Apr 4, 2026

Experienced researchers know something that newcomers learn the hard way. The peptide inside the vial matters less than the vendor behind it. You can have the most promising compound in the world, but if purity is inconsistent, if quantity deviates wildly from what the label claims, if third-party testing tells a different story than the certificate of analysis, then your research means nothing. NuScience Peptides has positioned itself as a premium supplier of retatrutide and other research compounds. The question is whether the data supports the claims.
This guide breaks down everything researchers need to know about NuScience Peptides retatrutide, from independent Finnrick testing scores and purity analysis to customer experiences and how the vendor stacks up against alternatives. We will examine eight individual test results, analyze quantity accuracy, review community feedback from forums and third-party platforms, and provide the context you need to make an informed sourcing decision.
Whether you are evaluating NuScience for the first time or comparing it against vendors you already use, the data here will give you a clear picture.
SeekPeptides tracks vendor quality data and testing results so researchers can make evidence-based sourcing decisions rather than relying on marketing claims alone.
What is NuScience Peptides?
NuScience Peptides is a United States based research peptide supplier that claims over 20 years of experience in the industry. The company positions itself as a provider of laboratory-grade peptides verified through HPLC and mass spectrometry analysis by independent third-party laboratories. Every batch, according to their marketing, undergoes rigorous testing to ensure premium purity and accuracy.
That is the pitch. Now for the substance.
The company offers a range of research peptides including retatrutide, semaglutide, tirzepatide, BPC-157, GHK-Cu, Melanotan II, PT-141, CJC-1295, Ipamorelin, and mazdutide. Their product catalog spans weight management peptides, recovery compounds, and cosmetic research peptides. All products carry the standard disclaimer that they are intended for research use only, not for human or veterinary application.
NuScience ships from within the United States and offers same-day shipping on orders placed before 12 PM EST. Orders above $200 qualify for free domestic shipping. The company publishes certificates of analysis on their website, though as we will discuss later, the reliability of vendor-published COAs versus independent third-party testing tells two very different stories sometimes.
What makes NuScience worth examining closely is not just their claims but the volume of independent testing data now available. Finnrick, a third-party product testing and supply chain transparency platform, has conducted 47 tests across 10 NuScience products. That sample size gives us something most vendor reviews lack: actual data.
NuScience retatrutide product overview
Before diving into test results, let us establish what NuScience actually sells when it comes to retatrutide. The company offers retatrutide (LY3437943, CAS 2381089-83-2) in four vial sizes: 2mg, 5mg, 10mg, and 20mg. This range gives researchers flexibility depending on the scale and duration of their retatrutide protocols.
The peptide arrives in lyophilized (freeze-dried) form, which is standard for research-grade peptides. Lyophilization preserves peptide stability during shipping and storage, and it is the format most researchers expect when ordering from any reputable supplier. If you have worked with lyophilized peptides before, the process is familiar. If you have not, reconstitution guides will walk you through mixing with bacteriostatic water.
NuScience claims 99%+ purity for their retatrutide. This is a common claim across research peptide vendors, and it is one that independent testing either confirms or challenges. We will get to those numbers shortly. The company also states that each batch comes with a certificate of analysis, though the specifics of who conducts that analysis and how it compares to independent results varies.
For researchers comparing retatrutide pricing across vendors, NuScience falls in the mid-to-premium range. The exact pricing fluctuates, but 5mg vials of retatrutide from research suppliers typically range from $250 to $400. Bulk orders generally reduce the per-unit cost by 15 to 25 percent.
Available vial sizes and their research applications
The 2mg vial works well for initial research protocols and pilot studies. It is the lowest commitment option and allows researchers to evaluate the product before scaling up. For those following starting dose protocols that begin at lower amounts, a 2mg vial may last through the initial phase.
The 5mg vial is the most commonly tested size. Seven of the eight Finnrick tests on NuScience retatrutide used 5mg vials. This is also the most popular size across the research peptide market generally, making it the easiest to compare across vendors.
The 10mg vial suits researchers running longer protocols or those who prefer fewer reconstitution events. Fewer vials means fewer opportunities for contamination and less time spent on preparation. The 20mg vial is designed for extended research applications or laboratory settings where higher volumes are needed.
Third-party testing and purity analysis
This is where claims meet reality. Independent third-party testing is the single most reliable way to evaluate any peptide vendor. What the company says on its website matters far less than what an unaffiliated laboratory measures in a controlled setting. Finnrick has tested NuScience retatrutide eight times between April 2025 and February 2026, and the results tell an interesting story.
The overall Finnrick rating for NuScience retatrutide is A (Great). Test scores average 7.2 out of 10, with no score falling below 6.0. That minimum threshold matters because it means none of the eight samples failed outright. Scores ranged from 6.1 at the low end to a perfect 10.0 at the high end.
Let us break that down test by test.
Individual test results
The most recent test, conducted on February 13, 2026, scored 7.5. The vial contained 4.89mg of retatrutide, which represents a -12.1% deviation from the labeled 5mg. Purity measured at 99.90%, and endotoxin testing came back below the limit of quantification. That endotoxin result is notable because not all vendors submit samples for endotoxin screening, and contamination at this level can compromise injection-based research.
The July 2025 test is the standout. Score: 10.0. Perfect. The vial contained 5.1mg (+2.0% from labeled), and purity measured at 100.00%. This is as good as it gets in peptide testing. However, one perfect score does not define a vendor. Consistency matters more than any single result.
Two tests from May 21, 2025 scored 6.9 and 6.4 respectively. The first vial contained 5.47mg (+9.4% over labeled amount) at 99.11% purity. The second contained 5.55mg (+11.0%) at 99.09% purity. Both showed overfill, which researchers generally prefer to underfill, but the purity numbers dipped slightly below the 99.5% mark that top-tier suppliers consistently hit.
The May 5, 2025 tests produced mixed results. One vial scored 6.1, the lowest in the dataset, with 6.11mg of peptide (+22.2% overfill) at 99.56% purity. The other scored 7.0 with 5.06mg (+1.2%) but only 93.68% purity. That 93.68% figure deserves attention. While still above 90%, it falls meaningfully short of the 99%+ claim on the label.
Two April 2025 tests scored 6.8 and 7.0. Quantities were 5.27mg (+5.4%) and 5.19mg (+3.8%). Purity measured 94.56% and 93.88% respectively. Again, both below the marketed 99%+ threshold.
What the data actually tells us
Three important patterns emerge from these eight tests. First, purity improved significantly over time. The earliest tests (April and May 2025) showed purities in the 93 to 95% range. By mid-2025, results climbed to 99%+. The February 2026 test measured 99.90%. This suggests NuScience either changed suppliers, improved their purification process, or both. Researchers ordering today are likely receiving higher-purity product than those who ordered in early 2025.
Second, quantity accuracy varies. The deviation from labeled amount ranged from -12.1% (underfill) to +22.2% (significant overfill). Most samples showed overfill, which means researchers generally received more peptide than they paid for. But the +22.2% result is concerning in a different way. It suggests inconsistent fill processes. A vial that contains 22% more than advertised might also sometimes contain 22% less. The retatrutide dosage calculator depends on accurate vial contents, so significant deviation in either direction introduces variables into research.
Third, the overall trajectory is positive. If we weight recent tests more heavily, as most researchers should, the picture looks good. The two most recent tests (February 2026 and July 2025) scored 7.5 and 10.0 with purities of 99.90% and 100.00%. That is strong performance by any standard.
Understanding retatrutide as a research peptide
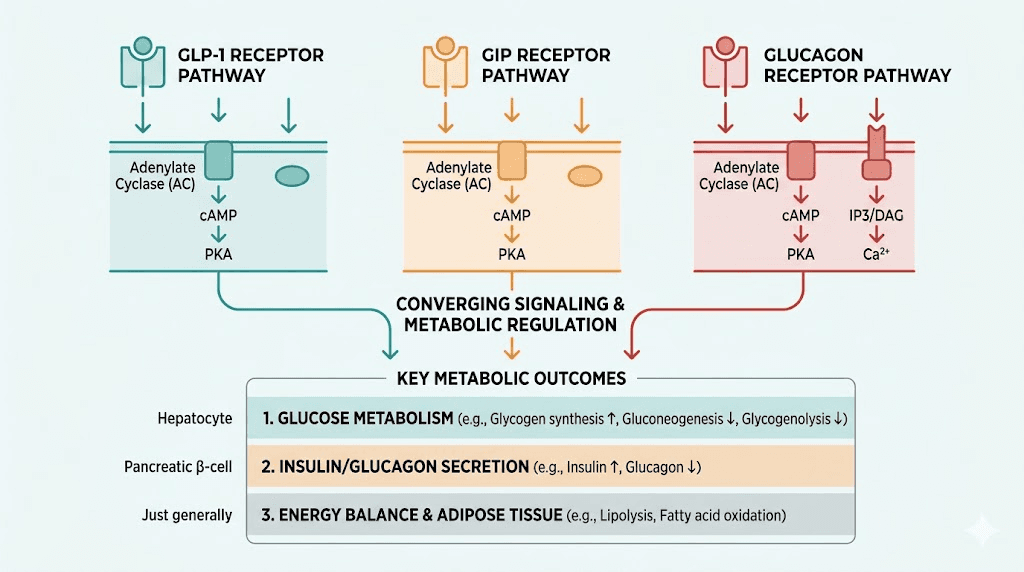
Retatrutide (LY3437943) represents the next generation of incretin-based research compounds. Unlike semaglutide, which acts as a single GLP-1 receptor agonist, or tirzepatide, which targets both GLP-1 and GIP receptors, retatrutide is a triple agonist. It activates GLP-1, GIP, and glucagon receptors simultaneously.
That triple mechanism is what makes retatrutide the most discussed research peptide in the weight management space right now.
Phase 2 clinical trial data showed body weight reductions of up to 24.2% over 48 weeks at the highest doses tested. To put that in perspective, semaglutide achieved approximately 15% and tirzepatide approximately 21% in their respective trials. The glucagon receptor component adds a metabolic dimension that previous compounds lacked, increasing energy expenditure while simultaneously reducing appetite through GLP-1 and GIP pathways.
Developed by Eli Lilly, retatrutide is currently in Phase 3 clinical trials for obesity and type 2 diabetes. It has not received FDA approval for any indication, which is why all commercially available retatrutide exists in the research-use-only category. This regulatory status means quality control falls entirely on the vendor and, more importantly, on independent testing platforms that hold vendors accountable.
Why vendor quality matters more for retatrutide
Retatrutide is a complex peptide. At approximately 4.2 kDa molecular weight, proper synthesis and purification require sophisticated manufacturing capabilities. The structural differences between retatrutide and simpler peptides mean that impurities from incomplete synthesis or degradation can vary more widely between batches.
This complexity makes third-party testing essential, not optional. A vendor that consistently delivers 99%+ purity on a relatively simple peptide like BPC-157 might struggle with the manufacturing demands of retatrutide. Finnrick testing data allows researchers to evaluate vendor performance on a peptide-by-peptide basis rather than assuming quality is uniform across the catalog.
For researchers exploring retatrutide dosing protocols, purity directly impacts research outcomes. A 5% impurity means 5% of the vial contents are not the compound being studied, introducing unknown variables into any protocol. The dosing schedule assumes a known quantity of active compound, so purity deviations compound over time.
The triple agonist mechanism explained
Understanding what retatrutide does helps explain why sourcing quality matters. The GLP-1 receptor activation reduces appetite and slows gastric emptying, similar to what semaglutide does. GIP receptor activation enhances insulin sensitivity and appears to improve the fat storage and mobilization balance. The glucagon receptor component is the differentiator, it increases hepatic glucose output and promotes thermogenesis, essentially telling the body to burn more energy.
These three pathways working together create a more comprehensive metabolic effect than either single or dual agonists. Research has shown that the glucagon component specifically targets visceral fat reduction and hepatic lipid metabolism, which are areas where single-agonist compounds show less activity.
For researchers transitioning from other compounds, understanding the switch from tirzepatide to retatrutide or timing considerations requires knowing exactly what is in the vial. This circles back to why vendor evaluation is not just a purchasing decision but a research integrity decision.
How NuScience compares to other retatrutide vendors
Vendor comparison requires apples-to-apples data, which is exactly what Finnrick provides. Looking at retatrutide specifically across the vendors they have tested, NuScience rating of A (Great) with 8 samples and a 7.2 average score places it in the upper tier. But it is not alone there.
Paradigm Peptides, one of the most extensively tested vendors on the platform, holds an A rating based on significantly more samples across their full product catalog. Paradigm Peptides retatrutide has been a popular choice among researchers for its consistency. Other vendors like Peptide Sciences and Simple Peptide also offer retatrutide, each with varying levels of independent testing data.
What separates NuScience from lower-tier vendors is the consistency of acceptable results. Eight tests with no score below 6.0 demonstrates baseline reliability. Some vendors show wild swings, scoring 9 on one test and 3 on the next. That kind of inconsistency makes research planning impossible.
NuScience product quality across their full catalog
A vendor is only as strong as their weakest product. Looking beyond retatrutide, NuScience Finnrick results across all 10 tested products show a mixed picture that researchers should understand.
The standout performers include tirzepatide (A rating, 8.0 average, 6 tests), Melanotan II (A rating, 7.5 average, 6 tests), and semaglutide (B rating, 7.8 average, 14 tests). The semaglutide result is particularly noteworthy because 14 tests provide high statistical confidence. A 7.8 average across that many samples suggests consistent quality.
The concerning results come from CJC-1295 (D rating, 4.1 average, 3 tests) and Ipamorelin (D rating, 4.3 average, 2 tests). These are tentative ratings due to small sample sizes, but D-grade scores indicate significant quality issues in those specific products. Researchers interested in NuScience for growth hormone secretagogue research should approach with caution until more testing data becomes available.
PT-141 received a C rating (5.7 average, 2 tests), which is middling. BPC-157 and mazdutide received tentative B ratings based on only 2 tests each.
The takeaway is clear. NuScience appears to perform well with the compounds that likely represent their highest sales volume, which are weight management peptides like retatrutide, semaglutide, and tirzepatide. Performance drops for less popular products in their catalog. This pattern is common across peptide vendors and likely reflects where they invest most heavily in quality control.
Pricing context in the retatrutide market
The retatrutide market has matured significantly since the compound first became available from research suppliers. Prices have come down as more vendors entered the space and manufacturing scaled up. NuScience pricing sits in the mid-to-premium range, which aligns with vendors that invest in quality control and third-party testing.
Budget vendors often undercut on price but deliver lower purity or inaccurate quantities. The Finnrick data showing some NuScience vials with 93-95% purity in early batches illustrates that even mid-tier vendors can have quality variation. For researchers deciding between a cheaper option and NuScience, the independent testing data provides a concrete basis for comparison rather than relying on vendor self-reported numbers.
When evaluating where to buy retatrutide, consider the total cost of research, not just the vial price. A cheaper vial with 93% purity delivers less active compound per dollar than a more expensive vial at 99.9% purity. The peptide cost calculator can help quantify this difference.
Reconstitution and storage for NuScience retatrutide
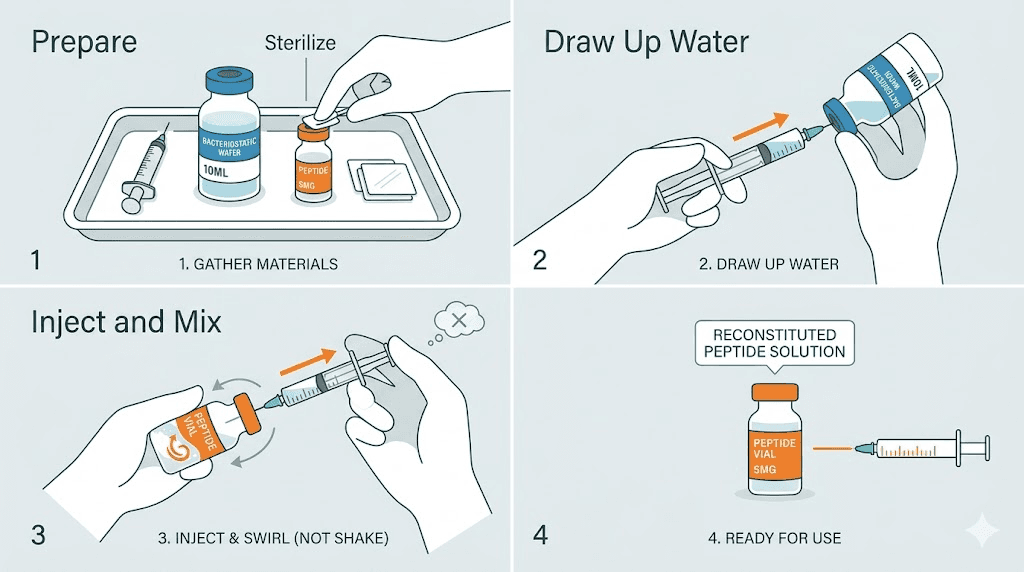
NuScience retatrutide arrives as a lyophilized powder, and proper reconstitution is critical for maintaining the purity that independent testing confirmed. Poor reconstitution technique can degrade even the purest peptide, making your vendor evaluation irrelevant if handling protocols are not followed.
The basic process is straightforward. Draw bacteriostatic water into a sterile syringe. Inject it slowly along the inside wall of the vial, allowing it to run down the glass rather than hitting the lyophilized cake directly. Let the solution sit for several minutes, gently swirling if needed. Never shake. Vigorous agitation can denature the peptide chains and reduce effective purity.
How much bacteriostatic water to use
The amount of bacteriostatic water you add determines the concentration of your reconstituted solution, which directly affects dosing accuracy. For a standard 5mg vial, common reconstitution volumes include 1ml and 2ml. Our reconstitution guide for 10mg retatrutide covers the math in detail, and the principles apply proportionally to other vial sizes.
With 1ml of bacteriostatic water in a 5mg vial, each 0.1ml (10 units on an insulin syringe) contains 500mcg of retatrutide. With 2ml, each 0.1ml contains 250mcg. The higher concentration (1ml) works well for protocols requiring larger doses, while the lower concentration (2ml) provides finer dose control for microdosing protocols or initial dose titration.
However, remember the quantity accuracy data from Finnrick testing. A vial labeled 5mg might contain 4.89mg or 6.11mg based on the test results we reviewed. This means your actual concentration could differ from the calculated concentration by up to 22%. The retatrutide dosage calculator assumes accurate vial contents, so researchers should factor in potential quantity deviation when interpreting their results.
Storage requirements
Unreconstituted lyophilized retatrutide should be stored in a freezer (-20 degrees Celsius) for long-term storage or in a refrigerator (2-8 degrees Celsius) for storage periods under a few months. The complete retatrutide storage guide covers optimal conditions in detail.
Once reconstituted with bacteriostatic water, the solution must be refrigerated and used within a reasonable timeframe. Most researchers aim to use reconstituted peptide within 4 to 6 weeks. Shelf life considerations depend on storage temperature, whether the vial was stored correctly before reconstitution, and how cleanly the reconstitution was performed.
Temperature exposure during shipping is another factor to consider. NuScience ships from within the United States, which typically means shorter transit times than international orders from Chinese manufacturers. Shorter shipping reduces the window for temperature-related degradation, especially during summer months when ambient temperatures can exceed safe storage ranges.
Dosing considerations for retatrutide research
Retatrutide dosing in clinical trials followed a gradual escalation approach, starting at 1mg weekly and increasing over time. The Phase 2 trial tested doses up to 12mg weekly, with the highest doses producing the most significant metabolic effects. Researchers designing protocols should understand the complete dosage chart for weight management applications before beginning.
The standard approach begins low. Very low.
Starting at 1mg per week allows assessment of tolerance and response. The dose schedule typically increases by 1mg every four weeks until reaching the target research dose. This slow titration minimizes gastrointestinal side effects, which are the most commonly reported adverse events with all GLP-1 receptor agonists.
For researchers coming from other compounds, the transition from high-dose tirzepatide to retatrutide requires specific considerations. The addition of the glucagon receptor component means retatrutide is not simply a stronger version of tirzepatide. It activates different metabolic pathways, and the body response profile can differ significantly from what researchers observed with dual agonists.
Why accurate vial contents matter for dosing
This is where vendor quality directly impacts research outcomes. Let us work through a concrete example. Suppose your protocol calls for 4mg of retatrutide weekly. You order a 5mg vial from NuScience, reconstitute with 2ml of bacteriostatic water, and calculate that 1.6ml of solution delivers your 4mg dose.
If the vial actually contains 4.89mg (as the February 2026 Finnrick test showed), your 1.6ml draw delivers 3.91mg instead of 4mg. That is a manageable 2.3% deviation. But if the vial contains 6.11mg (as the May 2025 test showed), the same 1.6ml draw delivers 4.89mg, which is a 22.2% overdose. Over a multi-week research protocol, that kind of deviation accumulates.
The practical implication is that researchers should not assume any vial contains exactly what the label states. Using the peptide calculator to determine theoretical doses is a good starting point, but understanding that actual content may deviate by 10-20% in either direction provides a more realistic framework for interpreting results.
Retatrutide for different research applications
The compound versatility has driven interest across multiple research domains. Bodybuilding and athletic research applications focus on the compound ability to reduce adipose tissue while potentially preserving lean mass. The question of whether retatrutide causes muscle loss remains an active area of investigation.
Weight management research represents the primary use case, with protocols designed around the weight-loss-specific dosage charts. The triple agonist mechanism provides theoretical advantages over dual agonists like tirzepatide (Mounjaro) for metabolic research.
Some researchers explore combination protocols, pairing retatrutide with compounds like tesamorelin or cagrilintide for specialized applications. These stacking protocols require even more precise dosing, which makes vendor quality and quantity accuracy critically important. Understanding how cagrilintide compares to retatrutide helps researchers determine whether combination approaches are warranted.
Customer experiences and community feedback
Third-party testing data tells you about the product. Customer reviews tell you about the experience. Both matter. NuScience Peptides has a presence across multiple review platforms and peptide forums, and the feedback follows a recognizable pattern.
The positives are consistent. Customers frequently praise shipping speed, customer service responsiveness, and product quality. One reviewer on Peptide Critic described the experience as exceptional, noting that customer service went above and beyond when handling a lost order, including assistance with insurance claims and a free upgrade to priority mail. Multiple forum posts on The Iron Den and ExcelMale reference fair prices, great communication, and quick turnaround.
But the picture is not uniformly positive.
Some users have reported concerns about certificate of analysis accuracy. One reviewer noted that they could not find third-party COA results on the products they checked, which they considered a significant red flag. Others reported minor product issues, including bacteriostatic water vials that contained 2ml instead of the labeled 3ml. While this does not directly relate to peptide quality, it raises questions about quality control across the product line.
A handful of users reported injection site reactions, including localized burning with certain peptides. However, injection site reactions can result from many factors, including reconstitution technique, injection depth, and individual sensitivity, so attributing these solely to product quality would be premature without controlled testing.
Forum sentiment analysis
Across bodybuilding forums, TikTok reviews, and peptide-specific communities, NuScience occupies a middle position in terms of reputation. They are not the most recommended vendor, a distinction that typically goes to long-established names with thousands of independent tests. But they are also not flagged as problematic. The general consensus is that NuScience is a legitimate vendor with acceptable product quality, especially for their higher-volume products like retatrutide and semaglutide.
The TikTok presence is worth noting. NuScience has generated significant discussion on the platform, with multiple review videos. Social media reviews should always be taken with skepticism, as some may be sponsored or incentivized, but the volume of organic discussion suggests genuine customer engagement.
For researchers who value peer feedback, checking current forum threads before ordering is always wise. Community sentiment can shift quickly if quality issues emerge, and real-time forum discussions often surface problems before they appear in formal reviews.
Red flags to watch for when buying retatrutide
Whether you are considering NuScience or any other vendor, certain warning signs should trigger caution. The grey market for research peptides includes both legitimate suppliers and operations that cut corners on quality, purity testing, or both.
The most serious red flag is a complete absence of third-party testing. Any vendor can produce an in-house certificate of analysis. The equipment exists, the templates exist, and the incentive to produce flattering numbers is obvious. What separates trustworthy vendors from the rest is submission to independent testing platforms like Finnrick, where the vendor has no control over the results. NuScience retatrutide has been tested eight times independently, which at minimum demonstrates willingness to be evaluated.
Pricing that seems too good to be true usually is. Retatrutide is an expensive peptide to synthesize properly. Vendors offering it at dramatically below market rates may be selling lower-purity product, underfilled vials, or degraded material. The Finnrick data showing some NuScience samples at 93-94% purity in early batches demonstrates that even mid-tier vendors can have purity issues, so budget vendors with no testing data are a gamble.
Specific warning signs
Watch for vendors that only provide internal COAs without independent verification. Watch for inconsistent or unavailable batch numbers. Watch for shipping from unexpected countries without cold chain packaging. Watch for products that arrive as liquid rather than lyophilized, as reconstituted peptides have much shorter shelf lives and are more susceptible to degradation during shipping.
Lack of customer service responsiveness is another warning. NuScience has received consistent praise for their customer service, which is actually one of the most reliable indicators of a legitimate operation. Vendors that are difficult to reach, that do not respond to quality complaints, or that lack a physical presence tend to be less reliable across the board.
For researchers exploring where to buy retatrutide, compiling testing data from multiple sources and cross-referencing community feedback provides the most complete picture. No single data point, whether testing scores, forum reviews, or pricing, should drive the decision alone.
How to evaluate any peptide vendor
SeekPeptides recommends a systematic approach to vendor evaluation that goes beyond marketing claims. The framework below applies to NuScience and any other supplier you might consider for retatrutide or other research peptides.
Step 1: Check independent testing data
Start with Finnrick or similar independent testing platforms. Look for the specific product you plan to order, not just overall vendor ratings. As we saw with NuScience, a vendor can earn an A rating for retatrutide while receiving D ratings for CJC-1295 and Ipamorelin. Product-level data matters more than vendor-level averages.
Key metrics to examine include purity percentage, quantity accuracy, and test score trends over time. A vendor whose scores are improving suggests investment in quality. A vendor whose scores are declining suggests the opposite. NuScience retatrutide purity improved from the 93-95% range to 99.9%+ over approximately 10 months, which is a positive trajectory.
Step 2: Verify certificate of analysis claims
Request the COA for the specific batch you are ordering. Compare the vendor-reported purity against any available independent test data for similar batches. Significant discrepancies between vendor COAs and independent results should raise questions. A vendor that claims 99.9% purity on their COA while independent testing shows 93% is either using a different analytical method or presenting inaccurate data.
Check whether the COA includes batch numbers, testing dates, analytical method details (HPLC column specifications, mobile phase composition), and the identity of the testing laboratory. Generic COAs without these details provide minimal assurance.
Step 3: Evaluate the product range context
Vendors that specialize in a narrow range of peptides often deliver higher quality than those offering enormous catalogs. A company selling 200 different peptides likely sources from multiple manufacturers with varying quality standards. NuScience offers a focused catalog of approximately 10 products, which suggests more concentrated quality control compared to vendors selling dozens or hundreds of compounds.
Look at which products receive the most testing attention on independent platforms. High-volume products typically receive more scrutiny and more investment in quality. For NuScience, semaglutide (14 tests) and retatrutide (8 tests) are the most thoroughly evaluated, while some products have only 2 tests, making those ratings less reliable.
Step 4: Assess shipping and handling practices
Domestic shipping (within the United States for US-based researchers) generally means shorter transit times and less temperature exposure. NuScience ships domestically with same-day processing for orders placed before noon. This reduces the risk of temperature-related degradation, particularly important for peptides like retatrutide that can lose potency when exposed to elevated temperatures.
Compare this to bulk retatrutide from international manufacturers, where shipping times can stretch to weeks and temperature control during transit is minimal. The price savings from international sourcing must be weighed against the risk of receiving degraded product.
Step 5: Monitor community feedback over time
A vendor reputation is not static. Companies can improve or decline in quality over time. Regular monitoring of forum discussions, review platforms, and independent testing results provides ongoing assurance that your chosen vendor maintains the standards that initially earned your business. NuScience improving purity from 93% to 99.9% over 10 months is exactly the kind of trajectory that ongoing monitoring reveals.
Retatrutide safety profile and side effects
No vendor review would be complete without addressing the compound safety profile, because the highest-purity peptide in the world still carries risks that researchers must understand. Retatrutide side effects in clinical trials were predominantly gastrointestinal, consistent with the GLP-1 receptor agonist class.
Nausea was the most commonly reported side effect, affecting a significant percentage of trial participants at higher doses. Diarrhea occurred in some research subjects, particularly during dose escalation phases. Decreased appetite, which is essentially the intended mechanism of action, was reported as a side effect by participants who experienced it as excessive. Vomiting, constipation, and injection site reactions rounded out the most frequent adverse events.
The question of whether retatrutide causes fatigue comes up frequently. Clinical trial data showed some incidence of fatigue and decreased energy, though distinguishing between a direct pharmacological effect and the metabolic consequences of significant caloric deficit can be challenging. GLP-1 related fatigue is a common concern across the entire compound class.
Hair loss concerns have also emerged in community discussions, though clinical trial data on this specific side effect is limited. The mechanism, if it exists, likely relates to the metabolic stress of rapid weight change rather than a direct effect of the peptide itself. Researchers monitoring for this should track changes systematically rather than attributing isolated observations to the compound.
The glucagon component and unique considerations
Because retatrutide is the first widely available triple agonist in the research peptide space, some of its effects do not have direct precedent from semaglutide or tirzepatide research. The glucagon receptor activation increases hepatic glucose output, which could theoretically cause blood sugar fluctuations in certain research contexts. It also promotes thermogenesis, meaning researchers may observe increased body temperature or sweating at higher doses.
Skin sensitivity has been reported by some researchers, potentially related to the metabolic shifts induced by the triple agonist mechanism. This is another area where long-term data is still accumulating.
The interaction between retatrutide and other compounds is not well characterized outside of clinical trial settings. Researchers considering combination protocols with tesamorelin, cagrilintide, or other peptides should proceed with caution and thorough documentation. The comparison between mazdutide and retatrutide provides context on how different multi-agonist approaches compare in terms of side effect profiles.
Injection technique and administration
Proper injection technique reduces the incidence of local side effects. Injection site selection matters, with subcutaneous administration into the abdomen, thigh, or upper arm being the standard approach. Rotating injection sites prevents lipodystrophy and reduces localized irritation.
The complete injection guide for retatrutide covers proper technique in detail, including needle gauge selection, injection depth, and post-injection care. For researchers using insulin syringes, understanding syringe specifications ensures accurate dose delivery.
Alternative administration routes like intranasal delivery are being explored in some research contexts, though subcutaneous injection remains the standard based on clinical trial protocols.
NuScience versus the broader vendor landscape
Putting NuScience in context requires understanding where they fit in the overall research peptide ecosystem. The market broadly divides into four tiers based on quality, testing, and reputation.
Tier one includes vendors with thousands of independent tests, consistently high scores, and deep community trust built over many years. These are the names that appear on every credible recommendation list. Tier two includes vendors with moderate testing data, generally positive results, and growing reputations. NuScience falls here. They have enough independent data to evaluate meaningfully, their results are predominantly positive, and community feedback trends favorably.
Tier three includes vendors with minimal independent testing, inconsistent results, or limited track records. Tier four includes vendors with no testing data, negative community feedback, or obvious quality concerns.
The progression from tier two to tier one requires time, continued testing submissions, and maintained quality. NuScience trajectory, with improving purity scores and expanding Finnrick testing coverage, suggests movement in the right direction. But they have not yet accumulated the depth of data that the most established vendors have.
When NuScience makes sense as a choice
For researchers specifically seeking retatrutide, NuScience A rating on that specific product makes it a defensible choice. The eight independent tests provide reasonable confidence in product quality, and the improving purity trend suggests recent batches are better than early ones. If retatrutide is your primary compound, the data supports NuScience as a viable option.
For researchers who need multiple peptides from a single vendor, the picture is more nuanced. NuScience excels with weight management peptides but shows concerning results for growth hormone secretagogues. If your research requires both retatrutide and CJC-1295, for example, you might consider NuScience for the retatrutide and a different vendor for the CJC-1295 based on product-specific testing data.
For bulk orders, the free shipping threshold of $200 provides modest cost savings on larger purchases. The bulk retatrutide purchasing guide covers additional considerations for high-volume research procurement.
Reconstitution math and practical calculations
Getting the math right prevents costly mistakes. Here is a practical walkthrough for NuScience retatrutide vials using the most common sizes.
5mg vial calculations
Adding 1ml of bacteriostatic water creates a concentration of 5mg/ml, or 5000mcg/ml. On a standard 100-unit insulin syringe, each unit equals 0.01ml, which contains 50mcg of retatrutide. For a 1mg (1000mcg) dose, you would draw 20 units. For a 2mg dose, 40 units. For a 4mg dose, 80 units.
Adding 2ml of bacteriostatic water creates a concentration of 2.5mg/ml, or 2500mcg/ml. Each syringe unit then contains 25mcg. A 1mg dose requires 40 units. A 2mg dose requires 80 units. This lower concentration provides more precise dose control, which is valuable during the initial escalation phase.
Remember, the peptide reconstitution calculator handles these calculations automatically. Enter your vial size, water volume, and desired dose, and it returns the exact draw volume in syringe units.
10mg and 20mg vial calculations
Larger vials follow the same mathematical principles but offer different concentration options. A 10mg vial reconstituted with 2ml of bacteriostatic water produces a 5mg/ml solution, identical to the concentration from a 5mg vial with 1ml. The advantage is fewer reconstitution events over the course of a longer protocol.
For the 20mg vial, reconstitution with 4ml creates the same 5mg/ml concentration while providing enough solution for extended research periods. Higher-volume reconstitutions require more bacteriostatic water, so researchers should ensure they have adequate supplies before beginning.
The retatrutide dosage calculator on SeekPeptides accommodates all vial sizes and reconstitution volumes, making these calculations straightforward regardless of which NuScience product you order.
Long-term research considerations
Researchers planning extended protocols face additional considerations beyond initial product quality. Batch-to-batch consistency becomes important when a protocol spans multiple vials purchased over weeks or months. The Finnrick testing data showing quantity deviations from -12% to +22% for NuScience retatrutide means that consecutive vials from different batches may deliver meaningfully different amounts of peptide per unit volume.
One approach to mitigating this variability is ordering all vials for a complete protocol from a single batch, if the vendor allows batch-specific ordering. This does not guarantee identical contents across vials, but it increases the likelihood of consistency compared to ordering vials manufactured at different times.
Storage duration also affects quality over time. Lyophilized peptide stored properly in a freezer maintains stability for months, but researchers who order months of supply should verify that their storage conditions remain consistent. Temperature fluctuations from frequent freezer openings, power outages, or improper seal integrity can degrade peptides faster than expected. The retatrutide storage guide covers best practices for maintaining potency over extended periods.
Tracking research outcomes
Given vendor variability, meticulous record-keeping becomes essential. Documenting the batch number, reconstitution date, water volume used, calculated concentration, and any observable characteristics of each vial (color, dissolution time, clarity of solution) creates a paper trail that helps identify quality issues if research outcomes become inconsistent.
Researchers should also note when they switch to a new vial and watch for changes in response that might indicate a different actual concentration. This is not unique to NuScience. Every research peptide vendor has batch variability. The difference is whether you account for it in your experimental design or discover it retroactively when results stop making sense.
SeekPeptides provides tracking tools and protocol management resources that help researchers maintain detailed logs throughout their programs.
This systematic approach to documentation transforms vendor variability from a confounding factor into a measured variable.
Comparing administration routes for retatrutide
While subcutaneous injection remains the standard administration route based on clinical trial methodology, the research community has explored alternatives. Intranasal retatrutide delivery has generated interest due to convenience and potential for altered pharmacokinetics.
Subcutaneous injection provides reliable bioavailability and predictable absorption kinetics. The clinical trial data that established retatrutide efficacy used this route exclusively, which means any departure introduces additional variables into research. For researchers using NuScience retatrutide, the injectable route maximizes comparability with published data.
The choice between injectable and oral peptide formats depends on the specific research question. Oral peptides face significant bioavailability challenges due to gastrointestinal degradation, though formulation advances are improving this limitation. For retatrutide specifically, the injectable format remains standard in virtually all research settings.
Needle gauge, injection site rotation, and proper aseptic technique all influence research outcomes independently of product quality. Even a perfectly pure vial of retatrutide can produce inconsistent results if injection technique varies. Our detailed injection guide provides standardized protocols that minimize technique-related variability.
The broader GLP-1 research landscape
Retatrutide exists within a rapidly expanding class of incretin-based research compounds. Understanding where it fits helps researchers make informed decisions about which compound best serves their specific research questions.
Semaglutide, tirzepatide, and retatrutide represent three generations of incretin-based compounds. Semaglutide targets GLP-1 receptors alone. Tirzepatide targets GLP-1 and GIP receptors. Retatrutide targets all three, adding glucagon. Each additional receptor target broadens the metabolic impact but also increases the complexity of the compound interaction with body systems.
For researchers who have already worked with semaglutide or tirzepatide, retatrutide offers a natural next step in studying multi-receptor agonism. The semaglutide versus tirzepatide comparison provides foundational context for understanding how dual agonism differs from single agonism, which then extends to understanding how triple agonism differs from dual.
Emerging compounds like survodutide and mazdutide add further options to the research toolkit. The competitive landscape continues to evolve, with new clinical data frequently reshaping understanding of optimal multi-receptor targeting strategies.
Why vendor quality matters across the GLP-1 class
As the research peptide market grows, so does the number of vendors offering these compounds. The quality range is enormous. Some vendors deliver pharmaceutical-grade purity consistently. Others sell degraded, underfilled, or contaminated product. The vendor you choose literally determines whether your research data is valid.
NuScience has demonstrated competency with weight management peptides specifically. Their Finnrick scores for retatrutide (A), tirzepatide (A), and semaglutide (B) suggest that this category represents their strength. Researchers working within the GLP-1 class can view these scores as a reasonable indicator of what to expect.
The complete GLP-1 purchasing guide covers vendor evaluation across the full compound class, including considerations specific to each peptide.
Practical tips for first-time NuScience customers
If you decide to order retatrutide from NuScience based on the data reviewed here, several practical steps can optimize your experience.
Order a small quantity first. A single 5mg vial allows you to evaluate product quality, shipping speed, packaging, and reconstitution characteristics before committing to a larger order. This initial order also provides a baseline for comparison if you order again later.
Request the COA for your specific batch. Compare the vendor-reported purity against the Finnrick testing data we reviewed. If NuScience recent batches are truly hitting 99.9% purity as the February 2026 test showed, the COA should reflect similar numbers. Significant discrepancies warrant further investigation.
Document everything about the vial upon receipt. Note the appearance of the lyophilized cake (color, texture, amount), the batch number, and any packaging details. Photograph the vial before and after reconstitution. This documentation becomes valuable if you need to troubleshoot unexpected results later.
Plan your reconstitution in advance. Have all supplies ready: bacteriostatic water, appropriate syringes and needles, alcohol swabs, and a clean workspace. Rushing the reconstitution process increases the risk of contamination and errors in water volume, both of which compromise your research.
Start with a conservative dose regardless of your experience level with other compounds. Retatrutide triple agonist mechanism means it interacts with the body differently than semaglutide or tirzepatide. The recommended starting approach exists for good reason, and even researchers experienced with other GLP-1 compounds should respect the titration schedule.
Understanding Finnrick testing methodology
Since we have relied heavily on Finnrick data throughout this review, understanding their testing methodology adds important context. Finnrick is an independent product testing and supply chain transparency platform that purchases samples directly from vendors under conditions that mirror real customer experiences. This means the product they test is the product you would receive.
Their testing evaluates three primary metrics. Purity, measured via HPLC, indicates what percentage of the vial contents is the advertised compound. Quantity accuracy compares the actual peptide mass against the labeled amount. Endotoxin testing, when performed, checks for bacterial contamination that could compromise research involving biological systems.
The Finnrick score combines these metrics into a single number on a 1-10 scale. Scores of 8-10 indicate excellent quality. Scores of 6-7 indicate acceptable quality with minor deviations. Scores below 6 indicate significant issues. The letter grade (A through F) reflects the overall vendor performance for that specific product based on all tests conducted.
NuScience retatrutide scoring profile, with no tests below 6.0, places it firmly in the acceptable-to-excellent range. The two tests scoring below 7.0 (6.1 and 6.4) were from May 2025, and both were affected by quantity overfill rather than purity issues at that level. The more recent tests show clear improvement, which is exactly the trajectory researchers want to see.
Frequently asked questions
Is NuScience Peptides a legitimate vendor?
Yes, NuScience Peptides is a legitimate US-based research peptide supplier with over 20 years of claimed industry experience. Independent Finnrick testing across 47 samples and 10 products confirms they deliver actual peptide products, though quality varies by product category. Their retatrutide specifically earned an A (Great) rating.
What is the purity of NuScience retatrutide?
Independent testing shows NuScience retatrutide purity ranging from 93.68% to 100.00% across eight samples. Recent tests (mid-2025 onward) consistently show 99%+ purity, while earlier batches from April-May 2025 measured in the 93-95% range. The improving trend suggests current batches are likely high-purity. Use the retatrutide dosage calculator for accurate dose planning.
How does NuScience retatrutide compare to other vendors?
NuScience Finnrick A rating for retatrutide places it in the upper tier of tested vendors. Paradigm Peptides and Peptide Sciences are also well-regarded options. The key differentiator is product-specific testing data, as vendor performance varies across different peptides.
What sizes does NuScience retatrutide come in?
NuScience offers retatrutide in 2mg, 5mg, 10mg, and 20mg vials. All arrive as lyophilized powder requiring reconstitution with bacteriostatic water before use. The 5mg vial is the most commonly ordered and most frequently tested size.
Does NuScience offer free shipping?
NuScience provides free US shipping on orders above $200 and offers same-day shipping for orders placed before 12 PM EST. Domestic shipping from within the United States reduces transit time and temperature exposure compared to international suppliers.
How should I store NuScience retatrutide?
Store unreconstituted lyophilized retatrutide in a freezer (-20 degrees Celsius) for long-term storage or refrigerator (2-8 degrees Celsius) for shorter periods. Once reconstituted, keep refrigerated and use within 4-6 weeks. The complete storage guide covers all conditions in detail.
Are NuScience COAs reliable?
NuScience publishes certificates of analysis for their products, but independent Finnrick testing sometimes shows different numbers than vendor-reported values. Some community members have noted difficulty finding third-party COAs on the website. Cross-referencing vendor COAs against independent testing data is always recommended.
What are the side effects of retatrutide?
Common side effects in clinical trials include nausea, diarrhea, decreased appetite, vomiting, and constipation. The triple agonist mechanism may also produce effects not seen with single or dual agonists, including increased thermogenesis. Concerns about hair loss and fatigue have been discussed in community forums.
What is the best starting dose for retatrutide?
Clinical trial protocols started at 1mg weekly with gradual escalation. The starting dose guide and dose schedule provide detailed escalation frameworks. Starting low and titrating slowly minimizes gastrointestinal side effects and allows for tolerance assessment.
Can I combine retatrutide with other peptides?
Some researchers explore combinations with tesamorelin or cagrilintide, though interaction data is limited. The triple agonist mechanism already provides broad receptor coverage, so combination protocols should be approached carefully. SeekPeptides members access detailed stacking guides and interaction information.
External resources
In case I do not see you, good afternoon, good evening, and good night. May your vials stay pure, your data stay consistent, and your research stay rigorous.