12 weeks as the body adjusts. They are consistent with what researchers already expect from GLP-1 class medications.
The dysesthesia signal: what researchers need to know
The more noteworthy finding from TRIUMPH-4 was dysesthesia. This is a type of abnormal skin sensation, tingling, burning, numbness, or heightened sensitivity to touch. At the highest dose (12mg), approximately 20.9% of participants reported this side effect. At 9mg, the rate was 8.8%.
This was not a significant finding in the Phase 2 trial, making it a genuinely new safety signal. The term covers a range of experiences. Some patients describe it as a tingling sensation. Others report that normal sensations, like clothing touching skin or water from a shower, feel unusual or mildly painful. The skin sensitivity with retatrutide appears to be related to the glucagon receptor component, which is the novel element not present in semaglutide or tirzepatide.
Several key points about the dysesthesia signal:
It is dose-dependent. The 12mg dose produced more than twice the rate of the 9mg dose, suggesting a clear relationship between glucagon receptor activation intensity and this side effect.
It is generally mild and self-limiting. Most cases resolved without treatment discontinuation. The events did not lead to significant dropout rates in the trial.
It warrants ongoing monitoring. Eli Lilly has acknowledged that dysesthesia requires continued surveillance across the remaining TRIUMPH trials. The FDA will certainly scrutinize this data when evaluating the NDA submission.
For researchers tracking retatrutide protocols, the dysesthesia data suggests that the 9mg dose may offer a better benefit-to-risk ratio than 12mg for some individuals. The weight loss difference between 9mg and 12mg exists but is smaller than the jump from lower doses, while the dysesthesia rate more than doubles.
A related study published in PMC examined allodynia and dysesthesia associated with GLP-1 medications more broadly, including semaglutide and tirzepatide. This suggests that skin sensitivity effects may be a class-wide phenomenon at some level, though the glucagon component in retatrutide clearly amplifies it.
How safety data affects the approval timeline
The dysesthesia signal will not necessarily prevent approval, but it will shape the labeling and potentially the approved dose range. The FDA frequently approves medications with known side effects as long as the benefit-risk ratio is favorable and the effects are well characterized.
What could delay approval is if the remaining TRIUMPH trials show higher rates of dysesthesia, more severe cases, or unexpected long-term consequences. The seven additional Phase 3 readouts expected this year will provide the larger safety database the FDA needs to make this determination.
Projected approval timeline
This is the question every researcher wants answered. When can we actually expect retatrutide to be available?
The realistic timeline
Based on the current state of the clinical program, regulatory precedent, and publicly available information from Eli Lilly, here is the most realistic projection:
Mid to late 2026: Remaining TRIUMPH, TRANSCEND, and SYNERGY Phase 3 results are reported. Seven major readouts are expected. These results will determine whether Lilly proceeds with filing.
Late 2026 to Q1 2027: NDA submission to the FDA, assuming all trials are successful. Lilly will need to compile the complete clinical data package, which takes several months after the last trial reports.
Q3 2027 to Q1 2028: FDA review period. The standard review timeline is 10 months from NDA acceptance. The PDUFA (Prescription Drug User Fee Act) date, which is the FDA's deadline to make a decision, would fall approximately 10 months after acceptance.
Late 2027 to early 2028: Potential FDA approval decision. If everything goes smoothly.
This timeline assumes no major setbacks. A failed trial, an unexpected safety signal, or a request for additional data from the FDA could push the timeline by 6 to 12 months or more.
What could speed things up
Priority Review designation from the FDA would shorten the review period from 10 months to 6 months. However, this designation is typically reserved for medications that offer a significant improvement over existing treatments for serious conditions. Given that semaglutide and tirzepatide are already approved for obesity, the FDA may not consider retatrutide sufficiently novel to warrant Priority Review.
Breakthrough Therapy designation, if granted, would facilitate more frequent FDA interactions and potentially accelerate the review. Lilly has not publicly announced whether they have sought or received this designation for retatrutide.
Exceptionally strong data from the remaining trials could also accelerate Lilly's internal timelines for NDA compilation and submission.
What could slow things down
Several factors could delay the projected timeline. If any of the remaining TRIUMPH trials fail to meet their primary endpoints, Lilly may need to redesign or repeat studies. If the dysesthesia rates are higher in the broader TRIUMPH population than in TRIUMPH-4, the FDA may request additional safety studies.
Manufacturing scale-up issues could also create delays. Producing a novel triple-agonist peptide at commercial scale requires specialized facilities and processes. Lilly has been investing heavily in manufacturing capacity, but any bottlenecks could push back the launch timeline even after approval.
A Complete Response Letter (CRL) from the FDA, essentially a request for more information or additional studies, would add at least 12 months to the timeline. While not expected based on current data, it remains a possibility.
Prediction markets and analyst estimates
As of early 2026, prediction markets place the probability of FDA approval this year at approximately 27%. This aligns with the realistic timeline described above. Most pharmaceutical analysts project approval in the 2027-2028 window.
Eli Lilly's own public statements have been measured. The company has confirmed the clinical program is on track but has not committed to specific filing dates. This is typical corporate caution, companies generally avoid making timeline commitments until they have complete Phase 3 data in hand.
What retatrutide approval would mean for the market
If approved, retatrutide would enter a rapidly evolving landscape of weight loss medications. Understanding the competitive dynamics helps researchers anticipate availability and access.
Market positioning
Retatrutide would likely be positioned as a premium option for patients who have not achieved adequate results with existing medications. With semaglutide plateau being a common concern and some patients finding that semaglutide stops working, a more potent alternative would fill a clear clinical need.
The additional liver disease indication, if the SYNERGY trials succeed, would open an entirely separate market. MASLD affects an estimated 30% of the global adult population, and currently has very limited treatment options. A medication that simultaneously treats obesity, diabetes, and liver disease would be unprecedented.
How it affects existing research
For researchers currently working with semaglutide or tirzepatide, retatrutide's approval would expand the toolkit significantly. The triple-agonist mechanism provides insights into metabolic pathways that single and dual agonists cannot access.
SeekPeptides members tracking the evolution of incretin-based therapies will find that retatrutide data also informs understanding of existing medications. The glucagon component, in particular, helps explain why some patients respond differently to GLP-1 therapy, and why medications sometimes stop working.
The comparison data between all three agents, semaglutide vs tirzepatide vs retatrutide, provides a framework for understanding dose-response relationships and receptor pharmacology that extends beyond any single compound.
Retatrutide dosing: what the trials used
Understanding the dosing protocols from clinical trials helps researchers contextualize the efficacy and safety data. It also provides insight into what the approved dosing might look like.
Phase 3 dose levels
TRIUMPH-4 tested three dose levels: 4mg, 9mg, and 12mg, all administered once weekly by subcutaneous injection. The dose escalation follows a titration schedule similar to other GLP-1 medications, starting low and increasing gradually over several weeks.
The retatrutide dose schedule typically involves starting at a low dose (often 1-2mg weekly) and increasing by small increments every 2-4 weeks until the target maintenance dose is reached. This gradual approach minimizes gastrointestinal side effects and allows the body to adjust to the medication.
For researchers interested in the mathematics of dosing, the retatrutide dosage calculator provides a useful tool. Understanding the relationship between vial concentration, injection volume, and target dose is essential for proper reconstitution and administration.
Efficacy by dose level
The TRIUMPH-4 data showed a clear dose-response relationship:
Dose | Weight loss at 68 weeks | Dysesthesia rate | GI side effects |
|---|---|---|---|
4mg | Moderate (specific % pending full publication) | Low | Mild |
9mg | Substantial | 8.8% | Moderate |
12mg | 28.7% | 20.9% | Moderate-High |
The 4mg dose is also being evaluated as a maintenance dose in some trials. The concept is that patients could achieve significant weight loss on higher doses and then step down to 4mg to maintain results with fewer side effects. This mirrors the maintenance dosing strategy being explored with tirzepatide and other medications.
Reconstitution and administration
Clinical trials used pre-filled syringes, but research-grade retatrutide peptide requires proper reconstitution. For researchers working with lyophilized retatrutide, the reconstitution process involves adding bacteriostatic water to the vial and following standard peptide preparation protocols.
Key details for researchers:
The amount of bacteriostatic water determines the concentration. For a 10mg vial, 2mL of bac water produces a 5mg/mL solution.
Storage after reconstitution requires refrigeration at 2-8 degrees Celsius. Reconstituted retatrutide remains stable for approximately 28-42 days when properly stored.
Injection technique follows standard subcutaneous protocols, with the abdomen, thigh, and upper arm as common injection sites.
The peptide reconstitution calculator helps ensure accurate concentration calculations.
Phase 2 trial: the study that started it all
The Phase 2 trial, published in the New England Journal of Medicine under the title "Triple Hormone Receptor Agonist Retatrutide for Obesity," is the foundation upon which the entire Phase 3 program was built. Understanding this trial explains why the pharmaceutical industry and research community became so excited about this compound.
Study design
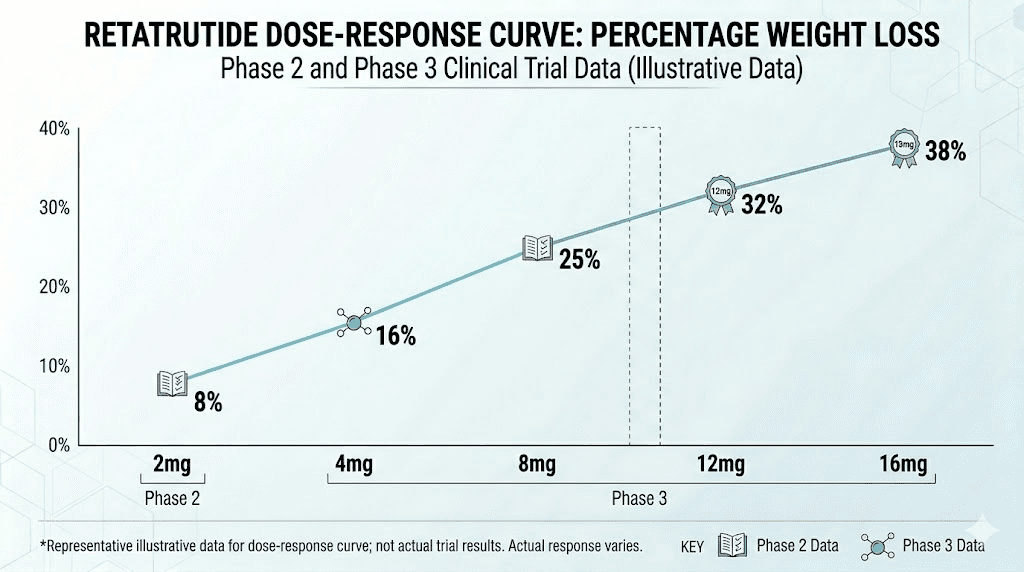
The Phase 2 trial was a randomized, double-blind, placebo-controlled study that enrolled 338 adults with obesity (BMI 30 or higher) or overweight (BMI 27 or higher) with at least one weight-related condition. Participants were randomized to placebo or one of several retatrutide dose groups: 1mg, 4mg, 8mg, or 12mg, administered weekly by subcutaneous injection.
The trial duration was 48 weeks, with a primary endpoint of percent change in body weight from baseline.
Key results
The results were dose-dependent and remarkable at every level:
1mg: Approximately 8.7% weight loss
4mg: Approximately 17.1% weight loss
8mg: Approximately 22.8% weight loss
12mg: Approximately 24.2% weight loss
Placebo: Approximately 2.1% weight loss
The 24.2% weight loss at 12mg over 48 weeks was unprecedented. No anti-obesity medication had ever produced results this dramatic in a clinical trial at that point. For context, this exceeds what semaglutide achieves in 68 weeks, and retatrutide did it in a shorter timeframe.
Additional findings
Beyond weight loss, the Phase 2 trial revealed several additional benefits. Improvements in HbA1c (blood sugar control) were significant, even in participants without diabetes. Liver fat reduction was dramatic, with up to 82% reduction in hepatic fat content among participants with fatty liver at baseline. This led directly to the SYNERGY liver disease trials.
Improvements in cardiovascular risk markers, including cholesterol, triglycerides, and blood pressure, were also observed. These findings supported the inclusion of the TRIUMPH-3 cardiovascular outcomes trial in the Phase 3 program.
What researchers are watching in the remaining trials
With seven Phase 3 readouts expected this year, researchers have specific questions they want answered. The data from these trials will determine not just whether retatrutide gets approved, but how it gets approved, which indications, what doses, and what safety warnings.
The TRIUMPH-1 readout
This is arguably the most important remaining trial. TRIUMPH-1 will provide the primary efficacy data for the general obesity indication. If the results match or exceed TRIUMPH-4 (28.7% weight loss), the case for approval becomes very strong. If the results are significantly lower, questions will arise about the TRIUMPH-4 population being particularly responsive due to the osteoarthritis component.
Researchers are also watching for the obstructive sleep apnea (OSA) sub-study results within TRIUMPH-1. OSA is extremely common in people with obesity, and a treatment that simultaneously reduces weight and improves sleep apnea could earn a specific labeled indication.
Safety data across larger populations
TRIUMPH-4 enrolled a specific population (obesity with knee osteoarthritis). The remaining trials cover broader and different populations. Researchers need to see whether the dysesthesia rate holds at approximately 20.9% for the 12mg dose across all populations, or whether certain groups are more or less susceptible.
The fatigue profile is also being monitored. Fatigue with retatrutide was reported in earlier studies, and the larger Phase 3 database will better characterize its frequency and severity.
Liver disease data from SYNERGY
The SYNERGY MASLD trial could be the most commercially significant readout. If retatrutide demonstrates resolution of steatohepatitis and improvement in liver fibrosis, it would be one of the first medications approved specifically for this condition. The market potential is enormous, given that MASLD affects roughly 30% of adults globally.
The Phase 2 liver data showing 82% fat reduction provides strong rationale, but the Phase 3 endpoints are more demanding. The FDA wants to see actual resolution of liver inflammation and fibrosis improvement, not just fat reduction. These are harder endpoints to achieve but more clinically meaningful.
Long-term maintenance data
Some TRIUMPH trials include extensions evaluating maintenance dosing. Specifically, the 4mg dose is being tested as a step-down option after initial weight loss on higher doses. Weight regain after discontinuation is a major concern with all GLP-1 medications. Getting off GLP-1 medications often leads to significant weight rebound, and researchers want to know if a lower maintenance dose of retatrutide can prevent this.
The concept of dose stepping, using a higher dose for initial weight loss and then transitioning to a lower dose for maintenance, could become the standard treatment paradigm if the data supports it. This approach would reduce side effects, lower cost, and potentially improve long-term adherence.
Retatrutide in the broader GLP-1 landscape
Retatrutide does not exist in a vacuum. Understanding where it fits within the rapidly expanding GLP-1 medication landscape helps researchers assess its significance and potential impact.
The evolution from single to triple agonism
The trajectory of incretin-based obesity pharmacotherapy shows a clear pattern of increasing complexity and efficacy:
First generation (single agonist): Liraglutide (Saxenda) and semaglutide (Wegovy) target GLP-1 only. These GLP-1 medications established the category and proved that receptor agonists could produce meaningful, sustained weight loss.
Second generation (dual agonist): Tirzepatide (Zepbound/Mounjaro) targets GLP-1 and GIP. The addition of GIP produced approximately 50% more weight loss than GLP-1 alone. The jump from tirzepatide to retatrutide follows this same pattern.
Third generation (triple agonist): Retatrutide targets GLP-1, GIP, and glucagon. The addition of glucagon produced approximately 25-30% more weight loss than dual agonism. This is where we stand now.
The pattern suggests that each additional receptor target provides incremental but meaningful improvements. However, it also introduces additional complexity and potential side effects, as the dysesthesia signal demonstrates.
Competitors in development
Retatrutide is not the only next-generation obesity medication in development. Several compounds are also in late-stage trials:
Survodutide (Boehringer Ingelheim) is a dual GLP-1/glucagon agonist in Phase 3 trials. It targets two of the three receptors that retatrutide hits but through a different molecular approach.
CagriSema (Novo Nordisk) combines semaglutide with cagrilintide, an amylin analog. This is a different approach entirely, combining a GLP-1 agonist with an amylin receptor agonist rather than adding GIP or glucagon.
Mazdutide (Innovent Biologics) is another dual GLP-1/glucagon agonist showing promising results in Chinese patient populations.
Orforglipron (Eli Lilly) is a small molecule oral GLP-1 agonist. While less potent than injectable options, the convenience of oral administration could make it a preferred first-line option. Comparing orforglipron to tirzepatide reveals a tradeoff between convenience and efficacy.
The competitive landscape means that by the time retatrutide reaches the market, it will compete not just with current medications but with several of these newer agents as well.
The role of research peptides
Because retatrutide is not yet approved, researchers who want to study it currently rely on research-grade peptide. Several vendors offer retatrutide for research purposes, and SeekPeptides provides comprehensive resources for understanding where to obtain research retatrutide, current pricing, and availability information.
Understanding the difference between research-grade and pharmaceutical-grade peptides is critical for researchers. Research peptides are not manufactured under the same GMP conditions as pharmaceutical products and are not intended for human use. The purity, stability, and consistency may vary between batches and vendors.
Research applications and ongoing studies
Beyond the TRIUMPH program, retatrutide is being studied in several contexts that expand understanding of triple agonism and metabolic pharmacology.
Body composition research
One of the most important questions in obesity pharmacotherapy is whether weight loss preserves or degrades lean body mass. With muscle preservation during GLP-1 therapy being a major concern, retatrutide's glucagon component is of particular interest. Glucagon promotes fat oxidation specifically, which theoretically could shift the weight loss composition toward greater fat loss and less muscle loss compared to pure appetite-suppressing approaches.
Some researchers are investigating whether combining retatrutide with creatine supplementation or resistance training can further optimize body composition outcomes. The interaction between exercise and GLP-1 therapy is an active area of investigation.
Cardiovascular research
The TRIUMPH-3 cardiovascular outcomes trial is the most direct study of retatrutide's heart effects. However, researchers are also examining secondary cardiovascular data from other trials. The improvements in non-HDL cholesterol, blood pressure, and inflammatory markers observed in TRIUMPH-4 suggest broad cardiovascular benefits.
For researchers monitoring cardiac parameters, understanding the cardiovascular effects of GLP-1 medications provides important context. The glucagon receptor component in retatrutide adds a variable not present in existing medications, making cardiovascular monitoring particularly important.
Liver disease research
The 82% liver fat reduction from Phase 2 represents some of the most dramatic results in MASLD research. Current treatments for liver disease are extremely limited, with only one drug (resmetirom) recently approved specifically for MASH. If retatrutide can demonstrate liver inflammation resolution and fibrosis improvement in Phase 3, it would represent a major advance.
Researchers working in the gut health and metabolic space are particularly interested in how the triple-agonist approach affects liver-gut axis signaling. The combination of improved glucose metabolism, reduced hepatic fat, and enhanced fat oxidation creates a multi-pathway approach to liver disease that monotherapy cannot replicate.
Autoimmune and inflammatory research
Emerging research suggests GLP-1 receptor agonists may have anti-inflammatory properties beyond their metabolic effects. Microdosing GLP-1 for autoimmune conditions is an area of growing interest, and retatrutide's triple-receptor approach may amplify these anti-inflammatory effects.
Glucagon receptor activation influences hepatic inflammation and immune cell function. Some preclinical data suggests this could be beneficial in conditions characterized by chronic low-grade inflammation, which includes most metabolic diseases. Whether this translates into clinical benefit for autoimmune conditions remains speculative but is being investigated.
Practical considerations for researchers
For researchers interested in retatrutide specifically, or in the broader field of incretin-based pharmacology, several practical considerations apply.
Working with research-grade retatrutide
Research peptide requires careful handling. Standard vial sizes include 5mg, 10mg, and 20mg. Proper reconstitution technique is essential to maintain peptide integrity. Key considerations:
Store lyophilized (unreconstituted) peptide at -20 degrees Celsius or colder for long-term storage.
Use bacteriostatic water for reconstitution, not sterile water, if the vial will be used multiple times.
Inject the water slowly down the side of the vial. Never shake the vial, as this can denature the peptide.
After reconstitution, store at 2-8 degrees Celsius and use within 28-42 days.
The reconstitution calculator ensures accurate concentration preparation.
Cost considerations
Research-grade retatrutide is currently more expensive than research-grade semaglutide or tirzepatide, reflecting its novelty and manufacturing complexity. Current retatrutide pricing varies by vendor and quantity. The peptide cost calculator can help researchers compare options.
If approved, the pharmaceutical version will likely be priced at a premium to tirzepatide, which itself is more expensive than semaglutide. The exact pricing strategy will depend on the approved indications, competitor pricing at the time of launch, and insurance coverage decisions.
Tracking updates
The retatrutide approval timeline will become clearer as Phase 3 results are reported throughout this year. Researchers should monitor:
Eli Lilly investor relations page for trial result announcements
ClinicalTrials.gov for individual trial status updates
FDA announcements for any regulatory interactions
Medical conferences (ADA, ObesityWeek, EASD) where data may be presented
SeekPeptides provides ongoing coverage of retatrutide developments, including dosing guides, availability updates, and comparison analyses that help researchers stay current with the latest information.
Understanding the regulatory process
For researchers less familiar with pharmaceutical regulation, a brief overview of the FDA approval process helps contextualize where retatrutide stands and what comes next.
Pre-NDA meeting
Before submitting the NDA, Eli Lilly will hold a Pre-NDA meeting with the FDA to discuss the submission package. This meeting typically covers which studies to include, what data the FDA considers sufficient, and any specific concerns about safety or efficacy. The outcome of this meeting often determines the submission timeline.
NDA submission
The NDA package will include all preclinical data, Phase 1, 2, and 3 trial results, manufacturing information, proposed labeling, and risk management plans. For a compound with as many ongoing trials as retatrutide, deciding which data to include and which to reference as pending is a strategic decision.
FDA review
During the standard 10-month review period, FDA reviewers examine the clinical data, statistical analyses, safety database, manufacturing processes, and proposed labeling. Advisory committee meetings may be convened to get external expert opinions, particularly if the safety profile raises questions (as the dysesthesia signal might).
PDUFA date and decision
The PDUFA date is the deadline by which the FDA must make a decision. The outcome can be approval, a Complete Response Letter (requesting more information), or rejection. For retatrutide, approval with specific labeling around the dysesthesia risk and recommended monitoring is the most likely positive outcome.
What happens after potential approval
Assuming retatrutide eventually receives FDA approval, several practical questions arise for the research and clinical communities.
Initial launch and availability
New drug launches typically start with limited availability, especially for injectable medications that require manufacturing scale-up. Eli Lilly has been investing in manufacturing capacity, including a $4 billion expansion of its manufacturing facilities, but initial supply constraints are still possible.
The experience with tirzepatide (Mounjaro/Zepbound) is instructive. Demand exceeded supply for the first several months after launch, leading to periodic shortages. Finding alternatives during shortages became a common research question. Retatrutide may face similar challenges.
Insurance coverage
Coverage for obesity medications remains inconsistent. While some insurers are expanding GLP-1 coverage, others continue to exclude weight loss medications. Coverage varies by plan and indication. If retatrutide is approved for multiple indications (obesity, diabetes, liver disease, osteoarthritis), coverage may be easier to obtain under non-obesity indications.
Researchers should also be aware that HSA and FSA eligibility for GLP-1 medications is evolving and may apply to retatrutide if approved.
Compounding considerations
Existing GLP-1 medications like semaglutide and tirzepatide are available through compounding pharmacies, which offer compounded versions at lower cost. Whether retatrutide will be available through compounding depends on its patent status, whether it appears on the FDA drug shortage list, and regulatory decisions about compounding rights.
Given Lilly's aggressive patent protection strategy (as seen with tirzepatide), affordable retatrutide options through compounding may be limited initially. The research peptide market would continue to serve researchers who need access before or alongside the pharmaceutical version.
Frequently asked questions
Is retatrutide FDA approved?
No. As of April 2026, retatrutide is not FDA approved. Eli Lilly has not submitted a New Drug Application. The compound remains in Phase 3 clinical trials, with multiple studies expected to report results throughout this year. The earliest realistic approval window is late 2027 to early 2028.
When will retatrutide be available to the public?
If all Phase 3 trials succeed and the NDA is submitted in late 2026, FDA approval could come in late 2027 to early 2028. After approval, manufacturing scale-up and insurance negotiations typically add several additional months before widespread availability. Researchers estimate early to mid 2028 as the earliest likely date for broad public access.
How much weight can you lose on retatrutide?
Phase 3 data (TRIUMPH-4) showed average weight loss of 28.7% body weight at the 12mg dose over 68 weeks, equivalent to approximately 71.2 pounds from an average starting weight of 248.5 pounds. Phase 2 data showed up to 24.2% weight loss at 48 weeks. Individual results vary based on dose, adherence, diet, and exercise habits.
Is retatrutide better than semaglutide or tirzepatide?
In terms of weight loss efficacy, clinical trial data shows retatrutide produces greater average weight loss than both semaglutide and tirzepatide. However, "better" depends on the individual. Retatrutide has a different side effect profile (including dysesthesia), is not yet approved, and may not be necessary for patients who respond well to existing medications. Each additional receptor target brings both increased efficacy and increased complexity.
What are the side effects of retatrutide?
Common side effects include nausea, diarrhea, vomiting, decreased appetite, and constipation, similar to other GLP-1 medications. A new side effect, dysesthesia (abnormal skin sensations like tingling or burning), emerged in Phase 3 trials at rates of 8.8% (9mg) to 20.9% (12mg). Skin sensitivity is believed to be related to the glucagon receptor component.
Can you buy retatrutide now?
Research-grade retatrutide peptide is available from peptide vendors for research purposes. This is not the same as pharmaceutical-grade retatrutide and is not intended for human use. The pharmaceutical version will not be available until after FDA approval. Purchasing retatrutide online requires understanding the difference between research and pharmaceutical peptides.
How does retatrutide work differently from tirzepatide?
Tirzepatide targets two receptors (GLP-1 and GIP). Retatrutide targets three (GLP-1, GIP, and glucagon). The glucagon receptor activation increases energy expenditure, promotes liver fat oxidation, and contributes to greater overall weight loss. This additional mechanism also introduces the dysesthesia side effect not seen with tirzepatide. For detailed comparison, see the tirzepatide vs retatrutide guide.
What dose of retatrutide will be prescribed?
Based on Phase 3 data, the likely approved doses will be 4mg, 9mg, and 12mg weekly. The dosing schedule will involve gradual titration from a low starting dose. The 4mg dose may serve as a maintenance dose after initial weight loss on higher doses. Final approved dosing will be determined by the complete Phase 3 data set.
External resources
In case I do not see you, good afternoon, good evening, and good night. May your research stay rigorous, your data stay clean, and your patience with regulatory timelines stay intact.