Mar 24, 2026

Before you dismiss that strange tingling on your arm as nothing, read this. Semaglutide skin sensitivity is more common than most prescribing guides suggest, and what starts as mild irritation can escalate into something that disrupts sleep, daily comfort, and your entire treatment plan. The problem is not that people experience skin changes on semaglutide. The problem is that nobody warns them it might happen.
Research published in the American Journal of Health-System Pharmacy documents a condition called allodynia, where normal touch becomes painful, developing specifically at higher semaglutide doses. A scoping review in the Journal of the American Academy of Dermatology catalogs everything from mild injection site redness to rare but serious conditions like bullous pemphigoid. And clinical trial data from oral semaglutide studies show that 2.7% of participants reported "sensitive skin" as a distinct adverse event, compared to 0% in the placebo group.
These are not negligible numbers. They represent real people who woke up one morning and found that their clothing hurt. That a shower felt like sandpaper. That the spot where they injected last Tuesday still burns.
This guide covers every documented type of semaglutide skin sensitivity, from common injection site reactions to rare immune-mediated conditions. It explains the mechanisms behind each one, identifies who faces the highest risk, and provides specific management strategies backed by clinical evidence. Whether you are dealing with skin sensitivity right now or want to prevent it before your next dose increase, the information here comes directly from published research, case reports, and dermatology reviews.
What skin sensitivity on semaglutide actually looks like
Skin sensitivity on semaglutide does not present as a single, predictable reaction. It shows up in different ways for different people, at different doses, and at different points in treatment. Understanding what you are dealing with is the first step toward managing it effectively. The complete semaglutide management guide covers many side effects, but skin reactions deserve their own deep examination because they are so frequently misunderstood.
Some people notice nothing more than mild redness at the injection site that fades within hours. Others develop widespread skin tenderness that makes wearing fitted clothing uncomfortable. A small percentage experience full-blown allergic reactions requiring emergency medical attention.
The clinical literature identifies several distinct categories of semaglutide-related skin changes. Each has different causes, different timelines, and different solutions. Lumping them all together under "skin sensitivity" does patients a disservice, because the management approach for an injection site reaction differs completely from the approach for allodynia or systemic hypersensitivity.
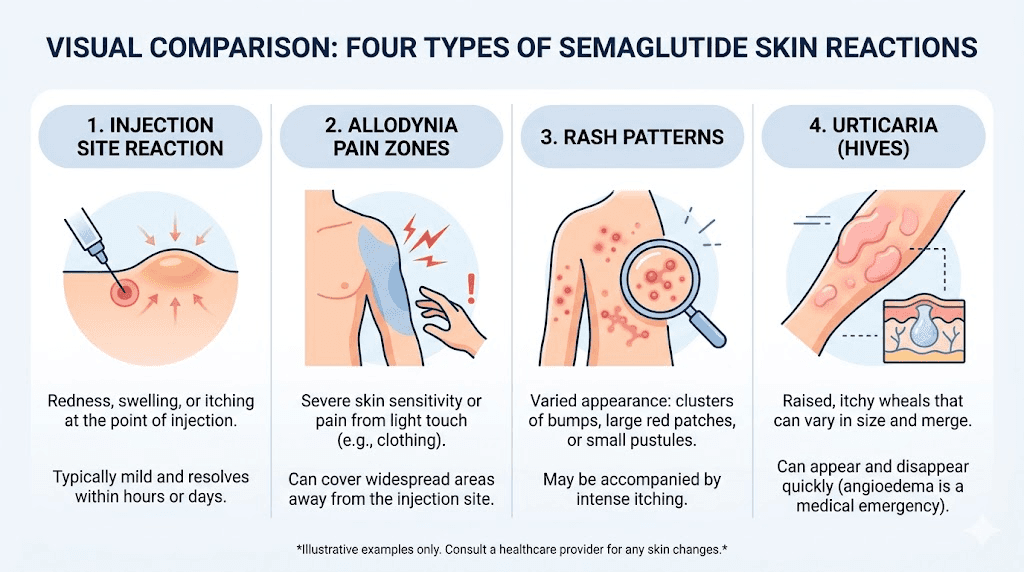
Injection site reactions
The most common skin complaint on semaglutide involves the injection site itself. Clinical trial data show injection site reactions in approximately 1.4% to 2.4% of patients using subcutaneous semaglutide products. These reactions typically manifest as localized redness, mild swelling, itching, or bruising that resolves within a few days. Most people who experience injection site issues find them annoying rather than debilitating.
But "common" does not mean "unimportant." Repeated injection site reactions at the same location can lead to lipodystrophy, where the subcutaneous fat tissue changes structure. A review in PMC documented 12 cases of lipodystrophy associated with GLP-1 receptor agonists. Once lipodystrophy develops, it can affect drug absorption from that site, creating unpredictable dosing outcomes.
Generalized skin tenderness and allodynia
This is the reaction that catches people completely off guard. Allodynia means pain from stimuli that should not normally cause pain, like the light touch of fabric against skin or the pressure of a seatbelt across the chest. A case series published in the American Journal of Health-System Pharmacy documented four patients who developed allodynia while on semaglutide, all at the 2.4 mg weekly dose.
Every single case showed a clear dose-response relationship. The symptoms appeared or worsened when the dose increased and, in two patients who discontinued treatment, resolved completely. The researchers scored each case 5 to 6 on the Naranjo Adverse Drug Reaction Probability Scale, placing them in the "probable" causation category. That is a significant finding. It means the connection between semaglutide and this painful skin sensitivity is not coincidental.
What makes allodynia particularly troublesome is that there is no visible sign on the skin. No rash. No redness. No swelling. The skin looks completely normal, but touching it produces genuine pain. This invisibility often leads to dismissal, both by the person experiencing it and by healthcare providers unfamiliar with this specific adverse effect.
Rashes and urticaria
Semaglutide can trigger various types of rashes beyond the injection site. The PMC dermatological review documented 22 cases of rash and 15 cases of urticaria (hives) across GLP-1 receptor agonist users. These rashes can appear as small red bumps, raised welts, widespread redness, or scaly patches anywhere on the body. Understanding the difference matters because each type suggests a different underlying mechanism and requires a different response.
Some rashes appear within hours of a dose and resolve quickly. Others develop gradually over weeks and persist. The timing and pattern of the rash provide critical diagnostic information. If you are tracking your GLP-1 injection schedule, noting any skin changes alongside your doses creates valuable data for your healthcare provider.
The science behind semaglutide skin reactions
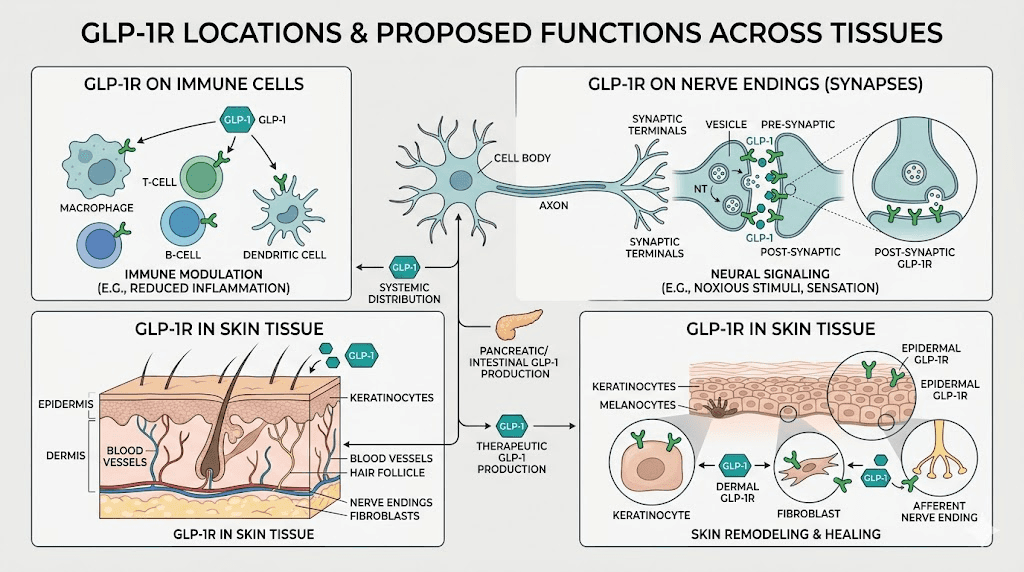
Understanding why semaglutide causes skin sensitivity requires examining several biological mechanisms. The peptide does not simply interact with GLP-1 receptors in the gut and pancreas. It affects inflammatory pathways, immune cell behavior, and potentially nerve signaling throughout the body. For anyone researching how semaglutide works, the skin effects reveal just how far-reaching GLP-1 receptor activation can be.
GLP-1 receptors and immune modulation
GLP-1 receptors exist beyond the digestive system. They appear on immune cells, including lymphocytes and macrophages, which play central roles in inflammatory skin responses. When semaglutide activates these receptors, it modulates the production of pro-inflammatory cytokines like TNF-alpha, IL-17, and IL-6. This immune modulation is actually beneficial in some skin conditions. Research shows GLP-1 effects on autoimmune conditions can be positive, with semaglutide reducing psoriasis severity scores by roughly 50% in clinical observations.
But immune modulation is a double-edged sword. The same mechanism that reduces psoriasis inflammation in one person can trigger hypersensitivity reactions in another. Individual genetic variations in immune function, pre-existing allergic tendencies, and the specific composition of compounded formulations all influence whether semaglutide calms or activates the immune system in a given person.
Mast cell activation and histamine release
Some semaglutide skin reactions appear to involve mast cells, the immune cells responsible for allergic responses. When mast cells degranulate (release their contents), they flood surrounding tissue with histamine and other inflammatory mediators. This produces the characteristic redness, swelling, and itching seen in urticaria and injection site reactions.
The connection between GLP-1 receptor agonists and mast cell behavior is not fully mapped. However, case reports of urticaria and angioedema following semaglutide administration suggest that mast cell activation plays a role in at least some reactions. This is why supplements that support immune balance during GLP-1 therapy have become a topic of growing interest.
Nerve signaling and peripheral sensitization
The allodynia cases present a different mechanistic puzzle. No pharmacological mechanism has been identified for semaglutide-induced allodynia, according to the case series authors. However, GLP-1 receptors exist on sensory neurons, and activation of these receptors could theoretically alter pain signaling thresholds. This would explain why normal touch becomes painful without any visible skin changes.
Peripheral sensitization, where nerve endings become hyper-responsive to stimuli, is a well-documented phenomenon in other conditions. Fibromyalgia, postherpetic neuralgia, and chemotherapy-induced neuropathy all involve similar mechanisms. The fact that semaglutide-induced allodynia resolves when the medication is discontinued (or sometimes even with continued use) suggests a reversible process rather than nerve damage.
This is an important distinction. Reversible sensitization means the nerves are temporarily "turned up too high" rather than structurally altered. For people experiencing this symptom, that knowledge alone can reduce anxiety significantly.
Excipient and formulation factors
Not all semaglutide products are identical. Brand-name products, compounded semaglutide formulations, and different administration routes contain different inactive ingredients. These excipients, including preservatives, stabilizers, and pH adjusters, can independently cause skin reactions in sensitive individuals.
Some compounded formulations include vitamin B12, glycine, or niacinamide as additional ingredients. Each of these can cause skin reactions independently of semaglutide itself. Niacinamide, for example, is well known for causing flushing in some people. If you developed skin sensitivity after switching formulations, the culprit might not be semaglutide at all. It might be something else in the vial.
Types of semaglutide skin sensitivity ranked by severity
Not all skin reactions carry the same level of concern. Some are nuisances. Others require immediate medical attention. Ranking them by severity helps you determine the appropriate response, whether that means adjusting your injection technique or calling emergency services.
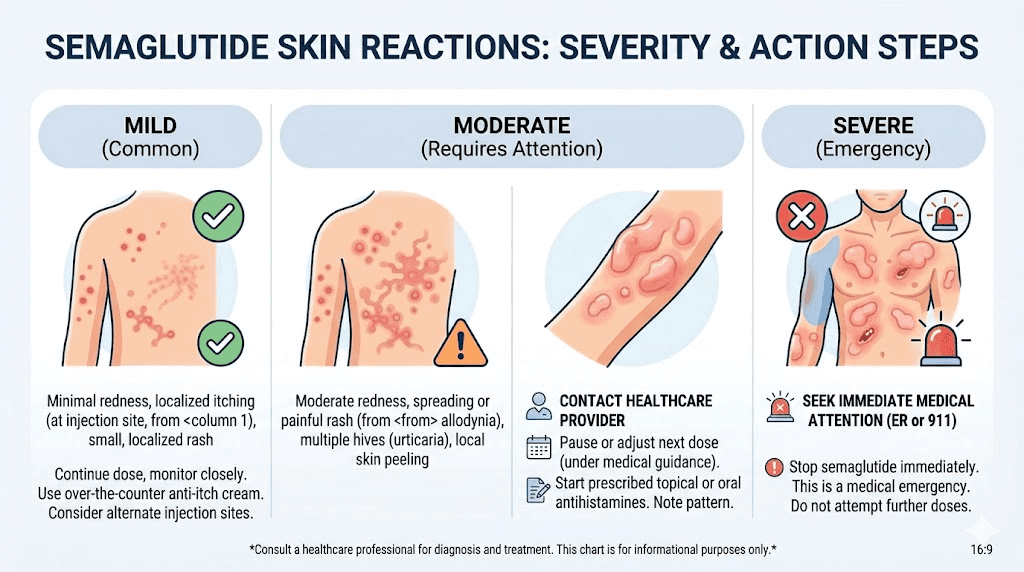
Mild reactions (self-manageable)
Injection site redness: Appears within minutes to hours of injection, resolves within 1 to 3 days. Affects 1.4% to 2.4% of subcutaneous semaglutide users. This is the most common skin reaction by far. Proper injection technique and consistent site rotation minimize occurrence.
Mild itching at injection site: Often accompanies redness. Usually resolves without treatment. If persistent, a cool compress applied for 10 to 15 minutes provides relief.
Bruising: Results from hitting a small blood vessel during injection, not from a drug reaction. Choosing the right syringe type and gauge reduces bruising frequency. Bruises at injection sites typically clear within 5 to 10 days.
Dry skin: Some users report increased skin dryness during semaglutide treatment. This may relate to changes in hydration status, altered nutrient absorption, or metabolic shifts during weight loss. Maintaining adequate water intake and following a proper semaglutide diet plan can help.
Moderate reactions (requires monitoring)
Allodynia (painful skin sensitivity): Documented at the 2.4 mg dose in four published cases. Skin appears normal but hurts with light touch. The Naranjo score of 5 to 6 indicates probable drug causation. Two of four patients saw resolution even while continuing treatment, which suggests this may be a temporary adaptation response in some individuals.
Persistent rash away from injection site: If a rash develops on areas of the body far from the injection site, this suggests a systemic rather than local reaction. The 75-year-old patient in the Journal of Drugs in Dermatology case report developed erythematous, scaly plaques on the back, chest, and legs after approximately 10 months on semaglutide. Her biopsy showed subepidermal blistering with eosinophils, indicating an immune-mediated process.
Altered skin sensations: Clinical trial data for oral semaglutide at the 50 mg dose reveal several distinct sensory changes: dysesthesia (abnormal sensation) at 1.8% versus 0% placebo, hyperesthesia (increased sensitivity) at 1.2% versus 0%, skin pain at 2.4% versus 0%, paresthesia (tingling or numbness) at 2.7% versus 0%, and "sensitive skin" at 2.7% versus 0%. Every single one of these occurred at rates significantly above placebo, confirming they are drug-related rather than coincidental.
These numbers might seem small. They are not. A 2.7% incidence rate means roughly 1 in 37 people on the 50 mg oral dose experience heightened skin sensitivity. If you are one of those 37, the percentage does not matter. What matters is managing it.
Severe reactions (requires medical intervention)
Angioedema: Deep tissue swelling, often affecting the face, lips, tongue, or throat. The PMC review documented 10 cases across GLP-1 receptor agonists. This is a medical emergency when it involves the airway. Stop the medication and seek emergency care immediately.
Bullous pemphigoid: An autoimmune blistering condition documented in 3 cases across GLP-1 agonist classes. Large, fluid-filled blisters develop on the skin. This requires dermatological evaluation and typically necessitates discontinuing the medication.
Anaphylaxis: A severe, life-threatening allergic reaction documented in 5 cases. Symptoms include widespread hives, difficulty breathing, rapid heartbeat, and drops in blood pressure. This requires immediate epinephrine and emergency medical services.
Leukocytoclastic vasculitis: Inflammation of small blood vessels in the skin. Presents as purplish spots (purpura) that do not blanch when pressed. Requires biopsy for confirmation and immediate medical management.
Who faces the highest risk for semaglutide skin sensitivity
Certain factors increase the likelihood of developing skin reactions on semaglutide. Knowing your risk profile before starting treatment allows you to monitor proactively and catch issues early. This is particularly important if you are just beginning your semaglutide protocol and want to know what to watch for.
Pre-existing skin conditions
People with eczema, psoriasis, dermatitis, or chronic urticaria already have hyperreactive skin immune systems. Adding a medication that modulates immune function can tip the balance in either direction. Interestingly, semaglutide sometimes improves psoriasis, with one study showing PASI scores dropping from 21 to 10 over 12 weeks. But for people with other inflammatory skin conditions, the response is less predictable.
If you have a history of skin conditions, document your baseline skin status before starting semaglutide. Take photographs. Note any active areas of irritation. This creates a reference point that makes it easier to identify new drug-related changes versus flares of existing conditions.
Allergic history
The PMC dermatological review specifically recommends that clinicians review patient histories regarding allergies and autoimmune diseases before initiating GLP-1 therapy. A history of drug allergies, food allergies, or allergic conditions like asthma increases the probability of hypersensitivity reactions to semaglutide or its excipients.
This does not mean people with allergies cannot use semaglutide. It means they should start with extra vigilance. Lower starting doses, slower titration schedules, and immediate-access antihistamines are reasonable precautions.
Rapid dose escalation
The allodynia case series provides the clearest evidence for dose-dependent skin sensitivity. All four patients developed symptoms at the 2.4 mg dose, the highest standard dose for weight management. The researchers explicitly note a "clear temporal and dose-response relationship" in each case.
This finding has practical implications. If you are following a standard semaglutide dosage chart and notice skin sensitivity appearing at higher doses, a slower titration approach may prevent or reduce the reaction. Some clinicians extend the time between dose increases from 4 weeks to 6 or 8 weeks to allow the body more adaptation time. Understanding how long semaglutide takes to work at each dose level helps set realistic expectations during a slower titration.
Compounded formulation variables
People using compounded semaglutide face additional variables. Different compounding pharmacies use different inactive ingredients, different concentrations, and different reconstitution processes. Some add methylcobalamin or other B vitamins. Others include amino acids like glycine as stabilizers.
If skin sensitivity appears after switching compounding pharmacies or formulations, the reaction may be to the new excipients rather than the semaglutide itself. This is an important diagnostic distinction because it means you might not need to abandon semaglutide entirely, just switch to a different formulation. When evaluating semaglutide formulation differences, color changes between batches can also signal formulation variations worth investigating.
Oral versus injectable route
The clinical trial data reveal a striking finding: oral semaglutide at 50 mg produces higher rates of skin sensory changes than subcutaneous injection. Dysesthesia, hyperesthesia, skin pain, paresthesia, and sensitive skin all appeared at measurable rates in the oral group versus 0% in placebo. This suggests that the oral delivery route, or its associated higher peak concentrations, may carry greater risk for certain types of skin sensitivity. If you are considering oral semaglutide options, this data point deserves consideration.
On the other hand, oral semaglutide eliminates injection site reactions entirely. There is no needle, so there is no local trauma. For people whose primary skin complaint is injection-related, switching to an oral or sublingual format may resolve the issue while potentially introducing different sensory considerations.
Dose-related skin sensitivity patterns
The relationship between semaglutide dose and skin sensitivity is not linear. It follows patterns that, once understood, become predictable and manageable. Tracking these patterns alongside your semaglutide dosage calculations gives you the data needed to make informed decisions about your protocol.
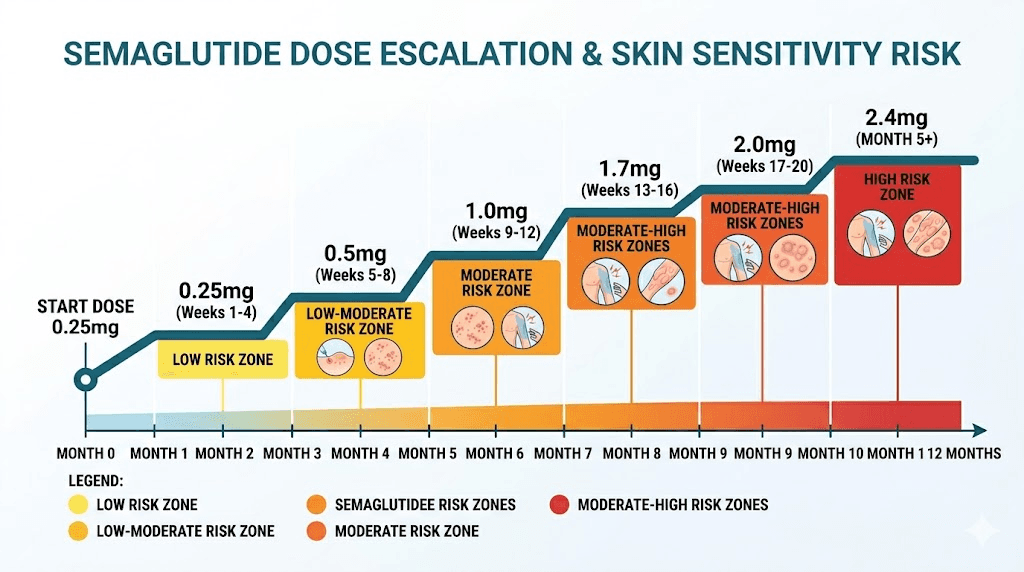
The dose escalation window
Most new skin reactions appear within 1 to 3 weeks of a dose increase.
This matches the pharmacokinetic profile of semaglutide, which takes approximately 4 to 5 weeks to reach new steady-state concentrations after a dose change. During that transition window, the body is adjusting to higher circulating levels of the medication.
Here is what typically happens at each dose level:
0.25 mg (starting dose): Skin reactions at this dose are uncommon. The primary purpose of this dose is GI adaptation, and the systemic levels are relatively low. Most people tolerate this dose without any skin changes whatsoever.
0.5 mg: Injection site reactions may first appear here as the dose doubles. Mild redness and occasional itching are the most common complaints. These usually resolve with proper injection technique and site rotation.
1.0 mg: The first clinically meaningful dose for weight loss. Some users report dry skin or mild sensitivity changes at this level. The timeline for semaglutide effects becomes more apparent, and so do side effects.
1.7 mg: Skin sensitivity reports increase at this dose. Understanding unit conversions at this dose helps ensure accurate administration, which matters because injection errors can contribute to local skin reactions.
2.4 mg: The dose at which allodynia cases were documented. All four published cases developed symptoms at this level. This is the highest standard dose, and it represents the point where dose-dependent skin effects become most likely. If you have been tolerating lower doses well but plan to titrate to 2.4 mg, heightened monitoring for skin changes during the first 4 to 6 weeks at this dose is prudent.
The adaptation curve
An encouraging finding from the allodynia case series: one of the four patients who continued semaglutide despite developing allodynia experienced complete symptom resolution after four months. This suggests that at least some skin sensitivity reactions represent a temporary adaptation period rather than a permanent side effect.
This adaptation phenomenon is consistent with what we see with other semaglutide side effects. Semaglutide fatigue, for example, commonly peaks in the first few weeks after a dose increase and then gradually improves. GI side effects follow a similar pattern. The body adapts.
However, adaptation is not guaranteed. Two of the four allodynia patients discontinued treatment because symptoms were intolerable. Deciding whether to push through a temporary reaction versus discontinuing requires balancing the severity of the skin sensitivity against the benefits of continued treatment.
How to manage semaglutide skin sensitivity
Management strategies depend entirely on what type of skin sensitivity you are experiencing. A blanket approach does not work because the underlying mechanisms differ. Here are specific, evidence-based strategies organized by reaction type.
Managing injection site reactions
Rotate injection sites systematically. Do not just inject in a slightly different spot. Create a deliberate rotation pattern across abdomen, thighs, and upper arms. Each injection should be at least 2 inches (5 cm) from the previous site. Some people find it helpful to track their injection locations in a log or app to ensure consistent rotation.
Check your injection technique. Injecting too quickly forces fluid into tissue faster than it can distribute, creating local pressure and irritation. Injecting too shallowly (intradermally rather than subcutaneously) causes more surface-level reactions. The proper injection technique guide covers angle, depth, and speed in detail.
Allow the medication to warm slightly. Cold injections cause more local irritation than room-temperature ones. Remove the vial from the refrigerator 15 to 20 minutes before injection. This is especially relevant if you are managing strict semaglutide storage protocols and injecting immediately after pulling the vial from cold storage.
Apply cool compresses after injection. A clean, cool (not frozen) compress applied for 10 to 15 minutes post-injection reduces local inflammation. Do not apply ice directly to skin, as this can cause its own irritation.
Clean the site with alcohol and let it dry completely. Injecting through wet alcohol pushes the antiseptic into subcutaneous tissue, which stings and can cause redness unrelated to the medication itself.
Managing allodynia and widespread skin tenderness
This is the trickiest type to manage because there is no rash to treat, no inflammation to reduce, and no well-established pharmacological intervention. Based on the published cases and general allodynia management principles, consider the following approaches.
Discuss dose reduction with your provider. Since allodynia appears to be dose-dependent, reducing from 2.4 mg to 1.7 mg while maintaining appetite suppression benefits may eliminate the skin sensitivity entirely. Many people achieve excellent results at lower doses.
Consider the 4-month adaptation window. One patient in the case series experienced complete resolution after four months of continued treatment at the same dose. If the allodynia is tolerable, giving it time may result in spontaneous improvement. This is a discussion to have with your healthcare provider, weighing symptom severity against treatment goals.
Wear loose, soft fabrics. Tight clothing exacerbates allodynia by maintaining constant pressure on sensitized skin. During episodes, choose loose-fitting garments made from soft materials like cotton or bamboo.
Avoid extreme temperatures. Both hot showers and cold exposure can intensify allodynia symptoms. Lukewarm water is the safer choice during active episodes.
Over-the-counter pain management. Topical lidocaine patches can provide local relief for the worst-affected areas. Oral gabapentin or pregabalin, prescribed by your healthcare provider, may help by modulating nerve signaling pathways. These are not first-line treatments for semaglutide-induced allodynia specifically, but they are well-established for allodynia from other causes.
Managing rashes and urticaria
When a rash develops, the management approach depends on its extent and severity.
Localized rash near injection site: Over-the-counter hydrocortisone cream (1%) applied twice daily can reduce inflammation. If the rash recurs with each injection at a particular site, that area may need to be excluded from your rotation permanently.
Widespread rash: Oral antihistamines (cetirizine or loratadine) can provide symptomatic relief. These are non-drowsy options that do not interact with semaglutide. Document the rash with photographs and contact your healthcare provider. A widespread rash after semaglutide administration may warrant dermatology referral, especially if biopsy could clarify the underlying mechanism.
Recurrent urticaria (hives): If hives appear repeatedly after semaglutide doses, this pattern strongly suggests a hypersensitivity reaction. Do not simply keep taking antihistamines and continuing. This warrants a serious conversation with your prescriber about whether to continue, reduce the dose, or switch medications. Some patients have successfully switched between GLP-1 medications when one caused reactions the other did not.
Supporting skin health during semaglutide treatment
Proactive skin care during semaglutide use can reduce the risk of reactions and improve tolerance.
Hydration is foundational. Semaglutide reduces appetite and food intake, which often means reduced fluid intake as well. Dehydrated skin is more reactive, more prone to irritation, and heals more slowly. Aim for at least 64 ounces of water daily, more if you are active. Your semaglutide food plan should include water-rich fruits and vegetables.
Protein protects skin integrity. Collagen production requires adequate protein intake, and rapid weight loss on semaglutide can deplete protein stores if intake is insufficient. The recommended protein intake on semaglutide is higher than standard recommendations specifically because the body needs building blocks for tissue maintenance during caloric restriction.
Supplement strategically. Zinc, vitamin C, and omega-3 fatty acids all support skin barrier function. The complete GLP-1 supplement guide covers which supplements to prioritize based on your specific protocol and goals.
Use fragrance-free products. During semaglutide treatment, sensitized skin reacts more strongly to fragrances, dyes, and chemical irritants in skincare products. Switching to fragrance-free, hypoallergenic products during treatment reduces confounding skin irritation.
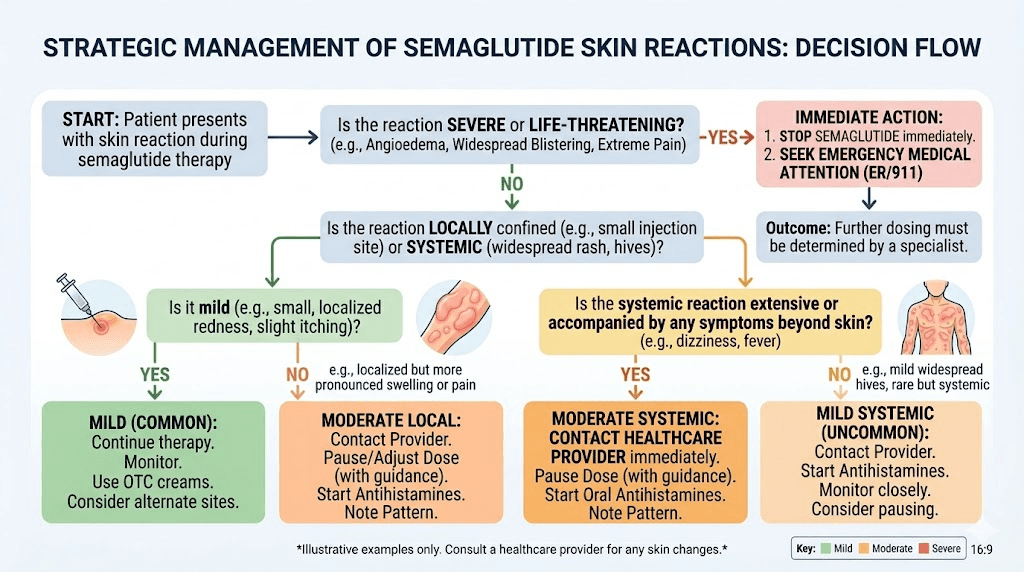
When skin sensitivity becomes a medical emergency
Most semaglutide skin reactions are manageable. But some require immediate emergency care. Knowing the difference could save your life.
Call emergency services immediately if you experience:
Swelling of the face, lips, tongue, or throat. This is angioedema and can compromise your airway within minutes. Do not wait to see if it improves.
Difficulty breathing or swallowing. Combined with skin changes, this pattern suggests anaphylaxis, a systemic allergic reaction that requires epinephrine.
Rapid onset of widespread hives covering large areas of the body, especially if accompanied by dizziness, rapid heartbeat, or a feeling of impending doom. This classic anaphylaxis presentation demands immediate intervention.
Large, fluid-filled blisters developing across multiple body areas. This could indicate bullous pemphigoid or another serious blistering condition that requires urgent dermatological evaluation.
Purple or dark spots on the skin that do not blanch (turn white) when pressed. These purpuric lesions may indicate vasculitis, inflammation of blood vessels that can affect multiple organ systems.
The FDA labeling for all semaglutide products includes warnings about serious hypersensitivity reactions including anaphylaxis and angioedema. These are rare, with only 5 cases of anaphylaxis and 10 cases of angioedema documented across all GLP-1 agonists in the PMC review. Rare does not mean impossible. If you have a history of severe allergic reactions to any medication, discuss an emergency action plan with your provider before starting semaglutide, and consider having an epinephrine auto-injector available during the first several doses.
Oral versus injectable semaglutide and skin reactions
The route of administration significantly influences which skin reactions you are likely to encounter. This distinction matters because it creates options. If one route causes intolerable skin effects, the other may not.
Injectable semaglutide skin profile
Subcutaneous semaglutide (brand names like Ozempic and Wegovy) causes injection site reactions in 1.4% to 2.4% of users. These are localized, predictable, and manageable with proper technique. Systemic skin reactions like widespread rash or allodynia are less common with injectable formulations at standard doses.
The advantage of injectable semaglutide is that the drug enters the bloodstream gradually from the subcutaneous depot, creating relatively stable blood levels over the week. This steady-state pharmacokinetics may produce fewer neural sensitization effects compared to the peak-and-trough pattern of oral absorption.
If you are using injectable semaglutide and experiencing injection site issues, perfecting your injection administration technique and ensuring proper reconstitution practices can make a significant difference. Many injection site reactions trace back to technique rather than drug sensitivity.
Oral semaglutide skin profile
Oral semaglutide eliminates injection site reactions entirely. However, the clinical trial data for the 50 mg oral dose show meaningfully higher rates of generalized skin sensory changes. Sensitive skin at 2.7%, paresthesia at 2.7%, skin pain at 2.4%, dysesthesia at 1.8%, and hyperesthesia at 1.2% all exceeded placebo rates of 0%.
These differences may relate to the pharmacokinetic profile of oral absorption, which creates different peak concentration patterns than subcutaneous injection. The sublingual semaglutide approach represents another oral delivery method with potentially different absorption characteristics, though comparative skin reaction data is limited.
Switching routes to manage skin reactions
If injectable semaglutide causes persistent injection site problems, switching to oral delivery eliminates that specific issue. If oral semaglutide causes generalized skin sensitivity, switching to injectable delivery may reduce those systemic sensory effects while introducing the possibility of injection site reactions.
The key insight is that these are different reaction profiles, not the same reactions from different routes. A person who develops allodynia on oral semaglutide may tolerate injectable semaglutide without skin issues, and vice versa. This creates a pathway to continue GLP-1 therapy even when one formulation causes problems.
Can you continue semaglutide with skin sensitivity
This is the question that matters most to people experiencing skin reactions. They are getting results from semaglutide. Their weight loss trajectory is on track. Their metabolic markers are improving. And now this skin problem threatens to derail everything.
The answer depends on the type and severity of the reaction.
Reactions that usually allow continued treatment
Mild injection site reactions. These are manageable with technique adjustments and typically do not worsen over time. Most people with injection site redness or bruising continue treatment indefinitely without problems.
Mild allodynia. The case series shows that some patients can continue treatment with symptom resolution occurring after several months. This requires close monitoring and regular reassessment, but discontinuation is not mandatory for mild cases.
Dry skin and minor sensitivity changes. These respond well to proactive skincare, adequate hydration, and supportive supplementation. They rarely require discontinuation.
Reactions that require dose reduction
Moderate allodynia interfering with daily activities. Dropping from 2.4 mg to 1.7 mg, or from 1.7 mg to 1.0 mg, may resolve dose-dependent skin sensitivity while maintaining clinically meaningful appetite suppression. Many people achieve excellent monthly results at moderate doses.
Recurrent rash at specific dose levels. If rash consistently appears after dose increases and resolves at lower doses, the maximum tolerated dose may be below the target dose. Staying at the highest tolerated dose is better than pushing higher and dealing with ongoing skin reactions.
Reactions that require discontinuation
Anaphylaxis or angioedema. These are absolute contraindications to continued use. Do not rechallenge with semaglutide after an anaphylactic or angioedema event.
Bullous pemphigoid or other serious immune-mediated skin conditions. The 75-year-old patient in the case report showed healing of existing lesions and no new lesion development within 3 weeks of discontinuing semaglutide. These conditions resolve with discontinuation but can worsen significantly with continued exposure.
Severe, intractable allodynia. Two of the four patients in the case series discontinued because symptoms were unbearable. If quality of life is severely impacted and dose reduction does not help, discontinuation is the appropriate choice.
Alternative GLP-1 options after discontinuation
Discontinuing semaglutide does not mean abandoning GLP-1 therapy entirely. Tirzepatide is a dual GIP/GLP-1 receptor agonist with a different molecular structure. While it can also cause skin reactions, a person who reacts to semaglutide may tolerate tirzepatide well, and the conversion process is well documented.
Other GLP-1 receptor agonists like liraglutide and dulaglutide have different excipient profiles and slightly different receptor binding characteristics. Cross-reactivity between GLP-1 agonists for skin reactions is not universal, meaning an allergy to one does not guarantee allergy to all. This is a conversation for your allergist or dermatologist, who can help determine whether a different GLP-1 agonist is safe to try.
For people who cannot tolerate any GLP-1 agonist, other weight management approaches exist, though they may not offer the same efficacy profile.
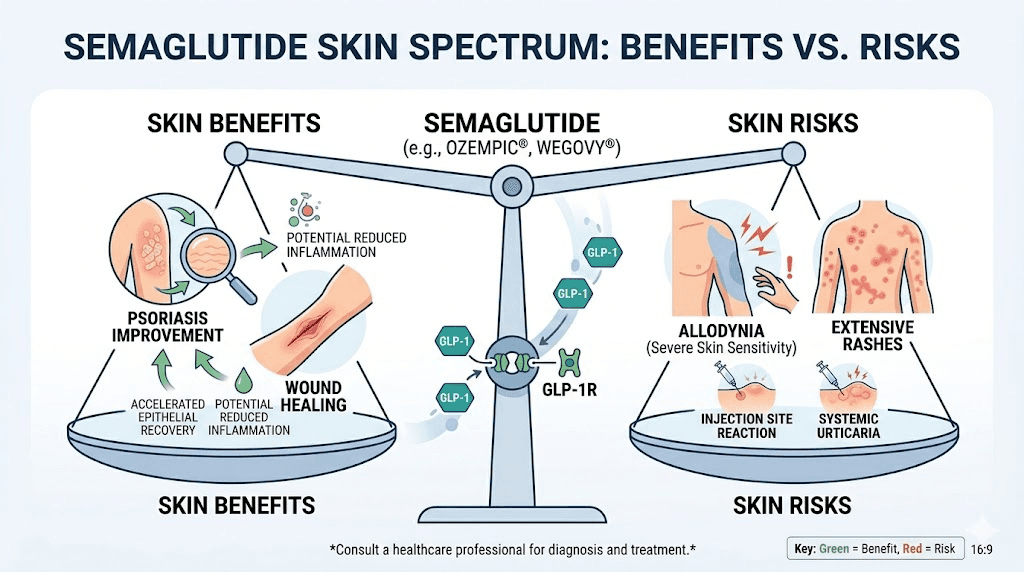
Skin benefits of semaglutide: the other side of the story
It would be incomplete to discuss semaglutide and skin without mentioning the documented benefits. The same immune-modulating properties that occasionally cause hypersensitivity reactions can also improve certain skin conditions significantly.
Psoriasis improvement
A randomized trial showed that semaglutide reduced median PASI (Psoriasis Area and Severity Index) scores from 21 to 10 across 12 weeks. Liraglutide, another GLP-1 agonist, showed even more dramatic improvement in one study, reducing PASI from 15.7 to 2.2 within 12 weeks. The mechanism involves suppression of pro-inflammatory cytokines TNF-alpha, IL-17, and IL-6, along with blockade of NF-kB signaling pathways. Histological examination showed decreased epidermal thickness, a direct measure of reduced psoriatic inflammation.
For people who have both obesity and psoriasis, semaglutide offers a rare dual benefit: weight loss and skin disease improvement from the same medication. This is a significant advantage over weight loss medications that have no effect on, or even worsen, inflammatory skin conditions.
Hidradenitis suppurativa reduction
Systematic reviews indicate that GLP-1 agonists reduce inflammatory nodules and disease flares in hidradenitis suppurativa. Response varies individually, but the anti-inflammatory mechanism provides a plausible explanation for improvement in this notoriously difficult-to-treat condition. People managing inflammatory conditions alongside weight loss goals may find this dual benefit particularly relevant.
Wound healing enhancement
GLP-1 receptor activation promotes angiogenesis (new blood vessel formation) and fibroblast activity, both of which accelerate wound healing. This benefit is particularly relevant for diabetic patients, who often experience delayed wound healing as a complication of their disease. The same monitoring tools that track weight loss can help track improvements in wound healing and skin health over time.
The paradox explained
How can semaglutide both help and harm the skin? The answer lies in the complexity of the immune system. GLP-1 receptor activation suppresses certain inflammatory pathways (beneficial for psoriasis and hidradenitis) while potentially activating others (harmful in hypersensitivity reactions). The net effect depends on individual immune genetics, pre-existing conditions, and specific triggers.
Think of it as a thermostat adjustment. For most people, the adjustment moves in the right direction. For some, it overshoots. And for a few, it triggers an alarm. Understanding which category you fall into, ideally before it becomes a crisis, is the goal of proactive monitoring.
Long-term skin monitoring on semaglutide
Skin sensitivity can develop at any point during semaglutide treatment, not just during dose escalation. The 75-year-old patient in the case report developed her skin condition after 10 months on semaglutide. This means ongoing vigilance is necessary even after you have tolerated the medication for months without issues.
Monthly skin check protocol
Once monthly, conduct a brief self-assessment. Check all injection sites for persistent changes like thickening, discoloration, or dimpling that could indicate lipodystrophy. Examine arms, legs, trunk, and face for any new rashes, spots, or texture changes. Note any changes in skin sensitivity to touch, temperature, or pressure. Document anything new with photographs for comparison.
This takes less than five minutes and creates a record that makes conversations with healthcare providers much more productive. Tracking this alongside your monthly progress metrics creates a complete picture of your treatment response.
When to alert your provider
Any new skin symptom that persists for more than two weeks deserves a mention to your healthcare provider. Sudden onset of widespread skin sensitivity, especially after a dose change, should be reported promptly. Skin changes accompanied by other new symptoms like unusual fatigue, dizziness, or persistent headaches warrant more urgent evaluation because they could indicate a systemic reaction rather than an isolated skin issue.
Maintaining treatment continuity
The goal of monitoring is not to find reasons to stop semaglutide. It is to catch problems early enough that they can be managed without disrupting treatment. Early intervention for skin reactions, whether through technique adjustment, dose modification, formulation change, or supportive care, preserves the long-term treatment trajectory that produces the best outcomes.
People who monitor proactively tend to stay on treatment longer and achieve better results than those who ignore symptoms until they become intolerable. A few minutes of monthly attention to skin health protects an investment of months or years of consistent treatment.
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions. SeekPeptides members access detailed side effect management guides, interaction databases, and direct support from experienced researchers who understand the nuances of GLP-1 therapy. When it comes to managing complex side effects like skin sensitivity, having access to SeekPeptides comprehensive protocol library and community knowledge can make the difference between abandoning treatment and finding a sustainable path forward.
Frequently asked questions
Does semaglutide cause skin sensitivity?
Yes. Clinical trial data show that oral semaglutide at 50 mg causes "sensitive skin" in 2.7% of users compared to 0% with placebo. A case series documents allodynia (painful skin sensitivity) at the 2.4 mg subcutaneous dose. Proper dose management and proactive monitoring help reduce risk.
Why does my skin hurt on semaglutide?
Painful skin on semaglutide may indicate allodynia, a condition where normal touch becomes painful. Published research shows this typically develops at higher doses (2.4 mg) and may involve altered nerve signaling through GLP-1 receptors on sensory neurons. Discuss dose adjustment with your healthcare provider.
How common are semaglutide skin reactions?
Injection site reactions occur in 1.4% to 2.4% of subcutaneous semaglutide users. Generalized skin sensory changes affect 1.2% to 2.7% of oral semaglutide users depending on the specific symptom. Serious reactions like angioedema and anaphylaxis are rare but documented. Understanding your dosage and administration helps contextualize your risk.
Will semaglutide skin sensitivity go away?
In many cases, yes. Injection site reactions typically resolve within days. Allodynia resolved in all patients who discontinued semaglutide and in one patient who continued treatment for four months. The semaglutide timeline for side effect resolution varies by reaction type and individual.
Can I switch to tirzepatide if semaglutide causes skin problems?
Potentially, yes. Tirzepatide has a different molecular structure and excipient profile. Cross-reactivity between GLP-1 agonists for skin reactions is not universal. Many people who cannot tolerate one GLP-1 medication tolerate another. Work with your healthcare provider and consider allergist consultation for serious reactions.
Does compounded semaglutide cause more skin reactions?
No direct comparison studies exist between compounded and brand-name semaglutide for skin reactions. However, compounded formulations contain different excipients, preservatives, and sometimes additional ingredients like B12 or glycine that could independently trigger reactions. If skin sensitivity appears after switching formulations, the excipients may be responsible.
Should I take an antihistamine before my semaglutide injection?
Pre-medication with antihistamines is not standard practice for semaglutide injections. However, if you have experienced mild urticaria or itching after previous doses, your healthcare provider may suggest taking cetirizine or loratadine 30 to 60 minutes before injection. This should be a medical decision, not self-directed.
Can semaglutide cause hair loss along with skin sensitivity?
Yes. Clinical trial data show alopecia rates of 6.9% with oral semaglutide 50 mg versus 0.3% with placebo. The GLP-1 hair loss guide covers this in detail. Hair loss and skin sensitivity may co-occur but likely involve different mechanisms, with hair loss related more to rapid weight loss and nutritional changes than to the same pathways driving skin sensitivity.
External resources
Allodynia associated with semaglutide: A case series (PubMed)
Dermal hypersensitivity reaction to semaglutide: Two case reports (PubMed)
A closer look at the dermatological profile of GLP-1 agonists (PMC)
Dermatologic findings associated with semaglutide use: A scoping review (JAAD)
Semaglutide-associated rash information (American Academy of Allergy, Asthma and Immunology)
In case I do not see you, good afternoon, good evening, and good night. May your skin stay comfortable, your doses stay tolerable, and your protocols stay effective.