Feb 28, 2026
On paper, cagrilintide and retatrutide look like they belong to the same family. Both target weight loss. Both require once-weekly injections. Both sit in the same conversations about next-generation weight loss peptides that go beyond what semaglutide and tirzepatide can deliver.
But that is where the similarities end.
Cagrilintide is an amylin receptor agonist, a synthetic version of a hormone your pancreas already produces every time you eat. Retatrutide is something entirely different, a triple hormone receptor agonist that activates three separate metabolic pathways simultaneously. One works through satiety signaling. The other rewires your metabolism from multiple angles at once. The clinical data tells a story of two fundamentally different approaches to the same problem, and the differences matter more than most comparison articles acknowledge. Understanding which peptide aligns with your research goals requires looking past the headline weight loss numbers and into the mechanisms, the side effect profiles, the dosing protocols, and the practical realities of working with each compound. SeekPeptides has analyzed the clinical trial data from both compounds extensively, and this guide breaks down everything researchers need to make an informed decision.
What is cagrilintide and how does it work
Cagrilintide is a long-acting amylin analogue developed by Novo Nordisk. Amylin is a 37-amino-acid peptide hormone that your pancreatic beta cells co-secrete alongside insulin after meals. Most people have never heard of amylin, but it plays a critical role in how your body regulates appetite suppression and glucose metabolism.
The natural hormone does three things. It slows gastric emptying, which means food stays in your stomach longer and you feel full for extended periods. It suppresses glucagon secretion from the pancreas, which prevents your liver from dumping excess glucose into your bloodstream after meals. And it signals satiety through the area postrema in the brainstem, telling your brain that you have eaten enough.
Cagrilintide takes that natural mechanism and extends it dramatically.
Through lipidation, a process where a fatty acid chain is attached to the peptide molecule, researchers gave cagrilintide a half-life long enough for once-weekly dosing. The native amylin hormone lasts only minutes in circulation. Cagrilintide lasts days. This modification allows the peptide to bind to amylin receptor subtypes 1 and 3 in the brain, specifically in the area postrema of the hindbrain, producing sustained appetite reduction without the peaks and valleys that come with shorter-acting compounds. Research published in eBioMedicine confirmed that cagrilintide reduces food intake and body weight by reducing relative fat mass while maintaining relative lean mass, an important distinction that separates it from many fat loss peptides that can compromise muscle tissue.
For researchers familiar with cagrilintide and semaglutide combinations, the amylin pathway represents a complementary mechanism to GLP-1 agonism. Where GLP-1 agonists like semaglutide work primarily through incretin-based signaling, cagrilintide approaches satiety from a completely different neurological pathway. This is why the combination of both, known as CagriSema, has shown results that exceed either compound alone.
Cagrilintide clinical trial results
The phase 2 dose-finding trial enrolled participants with overweight or obesity and tested cagrilintide doses ranging from 0.3 mg to 4.5 mg administered subcutaneously once weekly. Over 26 weeks, including a dose-escalation period of up to 6 weeks, researchers observed mean percentage weight reductions of 6.0% to 10.8% across the cagrilintide dose groups compared to just 3.0% with placebo.
Those numbers tell only part of the story.
The highest dose of 4.5 mg produced an average weight loss of 11.5 kg, roughly 25 pounds, in just 26 weeks. And this was cagrilintide alone, without any GLP-1 agonist combination. The weight loss curves had not plateaued at week 26, suggesting that longer treatment periods would yield even greater reductions.
The phase 3 REDEFINE 1 trial expanded on these findings dramatically. This 68-week trial enrolled 3,417 adults with obesity or overweight with obesity-related complications. CagriSema, the combination of cagrilintide 2.4 mg and semaglutide 2.4 mg, produced an estimated mean body weight reduction of 20.4% compared to 3.0% with placebo. Cagrilintide 2.4 mg alone achieved approximately 10.8% weight loss over the same period, while semaglutide 2.4 mg alone achieved about 16%.
The math here is important. CagriSema exceeded the additive effect of both compounds individually. That suggests the amylin and GLP-1 pathways create a synergistic interaction, not just a combined one. Novo Nordisk filed for FDA approval of CagriSema in early 2026, with a review decision expected later this year.
What is retatrutide and how does it work
Retatrutide, developed by Eli Lilly, represents a fundamentally different philosophy of peptide design. Rather than targeting one receptor pathway with high specificity, retatrutide activates three distinct hormone receptors simultaneously: GLP-1 (glucagon-like peptide-1), GIP (glucose-dependent insulinotropic polypeptide), and glucagon.
Think of it as attacking the weight loss problem from three directions at once.
The GLP-1 component does what researchers already know GLP-1 agonists do. It enhances insulin secretion in response to meals, slows gastric emptying, and promotes satiety. This is the same mechanism that drives semaglutide and the GLP-1 component of tirzepatide. The GIP component adds another layer. GIP receptors exist in fat tissue, the brain, and the pancreas. Activating them improves insulin sensitivity and may enhance fat metabolism through pathways that GLP-1 alone does not reach. This is the same dual-agonist approach that made tirzepatide so effective, but retatrutide goes further.
The glucagon receptor activation is what makes retatrutide truly unique.
Glucagon is typically thought of as an opposing hormone to insulin, something that raises blood sugar. But glucagon receptor activation also stimulates energy expenditure and fat oxidation in the liver. It increases thermogenesis. It promotes the breakdown of stored fat for energy. This third mechanism means retatrutide does not just reduce how much you eat. It actively increases how many calories your body burns, even at rest. No other approved or late-stage peptide combines all three of these pathways in a single molecule. That is what earned retatrutide the nickname "triple G agonist" in research circles.
Retatrutide clinical trial results
The phase 2 trial, published in the New England Journal of Medicine, tested retatrutide at doses ranging from 1 mg to 12 mg weekly in adults with obesity. The results at 48 weeks were striking. Participants receiving the highest dose of 12 mg lost an average of 24.2% of their body weight, compared to just 2.1% with placebo. That translates to roughly 58 pounds of average weight loss.
At the 12 mg dose, 93% of participants achieved at least 5% weight loss. Over 80% achieved at least 15% weight loss. And 63% achieved at least 20% weight loss. These numbers surpassed every other single-agent weight loss peptide tested to that point.
The weight loss came with broad metabolic improvements. Participants showed significant reductions in waist circumference, systolic and diastolic blood pressure, glycated hemoglobin, fasting glucose, insulin levels, and lipid profiles. A separate analysis in Nature Medicine found that retatrutide produced significant reductions in liver fat content in participants with metabolic dysfunction-associated steatotic liver disease (MASLD), suggesting benefits beyond simple weight reduction.
The phase 3 TRIUMPH-4 trial confirmed these findings. In adults with obesity or overweight and knee osteoarthritis, retatrutide at 12 mg produced 23.7% body weight loss (an average of 60 pounds) compared to 4.6% with placebo over a full treatment period. The 9 mg dose achieved 20.0% weight loss. Eli Lilly reported these results in mid-2025, and the retatrutide availability timeline now points toward potential FDA approval no earlier than 2027, as additional phase 3 trials continue.
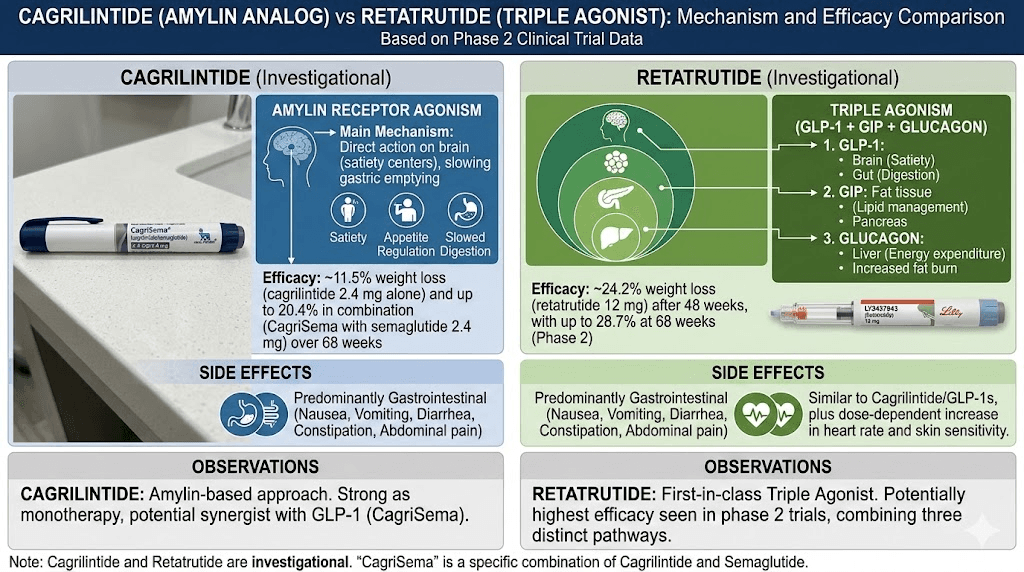
Mechanism of action comparison: amylin vs triple agonism
Understanding the mechanistic differences between cagrilintide and retatrutide is not just academic. These differences determine everything from side effect profiles to which patients respond best to each compound.
How cagrilintide targets satiety
Cagrilintide operates through a single, highly targeted pathway. The amylin receptors in the area postrema of the brainstem receive the signal and translate it into reduced hunger. This is a fundamentally different type of appetite suppression than what GLP-1 agonists provide.
GLP-1 agonists slow gastric emptying and affect appetite through gut-brain signaling. Amylin analogues act directly on the brain. The two pathways converge on the same outcome, reduced food intake, but they get there through different neurological routes. This is why combining cagrilintide with semaglutide produces synergistic effects. You are not doubling down on one pathway. You are activating two distinct satiety circuits simultaneously.
For researchers interested in cagrilintide dosing protocols, this mechanism has practical implications. The satiety effect tends to feel different from GLP-1-based appetite suppression. Where GLP-1 agonists often produce a sensation of fullness and food aversion (sometimes to the point of gastrointestinal discomfort), amylin-based satiety typically manifests as a more gentle reduction in hunger drive. Many researchers report that cagrilintide makes food less interesting rather than making eating feel unpleasant.
How retatrutide targets multiple metabolic pathways
Retatrutide takes the opposite approach. Instead of precision targeting one receptor system, it casts a wide net across three interconnected metabolic systems.
The GLP-1 component handles appetite suppression and insulin regulation, similar to what semaglutide does alone. The GIP component enhances insulin sensitivity in fat tissue and may promote beneficial fat metabolism, building on the same principle that makes tirzepatide dosing effective. But the glucagon receptor component is what truly separates retatrutide from everything else on the market.
Glucagon receptor activation increases hepatic lipid oxidation. Your liver burns more fat. It promotes thermogenesis, meaning your body generates more heat and expends more energy even at rest. And it shifts fuel utilization away from glucose and toward stored fat.
The practical result is a compound that reduces energy intake through appetite suppression while simultaneously increasing energy expenditure. This dual effect on both sides of the energy balance equation is why retatrutide produced the highest weight loss percentages of any single-agent trial. It is not just about eating less. It is about burning more.
Researchers considering retatrutide dosage protocols should understand that the three receptor interactions also mean a more complex pharmacological profile. The glucagon component can transiently raise blood glucose before the GLP-1 and GIP components bring it back down. This creates a unique metabolic dance that the body needs time to adjust to, which is why the retatrutide dose schedule uses a gradual titration over many weeks.
Receptor binding comparison table
Receptor target | Cagrilintide | Retatrutide | Effect |
|---|---|---|---|
Amylin receptors (1 and 3) | Primary target | Not targeted | Brain-based satiety, reduced hunger drive |
GLP-1 receptor | Not targeted | Active component | Gastric emptying, insulin, appetite |
GIP receptor | Not targeted | Active component | Insulin sensitivity, fat metabolism |
Glucagon receptor | Not targeted | Active component | Thermogenesis, fat oxidation, energy expenditure |
This table illustrates why these two peptides are not interchangeable. They operate through entirely non-overlapping receptor systems. A researcher choosing between them is choosing between two fundamentally different pharmacological strategies for addressing the same condition.
Weight loss results compared head to head
Numbers matter in this comparison. Here is what the clinical data actually shows when you line up the results side by side.
Cagrilintide monotherapy weight loss
In the phase 2 trial over 26 weeks, cagrilintide weight loss ranged from 6.0% to 10.8% of body weight depending on dose. The 2.4 mg dose, which became the standard dose carried forward into phase 3 trials, produced approximately 8% to 9% weight loss in 26 weeks. Over 68 weeks in the REDEFINE trials, cagrilintide 2.4 mg alone achieved approximately 10.8% body weight reduction. That is meaningful. It exceeds many GLP-1 therapies that have been on the market for years.
But it does not match the numbers that retatrutide puts up as a monotherapy.
Retatrutide weight loss at different doses
Retatrutide produced dose-dependent weight loss that scaled significantly with higher doses. At 4 mg weekly, participants lost approximately 17.5% body weight over 48 weeks. At 8 mg, losses approached 22%. And at the maximum studied dose of 12 mg, average weight loss hit 24.2% at 48 weeks.
The phase 3 TRIUMPH-4 data confirmed: 12 mg retatrutide produced 23.7% weight loss, translating to an average of 60 pounds lost. The 9 mg dose achieved 20.0%. Weight loss curves at the highest doses had not fully plateaued at the study endpoints, suggesting even greater losses with extended treatment.
For context, retatrutide vs semaglutide data shows retatrutide producing roughly 50% more weight loss than the highest approved semaglutide dose. And retatrutide vs tirzepatide comparisons suggest retatrutide edges ahead by 3 to 5 percentage points at comparable dose levels.
CagriSema combination weight loss
The most relevant comparison for cagrilintide, however, is not cagrilintide alone. It is CagriSema: cagrilintide 2.4 mg combined with semaglutide 2.4 mg. In the REDEFINE 1 trial, CagriSema produced 20.4% body weight loss over 68 weeks. In REDEFINE 2, which included participants with type 2 diabetes (a population that typically shows lower weight loss with any intervention), CagriSema achieved 13.7% weight loss compared to 3.4% with placebo.
These CagriSema numbers put it in direct competition with retatrutide monotherapy. Both approaches yield roughly 20-24% weight loss in their respective optimal protocols.
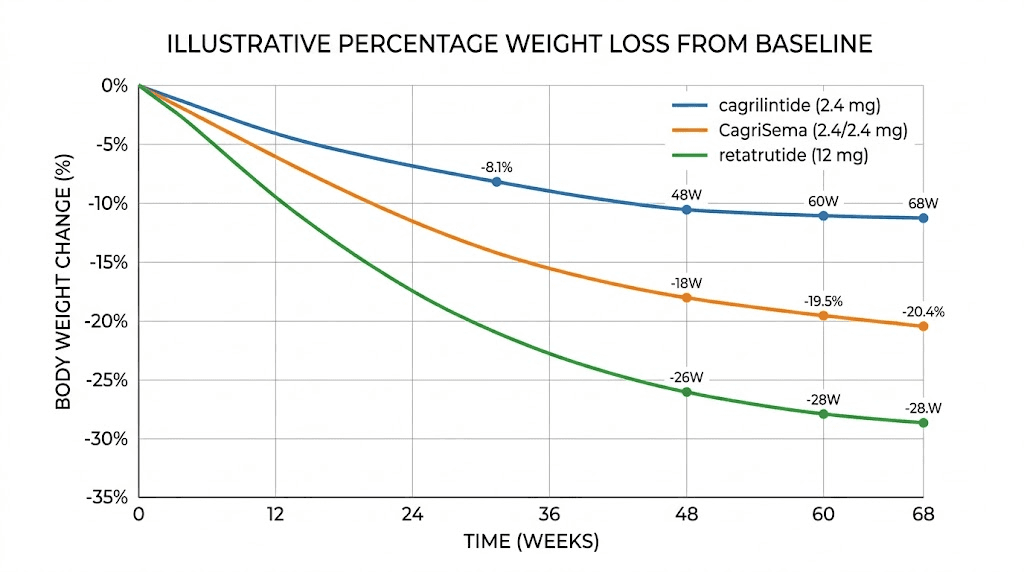
Weight loss comparison table
Compound | Dose | Trial duration | Mean weight loss | Trial phase |
|---|---|---|---|---|
Cagrilintide alone | 2.4 mg/week | 68 weeks | ~10.8% | Phase 3 |
Cagrilintide alone | 4.5 mg/week | 26 weeks | ~10.8% | Phase 2 |
CagriSema | 2.4 mg each/week | 68 weeks | ~20.4% | Phase 3 |
Retatrutide | 8 mg/week | 48 weeks | ~22.1% | Phase 2 |
Retatrutide | 12 mg/week | 48 weeks | ~24.2% | Phase 2 |
Retatrutide | 12 mg/week | Full treatment | ~23.7% | Phase 3 |
Retatrutide | 9 mg/week | Full treatment | ~20.0% | Phase 3 |
The takeaway is nuanced. Retatrutide as a single agent outperforms cagrilintide as a single agent by a significant margin. But CagriSema (cagrilintide combined with semaglutide) produces comparable results to retatrutide at moderate doses. The choice between them depends on whether a researcher prefers a single-compound approach or a combination protocol.
Dosing protocols compared
The practical aspects of dosing matter enormously for researchers. Both compounds use titration schedules designed to minimize side effects, but the specifics differ considerably.
Cagrilintide dosing schedule
The cagrilintide dosing protocol follows a gradual escalation over approximately 16 weeks. Treatment starts at 0.25 mg weekly and increases every four weeks through 0.5 mg, 1.0 mg, and 1.7 mg before reaching the maintenance dose of 2.4 mg per week. This mirrors the escalation approach used with semaglutide, which makes sense given that Novo Nordisk designed both compounds.
For researchers running CagriSema protocols, both the cagrilintide and semaglutide components are titrated simultaneously, though the specific escalation steps may differ slightly for each component.
The cagrilintide injection is administered subcutaneously, once weekly, at any time of day regardless of meals. Storage requirements follow standard peptide protocols: refrigerated at 2-8 degrees Celsius after reconstitution.
Retatrutide dosing schedule
The retatrutide dosage chart also follows a titration schedule, but it is longer and involves larger dose increments. Based on the phase 2 and phase 3 trial protocols:
Weeks 1-4: 1 mg once weekly
Weeks 5-8: 2 mg once weekly
Weeks 9-12: 4 mg once weekly
Weeks 13-16: 8 mg once weekly
Weeks 17 onward: 12 mg once weekly (maximum dose)
The starting dose of retatrutide is deliberately low because the triple-agonist mechanism produces more intense initial side effects than single-pathway compounds. Clinical data showed that participants who skipped titration steps or escalated too quickly experienced nearly double the rate of gastrointestinal adverse events.
This slower, more careful titration is a direct consequence of retatrutide activating three receptor systems. The body needs time to adapt to simultaneous GLP-1, GIP, and glucagon modulation. For detailed preparation instructions, researchers can reference our retatrutide reconstitution chart and bacteriostatic water mixing guide.
Dosing comparison table
Parameter | Cagrilintide | Retatrutide |
|---|---|---|
Frequency | Once weekly | Once weekly |
Starting dose | 0.25 mg | 1 mg |
Target maintenance dose | 2.4 mg | 8-12 mg |
Titration duration | ~16 weeks | ~16-20 weeks |
Dose steps | 0.25, 0.5, 1.0, 1.7, 2.4 mg | 1, 2, 4, 8, 12 mg |
Route | Subcutaneous injection | Subcutaneous injection |
Injection timing | Any time, regardless of meals | Any time, regardless of meals |
Researchers should note the significant difference in absolute dosing. Cagrilintide works at microgram-level potency (2.4 mg maintenance), while retatrutide requires milligram-level doses up to 12 mg. This reflects their different mechanisms. Amylin receptor activation requires far less compound than simultaneous triple receptor agonism. For those calculating doses, the retatrutide dosage calculator can simplify the math.
Side effects and safety profiles
Every peptide carries side effects. The question is not whether side effects occur but which ones, how severe, and how long they last. Here is where cagrilintide and retatrutide diverge significantly.
Cagrilintide side effects
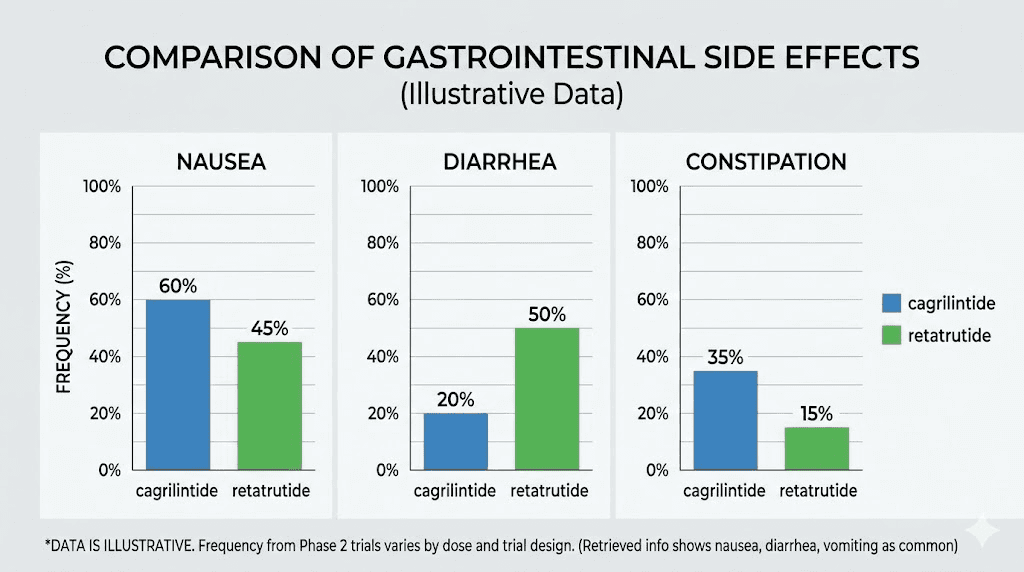
The cagrilintide side effect profile is relatively favorable compared to other weight loss peptides. In the phase 2 trial, 41% to 63% of participants receiving cagrilintide (0.3 to 4.5 mg) experienced gastrointestinal adverse events, compared to 32% with placebo. The most common issues were nausea (20% to 47% depending on dose), followed by constipation, diarrhea, and abdominal discomfort.
One notable finding from meta-analyses: cagrilintide demonstrated the lowest odds ratios for nausea, vomiting, and diarrhea when compared to other incretin-based therapies including semaglutide and tirzepatide. In other words, the gastrointestinal side effects were milder and less frequent than what researchers typically see with GLP-1 and GIP agonists.
Injection site reactions deserve separate mention. Cagrilintide showed a higher rate of injection site reactions compared to placebo, including mild redness and irritation. This is worth monitoring but rarely led to treatment discontinuation. The overall discontinuation rate due to adverse events was approximately 10%, which compares favorably to most compounds in this class.
For the CagriSema combination, gastrointestinal adverse events affected 79.6% of participants compared to 39.9% with placebo. This higher rate is expected when combining two appetite-suppressing compounds. However, the adverse events were described as mainly transient and mild-to-moderate in severity. Most resolved during the titration phase as the body adapted.
Retatrutide side effects
Retatrutide carries a heavier side effect burden, particularly during the initial weeks and dose escalation phase. The phase 3 TRIUMPH-4 data showed these rates at 9 mg and 12 mg doses respectively:
Nausea: 38.1% and 43.2% (vs 10.7% placebo)
Diarrhea: 34.7% and 33.1% (vs 13.4% placebo)
Constipation: 21.8% and 25.0% (vs 8.7% placebo)
Vomiting: 20.4% and 20.9% (vs placebo)
These numbers are higher than cagrilintide monotherapy across the board. The triple-agonist mechanism explains why. Activating GLP-1, GIP, and glucagon receptors simultaneously puts more stress on the gastrointestinal system during the adaptation period. The glucagon component can also cause transient increases in heart rate and blood glucose, effects that do not occur with amylin-based compounds.
Researchers familiar with retatrutide onset timelines know that the side effects typically peak during the first few weeks at each new dose level and then diminish. The gradual titration protocol exists specifically to manage this. Skipping dose steps dramatically increases the risk and severity of adverse events.
Hair loss has been reported by some retatrutide users, a side effect shared with rapid weight loss from any cause. The relationship appears to be with the rate and magnitude of weight loss itself rather than a direct drug effect. Similar patterns occur with GLP-1 related hair loss across multiple compounds.
Side effect comparison table
Side effect | Cagrilintide (2.4 mg) | Retatrutide (12 mg) | CagriSema |
|---|---|---|---|
Nausea | 20-47% | 43.2% | Higher (combined) |
Diarrhea | Lower than GLP-1s | 33.1% | Higher (combined) |
Constipation | Moderate | 25.0% | Moderate-high |
Vomiting | Lower than GLP-1s | 20.9% | Higher (combined) |
Injection site reactions | Notable increase vs placebo | Standard | Notable |
Discontinuation rate | ~10% | ~7-10% | ~10% |
Overall GI tolerability | Better than most GLP-1s | Similar to high-dose tirzepatide | Higher burden than either alone |
The bottom line on safety: cagrilintide alone has a milder side effect profile. Retatrutide produces more gastrointestinal distress but also delivers more weight loss as a monotherapy. CagriSema occupies a middle ground, with a higher side effect burden than cagrilintide alone but results that rival retatrutide. Researchers who are sensitive to gastrointestinal side effects or who have experienced significant issues with semaglutide constipation or tirzepatide GI issues may find cagrilintide monotherapy more tolerable, even at the cost of somewhat lower weight loss.
Who develops each compound: Novo Nordisk vs Eli Lilly
The corporate context matters for researchers tracking availability timelines and future access.
Cagrilintide is developed by Novo Nordisk, the same company behind semaglutide (Ozempic/Wegovy) and the CagriSema combination. Novo Nordisk has the infrastructure, regulatory experience, and manufacturing capacity from commercializing the most successful GLP-1 agonist on the market. Their filing for FDA approval of CagriSema in early 2026 puts them on track for a potential approval decision by late 2026 or early 2027.
Retatrutide is developed by Eli Lilly, the company behind tirzepatide (Mounjaro/Zepbound). Lilly has demonstrated their ability to bring complex peptide therapies through the regulatory process, and their phase 3 TRIUMPH trial program for retatrutide includes multiple studies across different patient populations. FDA approval for retatrutide is not expected before 2027, with some analysts projecting 2028.
Both companies have massive research pipelines. Both have proven they can manufacture and distribute peptide therapies at scale. The competition between them is driving unprecedented innovation in the weight loss peptide space.
Combination potential: stacking and synergy
One of the most important differences between cagrilintide and retatrutide is their potential for combination therapy.
Cagrilintide combination strategies
Cagrilintide was explicitly designed to be combined with other compounds. The cagrilintide and semaglutide combination (CagriSema) is the flagship application, and the clinical data confirms that the combination produces greater weight loss than either compound alone.
But the possibilities extend beyond CagriSema. Researchers are exploring cagrilintide dosage with tirzepatide as an off-label combination. The logic is straightforward: cagrilintide activates amylin receptors while tirzepatide activates GLP-1 and GIP receptors. There is zero receptor overlap, meaning both compounds can work at full efficacy without interfering with each other.
The cagrilintide dosage with retatrutide combination represents the ultimate multi-receptor stack. Cagrilintide covers amylin receptors 1 and 3. Retatrutide covers GLP-1, GIP, and glucagon receptors. Together, they would target five distinct metabolic pathways simultaneously. No clinical trial has tested this specific combination, but the theoretical basis for synergy is compelling.
For male researchers specifically, the combination potential of cagrilintide makes it an interesting component in broader peptide stacking protocols.
Retatrutide combination strategies
Retatrutide is a standalone powerhouse. Its triple-agonist mechanism means it already covers three of the most important metabolic pathways in a single injection. The question is whether adding anything to it provides meaningful benefit.
Some researchers are exploring combining tesamorelin with retatrutide to add growth hormone releasing hormone (GHRH) effects on top of the triple agonism. Others are investigating whether adding an amylin analogue like cagrilintide to retatrutide could provide additional satiety benefits beyond what the triple agonist achieves alone.
The challenge with combining retatrutide with other GLP-1-active compounds is receptor overlap. Adding semaglutide to retatrutide would duplicate GLP-1 receptor activation, which could increase side effects without proportionally increasing efficacy. This is why cagrilintide, which operates through completely different receptors, makes a more logical combination partner than additional GLP-1 agonists.
Researchers interested in the comparison between retatrutide and other compounds should also review the mazdutide vs retatrutide analysis and the Ozempic vs retatrutide comparison for additional context on where retatrutide fits in the broader landscape.
FDA status and availability timeline
Neither cagrilintide nor retatrutide is currently FDA approved. Both remain investigational. But their regulatory trajectories differ.
Cagrilintide, specifically as part of CagriSema, is further along in the regulatory process. Novo Nordisk filed the New Drug Application (NDA) for CagriSema in early 2026, based on positive results from the REDEFINE phase 3 trial program. If the FDA grants priority review, a decision could come by late 2026. Standard review would push the timeline into early 2027. Cagrilintide as a standalone drug does not appear to be pursuing separate FDA approval at this time, as Novo Nordisk is focusing on the combination product.
Retatrutide is still in the middle of its phase 3 trial program. While the TRIUMPH-4 results are in, additional trials including TRIUMPH-1, TRIUMPH-2, and TRIUMPH-3 are ongoing or planned. Eli Lilly has not yet filed an NDA. Most analysts expect a filing in late 2027 at the earliest, with potential approval in 2028.
For researchers who want to understand the current landscape of purchasing retatrutide or purchasing cagrilintide for research purposes, both compounds are available through various research peptide suppliers, though quality and purity vary significantly between vendors. Our guides on retatrutide online sources and Peptide Sciences retatrutide provide detailed vendor assessments.
Cost considerations for researchers
Cost is a practical factor that influences which compound researchers choose. While neither is commercially available as a prescription drug yet, research-grade peptide costs differ between the two.
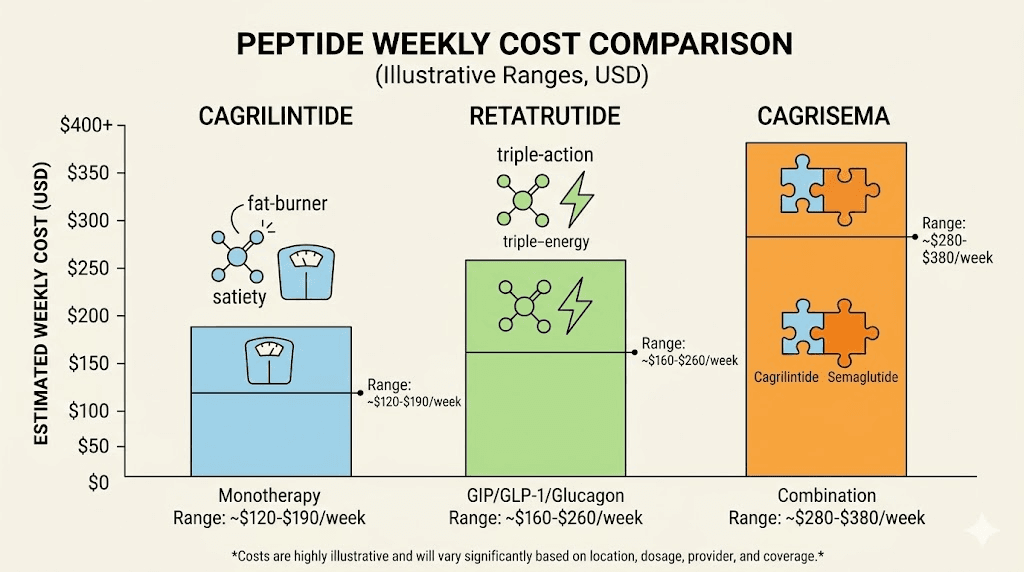
Cagrilintide requires lower absolute doses (2.4 mg weekly maintenance), which means less raw compound is needed per injection. At typical research peptide pricing, this translates to a lower per-dose cost. A 5 mg vial of cagrilintide provides roughly two weeks of maintenance dosing.
Retatrutide requires significantly higher doses (8-12 mg weekly at maintenance). A 10 mg vial provides roughly one week of dosing at the lower maintenance range. The retatrutide cost per week is higher in absolute terms, though the per-milligram price may be comparable or even lower depending on the vendor.
However, cost calculations get more complex when considering CagriSema protocols. Running both cagrilintide and semaglutide simultaneously means purchasing two separate compounds. The combined cost of CagriSema may exceed the cost of retatrutide monotherapy, even though retatrutide requires higher individual doses. Researchers should factor in the total cost of peptide therapy including reconstitution supplies, syringes, and storage equipment.
For those looking at budget-friendly options, bulk retatrutide purchasing can reduce per-milligram costs significantly. Similarly, researchers may find better pricing through the peptide cost calculator on SeekPeptides to compare overall protocol expenses.
Body composition effects: fat loss vs lean mass preservation
Weight loss alone does not tell the full story. What kind of weight you lose matters enormously. Losing muscle along with fat is counterproductive and potentially dangerous, especially for older adults.
Cagrilintide shows a particularly favorable body composition profile. The eBioMedicine research demonstrated that cagrilintide reduces relative fat mass while maintaining relative lean mass. This means the weight loss is preferentially coming from fat stores rather than muscle tissue. For researchers focused on simultaneous weight loss and muscle preservation, this characteristic makes cagrilintide an attractive option.
The amylin-based mechanism may contribute to this selectivity. Amylin signaling in the brain affects appetite and metabolism differently than GLP-1 signaling. The satiety produced by amylin analogues tends to reduce caloric intake without the extreme food aversion that sometimes occurs with high-dose GLP-1 agonists, which can lead to inadequate protein intake and subsequent muscle loss.
Retatrutide has less published data specifically on body composition. The phase 2 and phase 3 trials focused primarily on total body weight reduction. However, the glucagon receptor component theoretically favors fat oxidation over muscle catabolism. Glucagon promotes the breakdown of stored fat for energy while having relatively less impact on protein metabolism. The GIP component may also help preserve lean mass through its effects on insulin sensitivity in muscle tissue.
Early analyses suggest that retatrutide produces a body composition shift comparable to tirzepatide, where approximately 25-35% of total weight loss comes from lean mass and 65-75% from fat mass. This is within the typical range for rapid weight loss but better than what is seen with some less sophisticated approaches.
Researchers who prioritize body composition should consider supplementing either protocol with adequate protein intake. Our guides on optimal nutrition during peptide therapy and protein supplementation with GLP-1 therapy provide specific dietary frameworks. The supplement guides are equally applicable to researchers using either cagrilintide or retatrutide.
Metabolic effects beyond weight loss
Both cagrilintide and retatrutide affect metabolic parameters far beyond the number on the scale. These secondary benefits may influence which compound a researcher selects.
Blood sugar and insulin regulation
Cagrilintide suppresses post-meal glucagon secretion, which helps stabilize blood glucose levels. In clinical trials, cagrilintide alone improved glycemic parameters modestly. When combined with semaglutide in CagriSema, the glycemic improvements were substantial. In the REDEFINE 2 trial involving participants with type 2 diabetes, CagriSema significantly reduced HbA1c beyond what semaglutide alone achieved.
Retatrutide has a more complex relationship with blood glucose. The GLP-1 and GIP components improve insulin secretion and sensitivity, driving blood glucose down. But the glucagon component can transiently increase blood glucose by stimulating hepatic glucose output. The net effect in clinical trials was a significant improvement in glycemic parameters, including reductions in HbA1c, fasting glucose, and insulin levels. The body appears to find a new equilibrium where the insulin-promoting effects outweigh the glucagon-mediated glucose increases.
For researchers interested in metabolic health beyond weight loss, both compounds offer benefits. Retatrutide may have an edge for visceral fat reduction given the glucagon component effects on liver fat metabolism.
Liver fat reduction
Retatrutide demonstrated remarkable effects on liver fat in a dedicated study published in Nature Medicine. Participants with metabolic dysfunction-associated steatotic liver disease showed significant reductions in liver fat content with retatrutide treatment. This finding is particularly relevant because fatty liver disease affects a substantial percentage of people with obesity and can progress to serious conditions like cirrhosis and liver cancer.
Cagrilintide has not been specifically studied for liver fat reduction, though any compound that produces significant weight loss will likely reduce liver fat as a secondary benefit. The question is whether retatrutide glucagon component provides a specific, direct benefit to liver fat metabolism beyond what weight loss alone would produce. Early data suggests it does.
Cardiovascular markers
Both compounds showed improvements in cardiovascular risk markers in their respective trials. Retatrutide reduced blood pressure, improved lipid profiles, and decreased waist circumference significantly. Cagrilintide showed similar directional improvements, though the magnitude was generally proportional to the degree of weight loss rather than suggesting independent cardiovascular effects.
Researchers tracking comprehensive metabolic outcomes should monitor blood pressure, lipid panels, and inflammatory markers alongside weight when evaluating either compound. The GLP-1 plotter tools can help visualize metabolic trends over time.
Practical considerations for researchers
Beyond the clinical data, practical factors influence which compound works better in real-world research settings.
Reconstitution and preparation
Both compounds arrive as lyophilized (freeze-dried) powder that requires reconstitution with bacteriostatic water before injection. The reconstitution process is identical for both. Researchers familiar with preparing any injectable peptide will find nothing new here.
Cagrilintide concentrations per vial vary by supplier, but a typical 5 mg vial reconstituted with 2 mL of bacteriostatic water produces a concentration of 2.5 mg/mL. At a 2.4 mg maintenance dose, that is nearly one full milliliter per injection. The peptide reconstitution calculator simplifies these calculations.
Retatrutide vials are typically available in 10 mg or 20 mg configurations. A 10 mg vial reconstituted with 2 mL produces 5 mg/mL. At a 12 mg maintenance dose, researchers would use 2.4 mL per injection, requiring either larger syringes or split doses. The 20 mg retatrutide vial is often preferred because it allows higher concentration reconstitution and fewer vial changes.
Injection technique and sites
Both compounds use subcutaneous injection. Standard peptide injection techniques apply. Recommended injection sites include the abdomen (avoiding a 2-inch radius around the navel), the front of the thighs, and the back of the upper arms.
For cagrilintide, injection site reactions are slightly more common than with most peptides. Rotating injection sites regularly helps minimize this issue. The injection site selection guide covers rotation patterns that work for any subcutaneous peptide.
Retatrutide injections at higher maintenance doses (8-12 mg) may involve larger injection volumes depending on reconstitution concentration. Some researchers prefer a peptide injection pen for more precise dosing at higher volumes. Others split the weekly dose into two injections to reduce injection volume and potentially improve absorption consistency.
Storage requirements
Both compounds follow standard peptide storage protocols. Unreconstituted (lyophilized) powder should be stored in a cool, dark place. Reconstituted solutions must be refrigerated at 2-8 degrees Celsius. Most sources recommend using reconstituted solutions within 28 to 30 days.
Proper peptide storage is critical for maintaining potency. Temperature excursions degrade peptides rapidly. Researchers should also review our guides on peptide shelf life and reconstituted peptide stability for compound-specific storage recommendations.
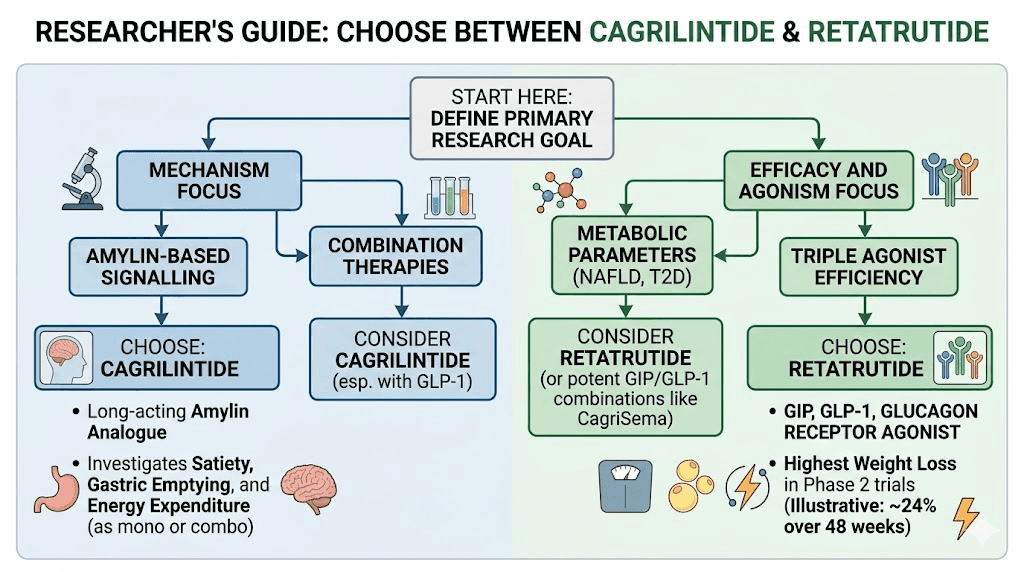
Which compound should you choose
The right choice depends on your specific research goals, tolerance for side effects, budget, and whether you prefer monotherapy or combination approaches.
Choose cagrilintide if
You want a compound with a milder side effect profile. Cagrilintide monotherapy produces less gastrointestinal distress than retatrutide, making it a better option for researchers who are sensitive to nausea, constipation, or diarrhea. If you have had difficulty tolerating semaglutide side effects or tirzepatide adverse events, cagrilintide alone may offer a smoother experience.
You are interested in combination protocols. Cagrilintide was designed for stacking. Its non-overlapping receptor profile makes it a logical addition to nearly any GLP-1 or GIP-based protocol without increasing receptor-specific side effects. If you want to build a multi-compound approach, cagrilintide is the more flexible building block.
You prioritize lean mass preservation. The data showing cagrilintide preferentially reduces fat mass while preserving lean mass is compelling for researchers who care about body composition, not just the scale number.
You want access to FDA-approved formulations sooner. CagriSema is likely to reach the market before retatrutide, given its more advanced regulatory status.
Choose retatrutide if
You want maximum weight loss from a single compound. Retatrutide at 12 mg produced 24.2% body weight loss in phase 2 trials, the highest of any single-agent compound tested. If your primary research goal is maximum weight reduction and you are willing to manage more side effects to get there, retatrutide is the strongest monotherapy option available.
You are interested in liver fat reduction. The published data showing retatrutide significantly reduced liver fat in participants with MASLD is unique among current weight loss peptides. If metabolic liver health is part of your research interest, retatrutide has specific evidence that other compounds lack.
You prefer protocol simplicity. Retatrutide is a single injection of a single compound once per week. CagriSema requires managing two separate compounds, two reconstitutions, and potentially two injection sites per week. The simplicity of retatrutide one injection, three mechanisms, has appeal for researchers who value straightforward protocols.
You are researching the glucagon pathway. Retatrutide is the only late-stage compound that provides significant glucagon receptor activation alongside GLP-1 and GIP agonism. If thermogenesis, energy expenditure, and fat oxidation through glucagon signaling are part of your research interest, retatrutide is the only option that provides this in a single molecule.
Consider both if
As discussed in the combination section, running cagrilintide alongside retatrutide represents the theoretical maximum in multi-receptor weight loss pharmacology. Five distinct metabolic pathways activated simultaneously. No clinical trial has tested this combination, but the pharmacological logic supports the approach for researchers operating at the cutting edge.
Future outlook: what comes next for both compounds
The peptide weight loss landscape is evolving rapidly, and both cagrilintide and retatrutide sit at the center of that evolution.
Novo Nordisk is advancing CagriSema toward commercial launch while also developing amycretin, a single-molecule dual GLP-1/amylin agonist that could eventually replace the two-injection CagriSema protocol with a single compound. Early data from amycretin showed up to 13.1% weight loss in just 36 weeks of a phase 1/2 trial, with the weight loss curve still descending steeply. If amycretin succeeds, it could make cagrilintide as a standalone product less relevant, though the amylin mechanism it pioneered would live on in the new molecule.
Eli Lilly continues to expand the retatrutide TRIUMPH trial program. Beyond the obesity indication, they are studying retatrutide for type 2 diabetes (TRIUMPH-2), obstructive sleep apnea (TRIUMPH-3), and cardiovascular outcomes. The breadth of these trials suggests Lilly sees retatrutide as a cornerstone product with applications far beyond weight loss.
Both companies are also investing in oral formulations, long-acting depot injections, and other delivery mechanisms that could transform how these peptides are administered. The current once-weekly subcutaneous injection model works, but once-monthly or even implantable formulations are on the horizon.
For researchers tracking the broader market, understanding how cagrilintide and retatrutide compare with existing options like semaglutide vs tirzepatide provides important context. The survodutide comparison and mazdutide comparison articles round out the competitive landscape.
How to get started with either compound
Researchers ready to begin working with cagrilintide or retatrutide should follow a systematic preparation process.
First, gather the necessary supplies. Both compounds require reconstitution equipment: bacteriostatic water, appropriate syringes (typically insulin syringes for cagrilintide and larger syringes for retatrutide high-dose protocols), alcohol swabs, and proper storage containers. The reconstitution calculator will help you determine exact water volumes for your specific vial size and desired concentration.
Second, plan your titration. Both compounds require gradual dose escalation. Do not skip this step. The clinical data is unambiguous: researchers who try to start at maintenance doses experience significantly worse side effects without faster results. Review the cagrilintide dosing guide or retatrutide dose guide for your chosen compound.
Third, source quality peptides. Purity matters enormously with research peptides. Third-party testing certificates (COAs) from independent labs should be verified before any research begins. Our vendor assessments cover options for both cagrilintide sources and retatrutide sources with emphasis on quality verification.
Fourth, establish monitoring protocols. Track weight, body composition, gastrointestinal symptoms, blood glucose if applicable, and any other relevant markers from the start. Consistent data collection makes protocol adjustments more informed and outcomes more meaningful. SeekPeptides members get access to tracking tools and protocol templates designed specifically for this purpose.
Switching between cagrilintide and retatrutide
Some researchers start with one compound and switch to the other based on their experience. Understanding how to transition safely matters.
Switching from cagrilintide to retatrutide involves moving from a single-receptor compound to a triple-receptor compound. The transition should include a washout period of at least 1-2 weeks after the last cagrilintide injection, followed by starting the retatrutide titration from the beginning (1 mg weekly). Do not attempt to jump to a higher retatrutide dose based on prior cagrilintide experience. The receptor systems are completely different, and tolerance to one does not predict tolerance to the other.
Switching from retatrutide to cagrilintide is somewhat simpler because you are moving from a broader mechanism to a narrower one. After completing a washout period, cagrilintide titration can begin at the standard 0.25 mg starting dose. Researchers who previously tolerated GLP-1 agonists may find that the amylin-based side effects of cagrilintide feel qualitatively different from what they experienced with retatrutide.
For researchers who have experience switching from tirzepatide to retatrutide or other peptide transitions, similar principles apply. Gradual titration from the lowest dose is always the safest approach when introducing a new compound with a different mechanism of action.
Frequently asked questions
Is cagrilintide more effective than retatrutide for weight loss?
As a standalone compound, no. Retatrutide monotherapy at 12 mg produced approximately 24% body weight loss compared to roughly 11% for cagrilintide 2.4 mg alone. However, when cagrilintide is combined with semaglutide (CagriSema), the combination produces approximately 20% weight loss, which approaches retatrutide results at moderate doses.
Can you combine cagrilintide and retatrutide together?
No clinical trial has tested this combination. Theoretically, the two compounds target completely different receptor systems (amylin receptors for cagrilintide, GLP-1/GIP/glucagon for retatrutide), which suggests they could work synergistically without receptor overlap. Our cagrilintide with retatrutide dosing guide covers the theoretical framework for researchers considering this approach.
Which compound has fewer side effects?
Cagrilintide monotherapy has a milder gastrointestinal side effect profile compared to retatrutide. Meta-analyses show cagrilintide has the lowest odds ratios for nausea, vomiting, and diarrhea among incretin-type therapies. Retatrutide produces higher rates of nausea (43% vs 20-47%), diarrhea (33% vs lower), and vomiting (21% vs lower) at their respective maintenance doses.
Are either of these peptides FDA approved?
Neither cagrilintide nor retatrutide is FDA approved as of early 2026. CagriSema (the cagrilintide plus semaglutide combination) has been filed for FDA review and could receive approval by late 2026 or early 2027. Retatrutide is still completing its phase 3 trial program, with FDA approval estimated for 2027 or 2028.
How do cagrilintide and retatrutide compare to semaglutide and tirzepatide?
Both represent a step beyond current FDA-approved options. Semaglutide (Wegovy) produces about 15-17% weight loss. Tirzepatide (Zepbound) achieves about 20-22%. CagriSema targets 20%+ and retatrutide targets 24%+. Each new generation adds either additional receptor targets or combination strategies that push weight loss outcomes further.
What is the difference between amylin and GLP-1 for weight loss?
Amylin and GLP-1 are two different hormones that both reduce appetite but through different brain pathways. GLP-1 agonists work primarily through gut-brain signaling and slowed gastric emptying. Amylin analogues like cagrilintide act directly on amylin receptors in the brainstem, producing satiety through the area postrema. This is why combining both pathways (as in CagriSema) produces greater weight loss than either alone.
How long does it take to see results with cagrilintide vs retatrutide?
Both compounds show measurable weight loss within the first 4-8 weeks of treatment, though most of this initial period involves dose titration rather than maximum efficacy. Significant results typically emerge by weeks 12-16 as maintenance doses are reached. Retatrutide tends to show steeper initial weight loss curves given its broader metabolic effects, while cagrilintide shows a more gradual, steady decline. For retatrutide specifically, our guide on how long retatrutide takes to work covers the expected timeline in detail.
Does cagrilintide preserve muscle mass better than retatrutide?
Published data from cagrilintide research specifically demonstrates reductions in relative fat mass while maintaining relative lean mass. Retatrutide does not yet have equivalent published body composition data. Based on the available evidence, cagrilintide appears to have a more favorable body composition profile, though the magnitude of difference between the two compounds remains to be determined through direct comparison studies.
External resources
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, detailed stacking protocols, dosing calculators, and a community of thousands who have navigated these exact questions about weight loss peptides and the latest next-generation compounds.
In case I do not see you, good afternoon, good evening, and good night. May your receptors stay responsive, your titrations stay gradual, and your research stay evidence-based.