Mar 24, 2026

Experienced researchers know something about retatrutide sourcing that newcomers learn the hard way. Not all vendors are equal. Not all products perform the same. And the difference between a reliable source and a questionable one can mean the difference between meaningful research data and months of wasted effort. Simple Peptide has positioned itself as a domestic option for retatrutide research compounds, but positioning and performance are two very different things.
The challenge with retatrutide is that it represents a genuinely novel class of compound. As a triple agonist targeting GLP-1, GIP, and glucagon receptors simultaneously, the purity demands are higher than single-target peptides. Contamination or degradation affects not one pathway but three. That makes vendor selection particularly important for anyone working with this compound, whether in metabolic research, receptor binding studies, or comparative analyses against semaglutide and tirzepatide.
This guide examines Simple Peptide from every angle that matters to researchers. Their product catalog, manufacturing claims, testing protocols, pricing structure, shipping reliability, and how they stack up against established alternatives. We will look at what the company gets right, where questions remain, and what you should verify before placing an order. SeekPeptides has reviewed dozens of peptide vendors, and Simple Peptide deserves the same thorough treatment.
What is Simple Peptide?
Simple Peptide operates under the legal name Melex Technologies. The company launched with a straightforward premise: make lab-tested research peptides accessible at reasonable prices. Their tagline, "Peptides Made Simple," reflects an approach that prioritizes ease of ordering over the complexity that defines many research chemical suppliers.
The company is based in the United States with a Florida contact number (561-501-1570). They maintain an active online presence across multiple social media platforms and operate exclusively through their website at simplepeptide.com. Account registration is required before browsing their full product catalog, which some researchers view as a minor inconvenience while others see it as a privacy measure.
What sets Simple Peptide apart from many competitors is their claim of domestic manufacturing. According to the company, all products are synthesized in a US-based laboratory following cGMP (current Good Manufacturing Practice) protocols. Each batch undergoes independent third-party testing for purity and potency verification. These claims align with industry best practices for peptide quality assurance, though independent verification remains the responsibility of each researcher.
Trust metrics paint a generally positive picture. ScamAdviser rates the domain at 95 out of 100 for safety, and customer reviews across platforms like Trustpilot and RatingFacts trend positive. Researchers report reliable shipping, accurate product descriptions, and responsive customer service. The company has been operational since 2019, giving it a moderate track record in an industry where many vendors appear and disappear within months.
Simple Peptide already has an established presence in the tirzepatide research market. Their expansion into retatrutide products represents a natural evolution as demand for triple agonist compounds continues growing among the research community.
Simple Peptide retatrutide products
Simple Peptide currently offers two retatrutide-related products for research purposes. Each targets a different research application, and understanding the distinction matters for protocol design.
GLP-3RT (retatrutide)
The primary retatrutide offering from Simple Peptide is marketed under the designation GLP-3RT. This is a synthetic tri-agonist peptide designed for laboratory research focused on multi-pathway metabolic signaling. The product interacts with three key receptor pathways: GLP-1, GIP, and glucagon receptors.
GLP-3RT comes in a 3mL vial format. The product is labeled strictly for Research Use Only (RUO), meaning it is intended exclusively for laboratory, investigational, and scientific research purposes. It is not approved by the FDA and is not intended for human or veterinary use, clinical applications, diagnostic procedures, or therapeutic treatment.
For researchers comparing this to other retatrutide sources, the GLP-3RT designation is simply Simple Peptide branding for what the research community broadly calls retatrutide or LY3437943 (the Eli Lilly investigational compound identifier). The underlying amino acid sequence and mechanism remain the same regardless of vendor naming conventions.
Reta/Cag blend (retatrutide plus cagrilintide)
The second product is a combination compound pairing retatrutide with cagrilintide at a 12.5mg/2.5mg ratio. This blend targets researchers studying the interaction between triple agonist signaling and amylin receptor agonism.
Cagrilintide is a long-acting amylin analog that has shown promise in weight management research. Combining it with retatrutide creates a four-pathway research compound, which is a relatively novel offering in the peptide vendor space. Few suppliers currently offer this specific combination, making it a differentiator for Simple Peptide.
The rationale behind this blend draws from emerging research on cagrilintide and retatrutide co-administration. Amylin works through appetite regulation and gastric emptying pathways that complement but do not duplicate the mechanisms of GLP-1, GIP, and glucagon activation. Researchers studying cagrilintide safety profiles alongside triple agonist compounds may find this pre-blended format convenient for controlled studies.
Both products ship domestically within the United States. Simple Peptide emphasizes fast processing times, though specific delivery windows depend on location and shipping method selected at checkout.
Understanding retatrutide: the science behind the triple agonist
Before evaluating any vendor, understanding what you are purchasing matters. Retatrutide is not just another GLP-1 peptide. It represents a fundamentally different approach to metabolic receptor research.
Triple receptor mechanism
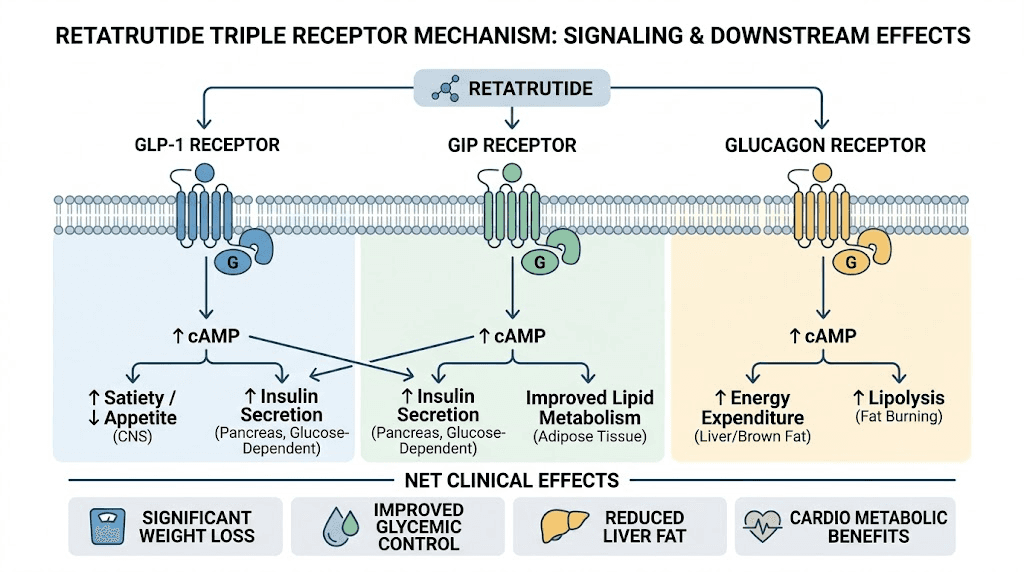
Most weight management peptides target one or two receptors. Semaglutide activates GLP-1 receptors exclusively. Tirzepatide targets both GLP-1 and GIP receptors, which earned it the "dual agonist" designation. Retatrutide goes further by simultaneously engaging three receptor systems.
GLP-1 receptor activation slows gastric emptying, enhances insulin secretion, and suppresses glucagon release from alpha cells. This is the mechanism that drives appetite suppression in GLP-1 compounds. GIP receptor activation amplifies insulin response and appears to play a role in fat metabolism that researchers are still fully characterizing. The glucagon component is what makes retatrutide truly unique.
Glucagon receptor activation increases energy expenditure, promotes hepatic fat oxidation, and drives lipolysis. This seems counterintuitive. Glucagon raises blood sugar. Why would activating it benefit metabolic research? The answer lies in balance. When GLP-1 and GIP agonism handle glucose regulation, the glucagon component can drive its metabolic benefits without the hyperglycemic risk. The three pathways work in concert rather than competition.
This mechanism explains why retatrutide shows different outcomes than semaglutide in research settings and why comparisons with tirzepatide consistently show the triple agonist producing more pronounced effects across multiple endpoints.
Clinical trial evidence
The evidence base for retatrutide is substantial and growing. The Phase 2 clinical trial, published in the New England Journal of Medicine, demonstrated remarkable outcomes. Participants receiving the highest doses achieved up to 24.2% mean body weight reduction over 48 weeks. In participants with type 2 diabetes, HbA1c improved by 2.2%, with 82% of participants reaching HbA1c levels at or below 6.5%.
Those numbers are significant. For context, semaglutide typically produces 15-17% weight reduction over similar timeframes, and tirzepatide achieves roughly 20-22%. Retatrutide consistently outperformed both in head-to-head comparisons.
The Phase 3 TRIUMPH-4 trial, announced by Eli Lilly, delivered even more dramatic results. Participants with obesity and knee osteoarthritis experienced average weight loss of 71.2 pounds alongside substantial relief from osteoarthritis pain. Additional Phase 3 results evaluating maintenance doses are expected throughout the remainder of the research program.
Perhaps the most striking finding involves liver fat. In the MASLD (metabolic dysfunction-associated steatotic liver disease) trial, retatrutide reduced liver fat by up to 82.4% at the 12mg dose level. The 8mg dose achieved 81.4% reduction. Even the 4mg dose produced a 57% reduction. Placebo showed essentially no change at 0.3%. These liver fat numbers exceed what any single-target or dual-target compound has demonstrated in comparable studies.
The safety profile was consistent with GLP-1 class compounds. Gastrointestinal side effects including nausea, diarrhea, vomiting, and constipation occurred at higher rates than placebo, particularly at higher doses. Nausea was the most frequently reported adverse event across all dose levels. These findings align with what researchers observe when studying GLP-1 and GIP agonist side effect profiles.
Quality and testing standards at Simple Peptide
Quality verification is the single most important factor when selecting a retatrutide vendor. A compound that targets three receptor systems demands exceptional purity. Contaminants or degradation products can bind unpredictably across receptor subtypes, creating noise in research data that is nearly impossible to troubleshoot after the fact.
Manufacturing claims
Simple Peptide states that all products are manufactured in a US-based laboratory following cGMP protocols. This is a meaningful distinction. Many research peptide vendors source their products from overseas facilities where manufacturing standards vary widely. Domestic synthesis under cGMP conditions implies standardized equipment calibration, environmental controls, documented procedures, and quality management systems.
The synthesis method described involves solid-phase peptide synthesis (SPPS), modern post-synthesis cleavage, and multi-stage purification. SPPS is the gold standard for peptide manufacturing and is the same approach used by Peptide Sciences and other established vendors. Multi-stage purification typically involves HPLC (high-performance liquid chromatography) to separate the target peptide from truncated sequences, deletion peptides, and other synthesis byproducts.
For retatrutide specifically, the synthesis complexity is higher than simple peptides. The compound contains multiple receptor-binding domains that must fold correctly. Purification must remove not only incomplete sequences but also misfolded variants that could produce unpredictable receptor binding patterns.
Third-party testing and COA verification
Simple Peptide claims each batch undergoes independent third-party testing for purity and potency. Third-party testing means an external, accredited laboratory with no financial stake in the product performs the analysis. This removes the conflict of interest inherent in self-testing.
When evaluating any vendor COA (Certificate of Analysis), researchers should verify several key elements. HPLC purity percentage indicates the proportion of the sample that is the target peptide versus other peptide-like impurities. Mass spectrometry confirmation verifies that the molecular weight matches the expected value for retatrutide. Net peptide content tells you how much of the vial weight is actual peptide versus water, salts, and other non-active components.
A robust COA will also include retention time data from HPLC analysis, which provides an additional identity confirmation. Some advanced COAs include amino acid analysis or sequencing data, though these are less common from research peptide vendors.
Researchers working with retatrutide from Simple Peptide should request lot-specific COAs before beginning any research protocol. Compare the reported molecular weight to the known value for retatrutide (approximately 4113.6 Da). Verify that HPLC purity exceeds 95% at minimum, with 98% or higher being preferred for receptor binding studies. Check that the testing laboratory is identified by name, not just described generically.
For additional guidance on evaluating vendor quality documentation, SeekPeptides provides comprehensive resources on peptide testing laboratories and quality verification.
What independent testing shows
Finnrick Analytics, an independent testing service that evaluates peptide vendors, maintains ratings for retatrutide across 143 vendors. Their database provides a useful benchmark for comparing Simple Peptide against the broader market. While individual test results for Simple Peptide from Finnrick were not available at the time of this review, the platform offers researchers a way to verify claims independently.
The research community generally recommends purchasing from vendors who provide batch-specific COAs, respond to quality inquiries, and maintain consistent product availability. Simple Peptide checks these boxes based on customer reports, though researchers should always conduct their own verification for critical studies.
How Simple Peptide compares to other retatrutide vendors
No vendor review is complete without context. How does Simple Peptide stack up against the alternatives? The retatrutide vendor landscape includes established names with long track records alongside newer entrants competing on price and service.
Vendor comparison table
Vendor | Product format | Manufacturing | Third-party testing | Track record | Overall |
|---|---|---|---|---|---|
Simple Peptide | GLP-3RT vial, Reta/Cag blend | US-based cGMP | Claimed, batch-specific | Since 2019 | 7/10 |
Lyophilized powder | US-based | In-house and third-party | 10+ years | 7/10 | |
Lyophilized powder | US-based | Third-party verified | Established | 8/10 | |
Varies | Mixed | Varies widely | Varies | 4-7/10 |
Paradigm Peptides consistently scores highest in independent testing databases, with an A rating based on over 2,100 laboratory tests. Peptide Sciences has the longest track record but has received mixed ratings in some independent analyses for retatrutide specifically. Simple Peptide occupies a middle position, offering competitive quality with the added benefit of unique product formats like the Reta/Cag blend.
The choice between vendors often comes down to specific research needs. Researchers requiring standard retatrutide for receptor binding studies might prioritize the vendor with the highest independent purity scores. Those studying cagrilintide and retatrutide interactions may find Simple Peptide unique blend offering more convenient than sourcing and mixing components separately.
For researchers exploring the broader vendor landscape, our guides on the best peptide vendors and where to buy retatrutide provide comprehensive comparisons across dozens of suppliers.

Retatrutide dosing protocols from clinical research
Understanding dosing protocols from published clinical trials helps researchers design appropriate study parameters. These protocols reflect the doses that produced meaningful outcomes in controlled settings.
Phase 2 dose escalation protocol
The Phase 2 trial evaluated multiple dose levels ranging from 0.5mg to 12mg administered once weekly via subcutaneous injection. The dose escalation schedule followed a gradual increase pattern designed to minimize gastrointestinal side effects while allowing researchers to identify optimal therapeutic ranges.
The starting dose for most treatment arms was 0.5mg weekly for the first two weeks. This is consistent with the approach used for tirzepatide starting doses and semaglutide initial dosing. From there, doses increased in 2-week intervals until reaching the target maintenance dose.
The dose levels tested included 1mg, 4mg, 8mg, and 12mg weekly. The 12mg group showed the most pronounced effects across all measured endpoints, but the 8mg group demonstrated a favorable balance between efficacy and tolerability. For researchers designing protocols, the retatrutide dose schedule guide provides detailed escalation timelines.
Reconstitution considerations
When working with lyophilized retatrutide from Simple Peptide or any vendor, proper reconstitution technique directly affects research outcomes. The general protocol involves adding bacteriostatic water slowly along the vial wall, allowing the lyophilized cake to dissolve without agitation.
For a standard 10mg retatrutide vial, the amount of bacteriostatic water added determines the concentration and, consequently, the volume per dose. Researchers use the retatrutide reconstitution chart to calculate exact volumes based on their target dose and preferred concentration.
Simple Peptide GLP-3RT product comes in a 3mL vial format, which may differ from the standard lyophilized powder vials that most peptide reconstitution guides reference. Researchers should confirm the product format and adjust their preparation protocol accordingly. The peptide reconstitution calculator at SeekPeptides can help determine exact volumes regardless of starting format.
Dosage reference from research literature
Based on published clinical data, here is a summary of the dose-dependent outcomes observed in controlled research settings:
Weekly dose | Weight reduction (48 weeks) | Liver fat reduction (24 weeks) | GI side effects |
|---|---|---|---|
1mg | ~8.7% | -42.9% | Mild |
4mg | ~17.1% | -57.0% | Moderate |
8mg | ~22.8% | -81.4% | Moderate to significant |
12mg | ~24.2% | -82.4% | Significant |
The diminishing returns between 8mg and 12mg for both weight and liver fat endpoints are noteworthy. The 8mg dose achieves approximately 94% of the weight effect and 99% of the liver fat effect seen at 12mg, but with meaningfully fewer side effects. This observation has influenced how researchers design their retatrutide dosing protocols.
For researchers transitioning from other compounds, the retatrutide dosage schedule after tirzepatide and switching protocols provide guidance on cross-compound transitions based on available research data.
Storage and handling for Simple Peptide retatrutide
Proper storage is not optional when working with triple agonist compounds. Retatrutide contains multiple receptor-binding domains that are sensitive to heat, light, and moisture. Degradation of any single domain changes the compound activity profile in ways that compromise research validity.
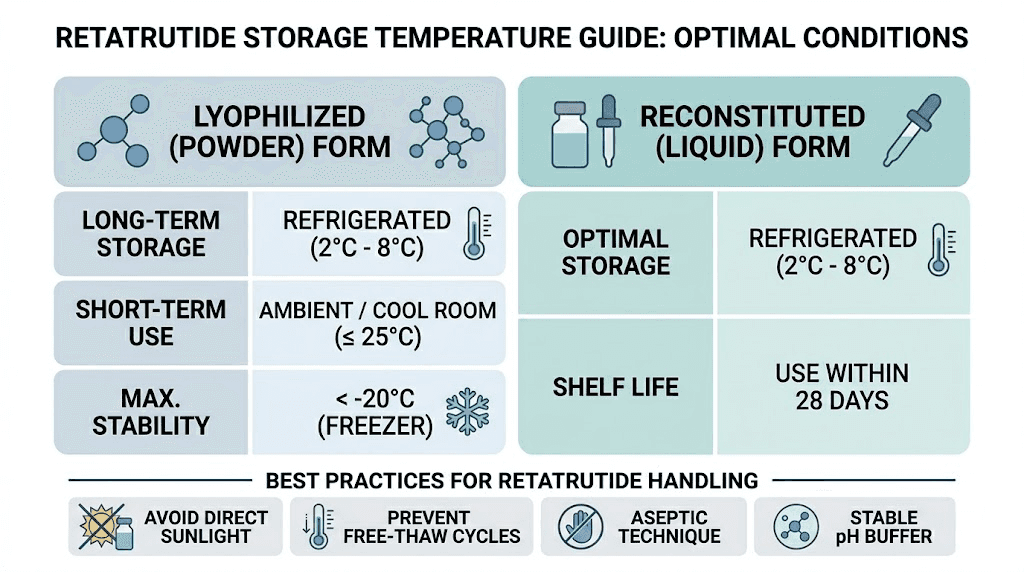
Pre-reconstitution storage
Lyophilized retatrutide should be stored at -20 degrees Celsius for long-term preservation. At this temperature, the peptide remains stable for extended periods with minimal degradation. Refrigeration at 2-8 degrees Celsius is acceptable for shorter storage windows of several weeks to a few months.
Room temperature storage should be avoided. Unlike some more stable peptides, the triple receptor binding domains in retatrutide are sensitive to thermal stress. Researchers who have experienced issues with peptides arriving warm during shipping should be especially cautious with retatrutide orders. If a package arrives warm, contact Simple Peptide immediately to discuss replacement options before using the product in any research protocol.
The complete retatrutide storage guide covers temperature considerations, light protection, and container selection in detail. For general peptide storage principles that apply across all compounds, see our comprehensive peptide storage resource.
Post-reconstitution handling
Once reconstituted, retatrutide must be refrigerated at 2-8 degrees Celsius. The reconstituted solution is significantly less stable than the lyophilized form. Most researchers plan to use reconstituted retatrutide within 4-6 weeks, though stability testing data specific to research-grade products varies by manufacturer.
Never freeze reconstituted peptide solutions. The freeze-thaw cycle damages peptide structure and can cause aggregation that is invisible to the naked eye but detectable on HPLC analysis. Aggregated peptides may show altered receptor binding characteristics.
Handle reconstituted vials with care. Avoid shaking, which can cause foaming and surface denaturation. Use sterile technique when withdrawing doses to prevent microbial contamination. The bacteriostatic water preservative (0.9% benzyl alcohol) inhibits but does not eliminate bacterial growth, particularly in protein-rich solutions like reconstituted peptides.
For researchers using Simple Peptide 3mL vial format, confirm whether the product arrives pre-reconstituted or as lyophilized powder. The storage requirements differ significantly between these two forms. The retatrutide shelf life guide provides specific timelines for both formats.
Side effects and safety profile from clinical data
Understanding the safety data is essential for any researcher working with retatrutide, regardless of vendor. The clinical trial data provides the most reliable information on adverse events and their frequency.
Gastrointestinal effects
The most commonly reported side effects in retatrutide clinical trials were gastrointestinal in nature. This is consistent with the entire GLP-1 agonist class, where constipation, bloating, and nausea represent the most frequent complaints.
Nausea occurred at significantly elevated rates across all dose levels compared to placebo. The incidence was dose-dependent, meaning higher doses produced more frequent and more intense nausea episodes. Most participants reported that nausea was most pronounced during dose escalation periods and tended to diminish with continued use at stable doses.
Diarrhea and vomiting also occurred at higher rates in treatment groups. Retatrutide-related diarrhea followed a similar pattern to what researchers observe with tirzepatide GI side effects, typically resolving within the first few weeks of a given dose level. Constipation was less common than with some single-target GLP-1 compounds, possibly due to the glucagon component stimulating gut motility.
Other reported effects
Beyond GI symptoms, clinical trial data reported several other effects worth noting for research protocol design. Hair loss concerns have emerged in community discussions, though clinical trial data did not identify this as a statistically significant adverse event. The hair loss discussion parallels similar conversations around GLP-1 compounds and hair thinning, where rapid weight reduction rather than the compound itself may be the primary driver.
Fatigue was reported by some participants, consistent with the general pattern seen with GLP-1 class compounds. Researchers studying this effect should note that caloric restriction itself produces fatigue independent of any pharmacological mechanism.
Muscle mass preservation is an active area of investigation. Some research suggests that the glucagon component may have muscle-sparing properties compared to pure GLP-1 agonists, but this requires further study. Researchers interested in body composition effects should include lean mass measurements in their protocols.
Skin sensitivity and injection site reactions have been reported at rates comparable to other injectable peptides. Standard injection technique and site rotation minimize these occurrences.
Monitoring recommendations
Researchers working with retatrutide should establish baseline measurements before initiating any protocol. Key parameters include fasting glucose, HbA1c, liver enzymes (ALT, AST), lipid panel, and body composition metrics. The onset timeline for retatrutide effects helps researchers plan when to schedule follow-up measurements for maximum data value.
The GLP-1 monitoring tools guide covers equipment and techniques for tracking key biomarkers throughout a research protocol. Regular monitoring is especially important during dose escalation phases when side effects are most likely to emerge.
Ordering from Simple Peptide
The ordering process at Simple Peptide involves several steps that researchers should be aware of before making their first purchase.
Account requirements
Simple Peptide requires account registration before allowing access to their full product catalog. This means you cannot browse products as a guest. While this adds friction to the initial experience, it serves as a basic identity verification step. Some researchers prefer vendors with open catalogs for easy comparison shopping, while others appreciate the additional layer of access control.
Registration requires standard information including name, email, and shipping address. The process is straightforward and typically takes less than five minutes to complete.
Shipping and delivery
Simple Peptide ships domestically within the United States. They emphasize fast processing and secure packaging. For temperature-sensitive compounds like retatrutide, packaging quality matters enormously. Researchers should verify that their order includes appropriate cold chain packaging, especially during summer months or when shipping to warm climates.
If you receive a package that appears to have been exposed to excessive heat, document the condition photographically before opening and contact customer support. Using a compromised product wastes both the compound and the research time invested in protocol preparation.
For researchers who travel with their peptide supplies, the GLP-1 travel case guide provides practical advice on maintaining cold chain integrity during transport.
Customer support and returns
Simple Peptide provides customer support through email (admin@simplepeptide.com) and phone (561-501-1570). Customer reviews across platforms indicate generally responsive communication, with most inquiries receiving replies within 24-48 hours.
Their website references a refund policy, though specific terms should be reviewed before purchasing. Research peptides are typically non-returnable once opened due to the inability to verify product integrity after breaking the seal. This is standard practice across the industry and not unique to Simple Peptide.
Retatrutide versus other weight management peptides
Researchers frequently need to select between multiple compound options for their studies. Understanding how retatrutide compares to the alternatives helps inform protocol design and vendor selection.
Retatrutide versus semaglutide
The comparison between these two compounds highlights the difference between single and triple agonism. Retatrutide consistently outperforms semaglutide across weight reduction, glycemic control, and liver fat reduction endpoints in published research.
Semaglutide targets only GLP-1 receptors. This makes it a cleaner compound for studying GLP-1 specific mechanisms but a less comprehensive tool for multi-pathway metabolic research. Semaglutide onset times are well characterized, with most studies showing measurable effects within 4-8 weeks.
Retatrutide onset appears comparable, with most researchers observing initial effects within similar timeframes. The difference becomes apparent at 12-24 weeks, where the triple agonist mechanism produces progressively larger separations from single-agonist compounds.
For researchers who need pure GLP-1 agonism without confounding from GIP and glucagon pathways, semaglutide remains the better choice. For those studying multi-pathway metabolic modulation, retatrutide offers a unique research tool. The comprehensive three-way comparison guide provides detailed analysis across dozens of parameters.
Retatrutide versus tirzepatide
This comparison is particularly relevant because tirzepatide already demonstrated significant improvements over semaglutide, making it the standard that retatrutide must beat. And in clinical research, it does.
Tirzepatide dual agonism (GLP-1 plus GIP) produces robust metabolic effects. Tirzepatide weight loss timelines show progressive improvement over 24-48 weeks. But the addition of glucagon receptor activation in retatrutide appears to provide an incremental but meaningful advantage, particularly for liver fat reduction and energy expenditure.
The dosage comparison between retatrutide and tirzepatide shows that retatrutide achieves similar or superior outcomes at comparable or lower milligram doses. This may have implications for research cost calculations, though pricing varies significantly across vendors including Simple Peptide.
Researchers studying the transition between compounds should review the tirzepatide to retatrutide switching protocol and the onset timeline when transitioning between these compounds.
Retatrutide versus mounjaro and other branded formulations
It is important to distinguish between research-grade retatrutide from vendors like Simple Peptide and pharmaceutical-grade formulations used in clinical trials. The retatrutide versus Mounjaro comparison highlights this distinction. Mounjaro is a branded tirzepatide product, while retatrutide is still in clinical development and not yet available as an approved pharmaceutical.
Research-grade compounds from vendors like Simple Peptide are synthesized for laboratory use and are not equivalent to clinical trial material. Purity specifications, excipient profiles, and formulation characteristics may differ. Researchers should design their protocols with these differences in mind and avoid making direct comparisons to clinical trial outcomes without appropriate caveats.
Stacking and combination research with retatrutide
Some researchers investigate retatrutide in combination with other compounds. This is an emerging area where Simple Peptide Reta/Cag blend represents a pre-formulated option for one specific combination.
Retatrutide and cagrilintide
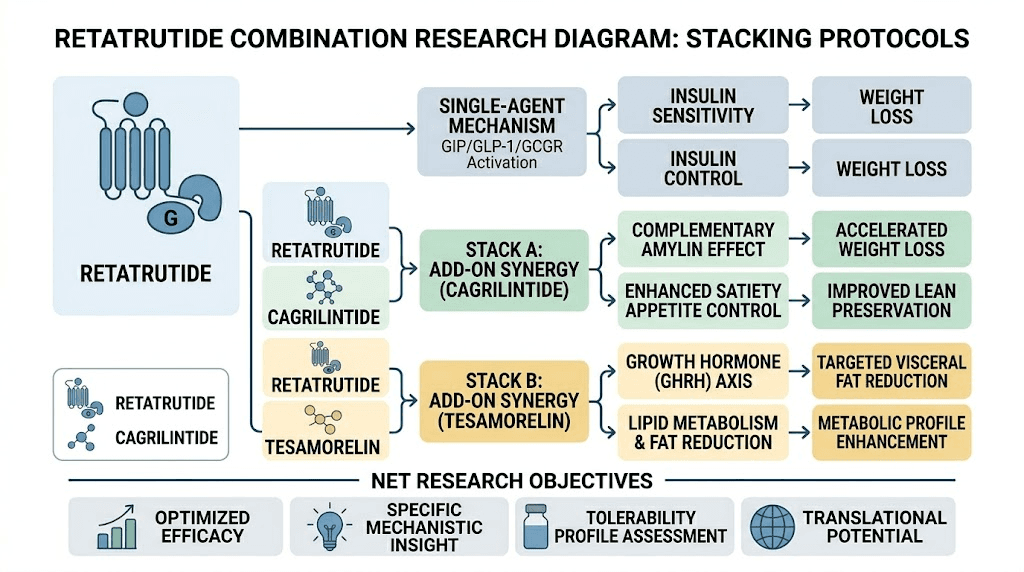
The cagrilintide and retatrutide combination represents one of the most discussed stacking protocols in the research community. Cagrilintide long-acting amylin agonism adds a fourth receptor pathway to the already comprehensive triple agonist activity of retatrutide.
Simple Peptide Reta/Cag blend (12.5mg/2.5mg) pre-mixes these compounds at a specific ratio. This offers convenience but removes the flexibility to adjust individual component doses. Researchers who need precise dose titration of each component may prefer sourcing them separately from cagrilintide suppliers and retatrutide vendors independently.
The CagriSema combination (cagrilintide plus semaglutide) has received more clinical attention, with Novo Nordisk advancing this combination through Phase 3 trials. The CagriSema dosing protocols provide a reference point for researchers designing their own cagrilintide combination studies.
Retatrutide and tesamorelin
Another combination under investigation involves tesamorelin co-administration with retatrutide. Tesamorelin is a growth hormone releasing hormone analog with specific effects on visceral fat reduction. The rationale for combining it with retatrutide centers on complementary mechanisms for body composition optimization.
This combination requires careful protocol design because both compounds affect metabolic pathways, and potential interactions have not been characterized in controlled clinical studies. Researchers should approach combination protocols with appropriate caution and comprehensive monitoring.
Important considerations for combination research
When combining retatrutide with any other compound, several factors require attention. First, verify that both compounds come from reliable sources with documented purity. Combining a high-purity retatrutide with a contaminated second compound invalidates the entire study. Second, establish baseline measurements for all relevant biomarkers before introducing either compound. Third, consider sequential introduction rather than simultaneous initiation, which allows attribution of effects and side effects to specific components.
The peptide stacking guide provides general principles for multi-compound research design. For guidance on concurrent peptide use, our comprehensive resource covers timing, compatibility, and monitoring considerations.
Retatrutide cost analysis: is Simple Peptide competitive?
Cost matters for researchers, particularly those running multi-week protocols that require sustained compound supply. Simple Peptide positions itself as an affordable option in a market where retatrutide pricing varies enormously.
Simple Peptide requires account creation to view pricing, which prevents direct price comparison without registration. This is a common practice among research peptide vendors, though researchers accustomed to transparent pricing from companies like Peptide Sciences may find it inconvenient.
When evaluating cost, do not just compare sticker prices. Calculate the cost per milligram of active peptide. A cheaper vial with lower net peptide content may actually cost more per usable dose than a more expensive product with higher purity. Request COAs showing net peptide content (not just HPLC purity) to make accurate cost comparisons.
The affordable retatrutide sourcing guide provides strategies for reducing research costs without compromising quality. For researchers on tight budgets, bulk purchasing options and international sourcing considerations offer additional pathways, each with their own quality verification requirements.
For overall research budget planning, the peptide cost calculator at SeekPeptides helps researchers estimate total protocol costs across different vendors and dosing schedules.
Red flags to watch for when ordering retatrutide
Simple Peptide has generally positive reviews, but the broader retatrutide market includes vendors of varying quality. Knowing what to watch for protects both your research and your investment.
Warning signs of low-quality vendors
Vendors who do not provide COAs upon request should be avoided entirely. Any legitimate peptide supplier will have batch-specific analytical data available. If a vendor provides only generic COAs without lot numbers, or if the COA appears templated rather than specific to your batch, treat the product with suspicion.
Unusually low pricing can indicate diluted product, lower purity, or sourcing from unverified manufacturers. While some legitimate vendors offer competitive pricing through efficient operations, prices significantly below market average warrant additional verification. Compare against the established price ranges for retatrutide to identify outliers.
Vendors who make therapeutic claims about their research compounds are violating regulatory requirements. Retatrutide is an investigational compound not approved for any therapeutic use. Any vendor marketing it with health claims is either ignorant of regulations or deliberately misleading customers. Neither reflects well on their quality control practices.
Check vendor longevity. Companies that appeared recently and offer unusually aggressive pricing may be liquidating substandard product or may not be around to honor quality complaints. Simple Peptide operational history since 2019 provides a moderate baseline, though it is shorter than established players in the market.
Verification steps before first use
Before using any retatrutide product in research, conduct basic quality checks. Visually inspect the product upon arrival. Lyophilized retatrutide should appear as a white to off-white powder or cake. Discoloration, clumping, or liquid in the vial (if expected to be lyophilized) may indicate degradation or contamination.
After reconstitution, the solution should be clear. Cloudiness, particulate matter, or visible aggregation are signs of a compromised product. Some researchers perform a basic solubility test by reconstituting a small amount first before preparing their full working solution.
For critical research applications, consider sending a sample to an independent testing laboratory for purity verification before beginning your protocol. The cost of independent testing is minimal compared to the cost of a compromised study. Our guide to peptide testing laboratories lists reputable facilities that offer peptide analysis services.
Who should consider Simple Peptide for retatrutide research?
Simple Peptide occupies a specific niche in the retatrutide vendor landscape. Understanding where they fit best helps researchers make informed sourcing decisions.
Best suited for
Researchers who value domestic US manufacturing and prefer dealing with a company that has a physical presence and verifiable contact information. The Florida-based operation provides a level of accountability that offshore vendors cannot match.
Those interested in the Reta/Cag combination blend, which is a relatively unique offering. Researchers studying multi-pathway metabolic modulation with four receptor targets will find this pre-blended product convenient for initial screening studies.
Researchers who prioritize customer service and communication. Review data suggests Simple Peptide is responsive to inquiries, which matters when questions arise about product specifications or shipping concerns.
May want to look elsewhere if
You require the highest independently verified purity scores. Vendors with more extensive third-party testing databases and higher independent ratings may be more appropriate for studies where absolute purity is critical.
You need transparent pricing without account creation. The registration requirement adds friction that some researchers prefer to avoid, especially when comparison shopping across multiple vendors.
You require international shipping. Simple Peptide appears focused on domestic US distribution. Researchers outside the United States should explore vendors with established international shipping capabilities.
For researchers who are still evaluating their options, the comprehensive retatrutide buying guide covers the full spectrum of sourcing options from domestic vendors to online purchasing considerations.
The future of retatrutide research
Retatrutide is still in active clinical development by Eli Lilly under multiple Phase 3 trial programs. The TRIUMPH study series is evaluating the compound across several indications including obesity, type 2 diabetes, osteoarthritis, obstructive sleep apnea, chronic low back pain, cardiovascular and renal outcomes, and MASLD.
If retatrutide receives regulatory approval, the research landscape will shift significantly. Approved pharmaceutical formulations will become available through traditional medical channels, potentially reducing demand for research-grade products. However, the timeline for approval remains uncertain, and current Phase 3 results are still being reported.
For the research community, vendors like Simple Peptide fill an important gap between clinical trial availability and potential future commercial approval. They provide access to compounds that researchers need for ongoing studies, receptor characterization, and comparative analyses.
The emergence of combination products like Simple Peptide Reta/Cag blend suggests that vendors are anticipating research trends toward multi-compound protocols. As understanding of retatrutide mechanisms deepens, expect to see more specialized product offerings targeting specific research applications.
Researchers serious about staying current with retatrutide developments should follow both the clinical trial registry (ClinicalTrials.gov) and independent testing databases. The compound profile is still evolving, and new data regularly updates the understanding of optimal dosing, safety parameters, and combination possibilities.
For researchers committed to optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions about vendor selection and research design.
Frequently asked questions
Is Simple Peptide a legitimate vendor?
Simple Peptide (operated by Melex Technologies) has been operational since 2019 and maintains a trust score of 95 out of 100 on ScamAdviser. Customer reviews are predominantly positive, and the company provides verifiable contact information including a Florida-based phone number. They are a legitimate research peptide vendor, though researchers should always verify product quality independently through COA review and, when possible, independent testing.
What is GLP-3RT from Simple Peptide?
GLP-3RT is Simple Peptide branded name for retatrutide, a synthetic tri-agonist peptide targeting GLP-1, GIP, and glucagon receptors. The compound is identical in mechanism to what the research community calls retatrutide or LY3437943. It comes in a 3mL vial format for research use only. For more on how this compound works, see our GLP-3 peptide guide.
Does Simple Peptide provide COAs for their retatrutide?
Simple Peptide claims to provide batch-specific Certificates of Analysis for all products, with testing performed by independent third-party laboratories. Researchers should request COAs before purchasing and verify that the documentation includes HPLC purity data, mass spectrometry confirmation, and net peptide content. For guidance on reading and interpreting COAs, see our peptide testing guide.
How does Simple Peptide retatrutide compare to Peptide Sciences?
Both vendors offer research-grade retatrutide with domestic manufacturing claims. Peptide Sciences has a longer track record and more extensive documentation, while Simple Peptide offers unique product formats like the Reta/Cag blend. Paradigm Peptides currently scores highest in independent testing databases with an A rating. The right choice depends on your specific research requirements, budget, and quality verification preferences.
What is the Reta/Cag blend from Simple Peptide?
The Reta/Cag blend combines retatrutide (12.5mg) with cagrilintide (2.5mg) in a single vial. This creates a four-pathway research compound targeting GLP-1, GIP, glucagon, and amylin receptors. It is designed for researchers studying the interaction between triple agonist and amylin receptor signaling. For more on this combination, see our cagrilintide and retatrutide dosing guide.
How should I store retatrutide from Simple Peptide?
Store lyophilized (powder form) retatrutide at -20 degrees Celsius for long-term storage or 2-8 degrees Celsius for short-term use. After reconstitution, refrigerate at 2-8 degrees Celsius and use within 4-6 weeks. Never freeze reconstituted solutions. Protect from light and avoid temperature fluctuations. See our complete retatrutide storage guide for detailed instructions.
Is retatrutide better than tirzepatide for research?
Clinical trial data shows retatrutide producing greater weight reduction (24.2% vs approximately 20-22%) and more dramatic liver fat reduction (82.4% vs approximately 50-60%) compared to tirzepatide. However, "better" depends on research objectives. Tirzepatide is more extensively studied, has regulatory approval for specific indications, and may be preferred for studies requiring a more established evidence base. See our detailed comparison for a comprehensive analysis.
Can I use Simple Peptide retatrutide for personal use?
No. All Simple Peptide products are labeled Research Use Only (RUO) and are intended strictly for laboratory, investigational, and scientific research purposes. They are not approved by the FDA and are not intended for human or veterinary use, clinical applications, diagnostic procedures, or therapeutic treatment of any kind. This applies to all research peptide vendors, not just Simple Peptide.
External resources
PubMed Central: Systematic Review of Retatrutide Safety and Efficacy
Nature Medicine: Retatrutide for Metabolic Dysfunction-Associated Steatotic Liver Disease
In case I do not see you, good afternoon, good evening, and good night. May your research stay rigorous, your peptides stay pure, and your protocols stay productive.