Apr 4, 2026

At the cellular level, retatrutide triggers something no other weight loss peptide does. Three receptors activate simultaneously. GLP-1. GIP. Glucagon. And that third receptor, the glucagon receptor, sends a signal straight to your heart that changes its rhythm. Not dramatically. Not dangerously, in most cases. But measurably. Consistently. And in a way that separates retatrutide from every other compound in its class.
This is not a theoretical concern. The phase 2 trial published in the New England Journal of Medicine documented heart rate increases across every dose group. Researchers measured it. Quantified it. Tracked it over 48 weeks. The numbers tell a specific story, one that most summaries of retatrutide gloss over entirely. A resting heart rate increase of 5 to 10 beats per minute might sound trivial on paper. For someone with a healthy cardiovascular system, it probably is. But for researchers with pre-existing conditions, those extra beats raise real questions about monitoring, dose adjustment, and long-term safety that deserve thorough answers.
This guide breaks down everything the clinical data reveals about retatrutide and heart rate. The mechanism behind the increase. How it compares to semaglutide and tirzepatide. When it peaks. When it fades. What the cardiovascular safety data actually shows. And what SeekPeptides members should know before starting or adjusting a retatrutide protocol.
Why retatrutide affects heart rate differently than other GLP-1 compounds
Every GLP-1 receptor agonist nudges heart rate upward to some degree. That is a known class effect. Semaglutide typically increases resting heart rate by 2 to 3 beats per minute. Tirzepatide raises it by roughly 2 to 4 beats per minute. These are modest changes that most people never notice without a heart rate monitor.
Retatrutide is different.
The average increase ranges from 5 to 10 beats per minute, with the highest dose groups seeing the largest changes. That is roughly double what semaglutide produces. And the reason comes down to one receptor that neither semaglutide nor tirzepatide activates: the glucagon receptor.
Retatrutide is a triple agonist. It binds GLP-1 receptors for appetite suppression and glucose control, GIP receptors for enhanced insulin sensitivity and fat metabolism, and glucagon receptors for direct energy expenditure and thermogenesis. This triple mechanism is what makes retatrutide potentially more effective for weight loss than dual or single agonists. But that glucagon receptor activation comes with a cardiovascular signature that researchers need to understand.
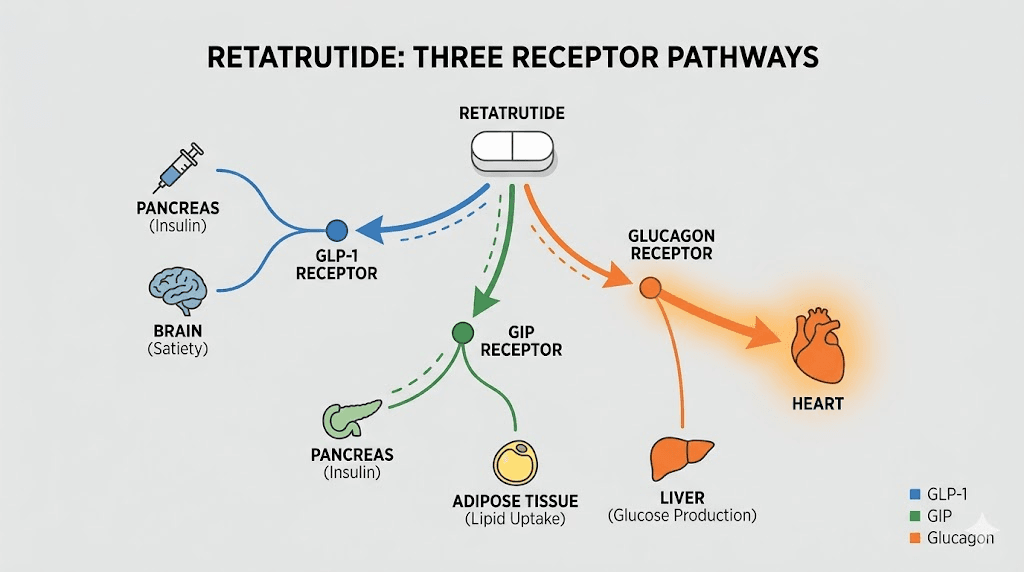
Glucagon has been known to increase heart rate for decades. When the glucagon receptor activates on cardiac tissue, it triggers a cascade involving cyclic adenosine monophosphate, or cAMP. This messenger molecule increases both the force of heart contractions (positive inotropic effect) and the rate of contractions (positive chronotropic effect). The heart beats harder. It beats faster. This is not a bug in retatrutide. It is a predictable pharmacological consequence of activating a receptor that was always going to do this.
Research published in Naunyn-Schmiedeberg Archives of Pharmacology confirmed this in isolated human atrial tissue. Retatrutide produced concentration-dependent increases in contractile force starting at 10 nanomolar concentrations and reaching plateau at 100 nanomolar.
The key finding was that plasma concentrations in patients taking 8 mg of retatrutide (approximately 182 nanomolar) exceeded the effective concentrations observed in the laboratory, suggesting these cardiac effects are clinically relevant at therapeutic doses.
The cAMP-PKA signaling pathway
Understanding why retatrutide affects the heart requires a brief look at the molecular chain of events. When retatrutide binds the glucagon receptor on cardiac cells, it activates adenylyl cyclase. This enzyme converts ATP into cAMP. Rising cAMP levels then activate protein kinase A (PKA), which phosphorylates calcium-handling proteins in the heart muscle.
The result is threefold. Calcium release from the sarcoplasmic reticulum increases, making each contraction stronger. L-type calcium channels open more readily, allowing more calcium to flow into the cell. And the rate of tension relaxation increases, meaning the heart can beat faster because each individual contraction cycle completes more quickly. Researchers at the University of Halle demonstrated that H89, a PKA inhibitor, could block retatrutide chronotropic effects but not those of isoprenaline (a beta-adrenergic agonist), confirming that retatrutide works through a distinct PKA-dependent mechanism rather than through the sympathetic nervous system.
This matters for a practical reason. Beta blockers, which are commonly prescribed for elevated heart rate, work by blocking the sympathetic nervous system. They may be less effective at controlling retatrutide-induced heart rate increases because retatrutide does not work through that pathway. Dose adjustment remains the primary tool for managing cardiovascular effects.
Why GLP-1 and GIP receptors are not the main drivers
Mouse atrial preparation studies provided an important clarification. When researchers tested pure GLP-1 receptor agonists, including semaglutide, liraglutide, and exenatide, none of them increased the beating rate of isolated heart tissue. This was surprising given that GLP-1 agonists do increase heart rate slightly in clinical settings. The explanation is that GLP-1 likely affects heart rate indirectly, through autonomic nervous system modulation rather than direct cardiac effects.
When researchers applied adomeglivant, a selective glucagon receptor antagonist, it blocked the chronotropic effects of retatrutide. This confirmed the glucagon receptor as the primary driver of the direct cardiac effects. The dose of retatrutide matters enormously here because higher doses mean more glucagon receptor activation and proportionally greater heart rate effects.
The clinical data: exactly how much does retatrutide increase heart rate
The most authoritative data comes from the phase 2 randomized controlled trial published in the New England Journal of Medicine (Jastreboff et al.). This trial enrolled 338 adults with obesity and tracked them for 48 weeks across multiple dose groups. Heart rate was measured at regular intervals throughout.
Here is what the numbers show.
Heart rate increases were dose-dependent and followed a consistent pattern across all treatment groups. The lowest doses produced the smallest increases. The highest doses produced the largest. This is exactly what you would expect from a drug whose cardiac effects are driven by receptor activation, more drug means more receptor binding means more cAMP means faster heart rate.
Heart rate changes by dose group
At the 12 mg dose (the highest tested), the average heart rate increase peaked at approximately 6.7 beats per minute at week 24. This was the highest average increase observed across any dose group at any time point. At lower doses, the increases were proportionally smaller, generally ranging from 2 to 5 beats per minute depending on the specific dose and titration schedule.
The 4 mg dose group, which many researchers consider the starting therapeutic range for retatrutide protocols, showed more modest heart rate elevations in the 2 to 4 beat per minute range. This is comparable to what tirzepatide produces at therapeutic doses and may represent an acceptable cardiovascular profile for most healthy individuals.
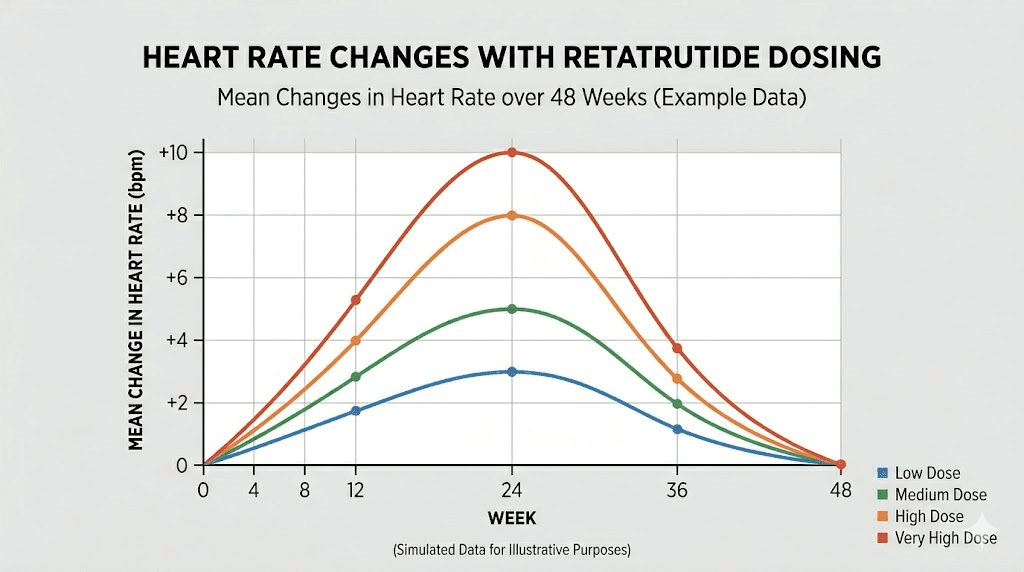
The week 24 peak and subsequent decline
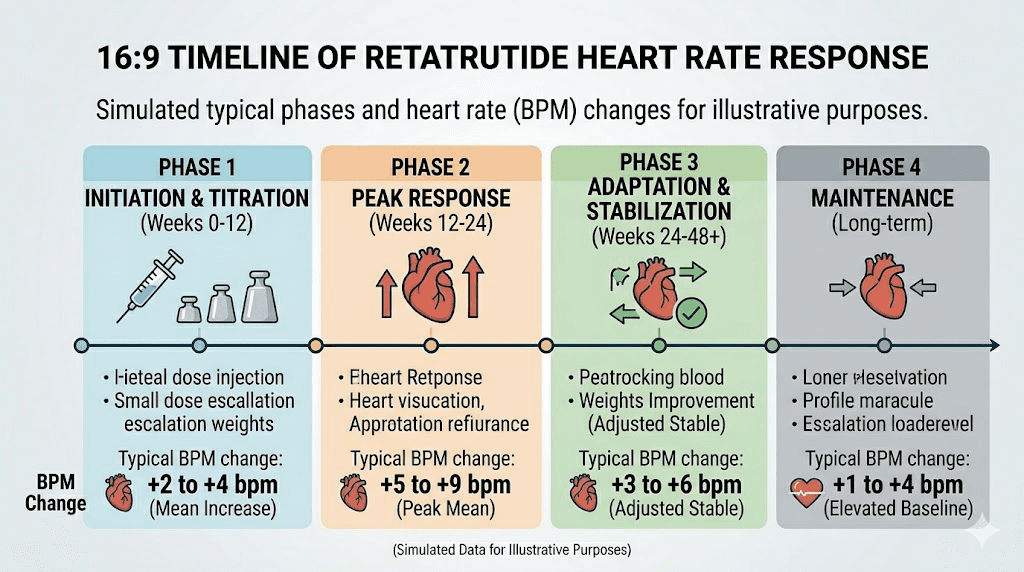
One of the most important patterns in the data is the temporal profile. Heart rate did not simply go up and stay up. It rose gradually during the dose-escalation phase, peaked around week 24, and then declined at weeks 36 and 48 even though participants continued taking the same dose.
This pattern suggests physiological adaptation. The body appears to recalibrate its cardiovascular response over time, potentially through downregulation of glucagon receptor sensitivity in cardiac tissue or compensatory autonomic adjustments. For researchers planning long-term retatrutide protocols, this is encouraging. The peak heart rate effect is temporary, not permanent.
The decline after week 24 does not mean heart rate returns completely to baseline while on treatment. Most participants maintained a small residual elevation above their pre-treatment heart rate throughout the study. But the acute peak, the period of maximum cardiovascular change, appears limited to roughly the first six months of treatment.
Individual variation matters
Averages obscure individual responses. Some participants in the clinical trial experienced heart rate increases well above 10 beats per minute, while others showed minimal change. Factors that likely influence individual response include baseline heart rate, cardiovascular fitness level, autonomic nervous system tone, body composition, and potentially genetic variations in glucagon receptor density or sensitivity.
Researchers who are physically active and have low resting heart rates (below 60 beats per minute) may notice the increase more because they are more attuned to their cardiovascular baseline. Someone with a resting rate of 55 who jumps to 65 will likely notice that shift. Someone with a resting rate of 75 who moves to 80 may not notice at all, even though both experienced a similar absolute change. If you are tracking your response to retatrutide, measuring resting heart rate first thing in the morning before any activity provides the most consistent comparison point.
Retatrutide vs semaglutide vs tirzepatide: a heart rate comparison
Comparing cardiovascular effects across these three compounds helps put retatrutide heart rate data into context. Each drug has a different receptor profile, which translates into different cardiovascular signatures.
Semaglutide: the single agonist baseline
Semaglutide activates only the GLP-1 receptor. In clinical trials, it produces an average heart rate increase of 2 to 3 beats per minute, with some individuals reaching 10 beats per minute or more. The SUSTAIN and STEP trial programs documented these changes across thousands of participants. Cardiovascular side effects with semaglutide are generally mild, and the SELECT trial demonstrated a 20% reduction in major adverse cardiovascular events (MACE) in people with established heart disease.
The mechanism of heart rate increase with semaglutide is primarily indirect, mediated through autonomic nervous system effects rather than direct cardiac stimulation. This explains why the magnitude is smaller than retatrutide, which adds direct glucagon receptor-mediated cardiac effects on top of the indirect GLP-1 effects.
Tirzepatide: the dual agonist middle ground
Tirzepatide activates both GLP-1 and GIP receptors. The SURPASS clinical trial program showed mean heart rate increases of approximately 2 to 4 beats per minute. Side effects with tirzepatide are primarily gastrointestinal, with cardiovascular effects being relatively modest. Ambulatory blood pressure monitoring showed that tirzepatide actually reduces both systolic and diastolic blood pressure, partially offsetting any heart rate increase from a cardiovascular risk perspective.
The addition of GIP receptor agonism in tirzepatide does not appear to significantly amplify heart rate effects compared to GLP-1-only drugs like semaglutide. This supports the conclusion that it is specifically the glucagon receptor, present in retatrutide but not in tirzepatide, that drives the larger heart rate signal.
The comparison table
Parameter | Semaglutide | Tirzepatide | Retatrutide |
|---|---|---|---|
Receptors activated | GLP-1 only | GLP-1 + GIP | GLP-1 + GIP + Glucagon |
Average HR increase | 2-3 BPM | 2-4 BPM | 5-10 BPM |
Peak HR effect timing | Early weeks | Early weeks | Week 24 |
HR effect declines over time | Persists | Persists | Yes, after week 24 |
Direct cardiac effect | Minimal | Minimal | Yes (glucagon receptor) |
Blood pressure effect | Mild decrease | Moderate decrease | Significant decrease (up to 14 mmHg systolic) |
Max weight loss (trials) | ~15-17% | ~20-25% | ~24-26% |
The table reveals an important tradeoff. Retatrutide produces the largest heart rate increase but also the most significant blood pressure reduction and the greatest weight loss. For many researchers, the net cardiovascular effect may actually be favorable despite the higher heart rate, because blood pressure reduction and weight loss both independently reduce cardiovascular risk. Understanding the specific dose you are using helps calibrate expectations for both benefits and side effects.
When heart rate changes become concerning
Not every heart rate increase requires intervention. The human heart is remarkably adaptable. It speeds up when you stand, when you exercise, when you drink coffee, when you feel anxious. A 5 to 10 beat per minute resting increase from retatrutide falls within the range of normal physiological variation for most healthy people.
But some situations warrant attention.
Red flags that require medical evaluation
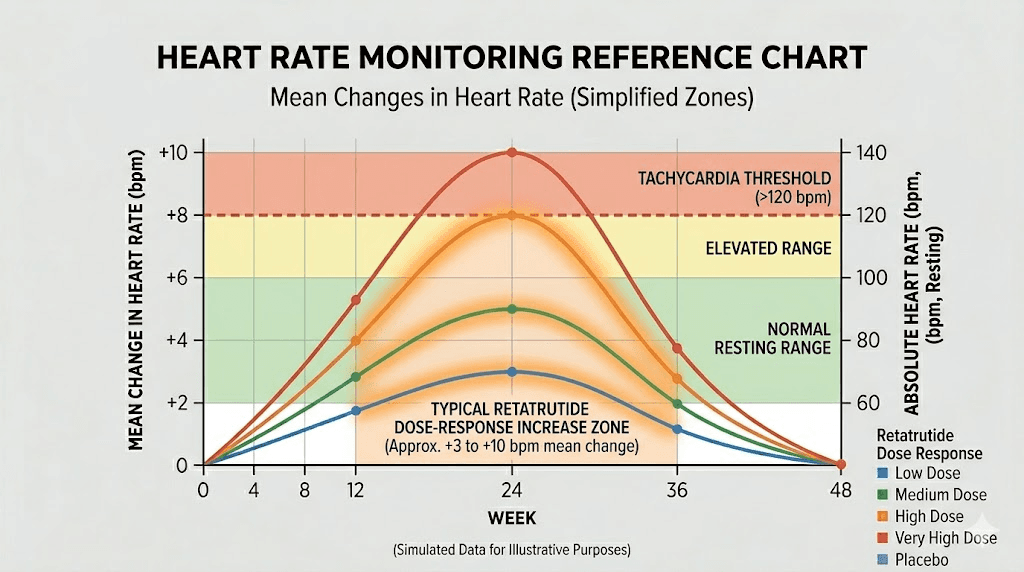
A sustained resting heart rate above 100 beats per minute (tachycardia) at rest is never normal and should prompt medical evaluation regardless of the cause. If retatrutide pushes someone from a resting rate of 92 to 102, that crosses into a range where cardiac workload becomes a genuine concern, especially over months of treatment.
Palpitations, the sensation of feeling your heart beat irregularly or unusually forcefully, deserve attention even if your measured heart rate remains in a normal range. Some retatrutide users report awareness of their heartbeat, particularly during the dose-escalation phase. Most of the time this reflects the positive inotropic effect (stronger contractions) rather than any dangerous rhythm abnormality. But palpitations accompanied by dizziness, chest pain, shortness of breath, or fainting require immediate medical evaluation to rule out arrhythmias.
An increase in the incidence of cardiac arrhythmias, mainly supraventricular, was noted in some trial arms. The phase 2 data showed arrhythmias in 4 to 14% of retatrutide groups compared to 2 to 3% in the placebo group. These were generally mild and not classified as serious adverse events, but they establish that retatrutide does have a measurable effect on cardiac rhythm beyond simple rate increase.
Pre-existing conditions that increase risk
Researchers with the following conditions should discuss retatrutide heart rate effects with their physician before starting a protocol:
History of atrial fibrillation or other arrhythmias - the increased contractile force and rate from glucagon receptor activation could potentially trigger or worsen existing rhythm disorders
Heart failure - while increased contractility might seem beneficial, the increased cardiac workload from higher heart rate can worsen heart failure in some cases
Hyperthyroidism - thyroid hormone excess already increases heart rate, and adding retatrutide could produce additive effects
Anxiety disorders or panic disorder - heightened awareness of cardiac sensations can trigger anxiety episodes in susceptible individuals
Use of stimulant medications - amphetamines, methylphenidate, and similar drugs increase heart rate through sympathetic activation, and combining them with retatrutide glucagon receptor effects could produce larger-than-expected increases
For most healthy adults without these conditions, a gradual dose escalation starting at lower doses allows the cardiovascular system to adapt and minimizes the risk of abrupt, uncomfortable heart rate changes. The retatrutide dosage calculator can help determine appropriate starting points based on individual parameters.
Managing heart rate increases on retatrutide
The clinical trial data suggest several evidence-based approaches to managing retatrutide heart rate effects. These are not theoretical recommendations. They are derived from patterns observed across hundreds of trial participants and from the pharmacological profile of the drug.
Slow dose escalation is the primary strategy
The most effective way to minimize heart rate effects is slow, gradual dose escalation. Researchers who jump to high doses quickly experience more pronounced cardiovascular effects because the glucagon receptor gets activated intensely before the body can adapt. A slower titration schedule, spending more time at each dose level before increasing, gives the cardiovascular system time to recalibrate.
Clinical trial protocols typically used 4-week intervals between dose increases. Some researchers choose even longer intervals, spending 6 to 8 weeks at each dose level, particularly if they notice heart rate increases above 10 beats per minute from baseline. The recommended approach to retatrutide dosing prioritizes tolerability over speed. There is no benefit to reaching target dose faster if cardiovascular side effects become limiting.
The microdosing approach to retatrutide has gained attention precisely because it minimizes side effects including heart rate changes. Starting at very low doses and increasing in small increments allows the most gradual cardiovascular adaptation possible.
Timing and lifestyle factors
Heart rate naturally fluctuates throughout the day, and certain factors can amplify the effects of retatrutide on cardiac rate:
Caffeine is an adenosine receptor antagonist that independently increases heart rate. Combining high caffeine intake with retatrutide can produce additive effects. Researchers who notice uncomfortable heart rate increases should consider reducing caffeine intake, particularly in the first few months of treatment when cardiovascular effects are most pronounced. Switching from coffee to tea, reducing intake by half, or avoiding caffeine in the afternoon are practical modifications.
Dehydration increases heart rate because the heart must beat faster to maintain blood pressure with reduced blood volume. Gastrointestinal side effects of retatrutide, including nausea and diarrhea, can contribute to dehydration. Adequate fluid intake becomes even more important when cardiovascular effects are a concern. A reasonable target is 2 to 3 liters of water daily, adjusted upward for physical activity and warm environments.
Sleep quality affects autonomic nervous system balance. Poor sleep shifts the autonomic balance toward sympathetic dominance, which increases resting heart rate independently. Researchers experiencing fatigue on retatrutide may also have disrupted sleep patterns that compound cardiovascular effects. Prioritizing 7 to 9 hours of quality sleep supports cardiovascular adaptation.
Exercise is a powerful tool for managing resting heart rate. Regular aerobic exercise improves vagal tone and lowers resting heart rate over time. Researchers who maintain a consistent exercise routine while on retatrutide often see their resting heart rate stabilize or even decrease below baseline over months, despite the pharmacological effect pushing it upward. The role of exercise in retatrutide protocols extends well beyond weight loss, it is a genuine cardiovascular management strategy.
Monitoring protocol
A structured approach to heart rate monitoring provides early warning if effects are trending toward concerning territory:
Baseline measurement: Before starting retatrutide, measure resting heart rate for 5 consecutive mornings immediately upon waking (before getting out of bed, before coffee, before checking your phone). Average these five readings. This is your true resting baseline. Many people have never measured their resting heart rate accurately and are surprised by their starting point.
Weekly monitoring during escalation: During dose increases, measure resting heart rate each morning using the same protocol. Track the numbers in a simple log. A consistent upward trend of more than 15 beats per minute above baseline, or any reading consistently above 95 at rest, warrants a pause in dose escalation and consultation with a physician.
Monthly monitoring at stable dose: Once you reach your target dose and heart rate has stabilized (typically after 2 to 3 weeks at the same dose), weekly monitoring can shift to monthly spot checks. Continue the same morning measurement protocol.
Wearable devices like fitness trackers and smartwatches can supplement manual measurements, but morning resting measurements taken before activity remain the gold standard. Continuous wearable data can show trends but is easily confounded by movement, stress, and measurement artifact. The home monitoring tools guide covers device options in more detail.
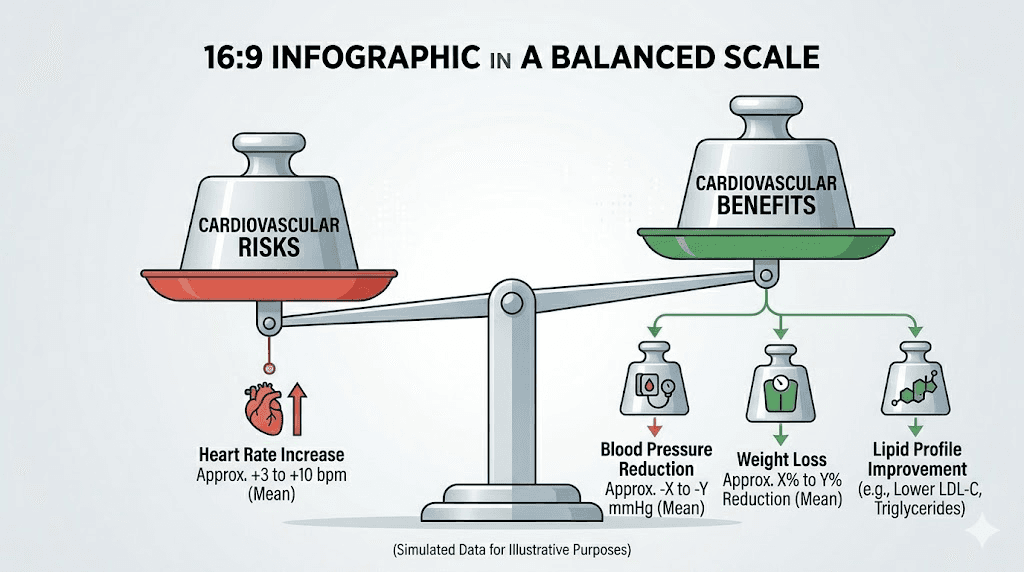
Cardiovascular benefits that offset the heart rate concern
Focusing only on heart rate increase gives an incomplete picture. The cardiovascular effects of retatrutide are multidimensional, and several of them are strongly protective.
Blood pressure reduction
TRIUMPH-4 data from December showed retatrutide reducing systolic blood pressure by 14 mmHg at the 12 mg dose. That is a clinically significant reduction, comparable to what you would expect from a dedicated blood pressure medication. Even at lower doses, meaningful blood pressure reductions were observed.
This matters enormously. Elevated blood pressure is one of the strongest predictors of heart attack, stroke, and cardiovascular death. A sustained 14 mmHg reduction in systolic pressure translates to roughly a 40 to 50% reduction in stroke risk and a 20 to 30% reduction in coronary events over the long term. These benefits vastly outweigh the theoretical risk of a modest heart rate increase in most people.
The mechanism behind blood pressure reduction is multifactorial. Weight loss itself reduces blood pressure. GLP-1 receptor activation promotes vasodilation. Improved insulin sensitivity reduces the arterial inflammation and stiffness that drives hypertension. All three of retatrutide receptor pathways contribute to blood pressure improvement through different mechanisms.
Lipid improvements
Retatrutide produces substantial improvements in lipid profiles. Trial data showed significant drops in non-HDL cholesterol, triglycerides, and high-sensitivity C-reactive protein (a marker of cardiovascular inflammation). The glucagon receptor activation that drives heart rate increase also drives hepatic fat oxidation, which is a primary mechanism behind the lipid improvements.
This creates an interesting pharmacological paradox. The same receptor activation that increases heart rate also drives some of the most potent cardiovascular benefits. You cannot have one without the other with retatrutide. The question is whether the net effect is positive or negative, and the aggregate clinical data strongly suggests it is positive for most people.
Weight loss as cardiovascular medicine
Retatrutide produces the most dramatic weight loss of any peptide studied to date, with mean reductions of 24 to 26% body weight at the highest doses. Weight loss of this magnitude is transformative for cardiovascular risk. Every 5 kg of weight loss independently reduces systolic blood pressure by approximately 3 to 5 mmHg, reduces triglycerides, improves HDL cholesterol, reduces insulin resistance, decreases systemic inflammation, and reduces mechanical strain on the heart.
For someone with obesity, the cardiovascular risk reduction from losing 25% of their body weight almost certainly exceeds the risk from a 5 to 10 beat per minute resting heart rate increase. This is not to dismiss the heart rate effect, but rather to contextualize it within the broader cardiovascular picture. Researchers evaluating whether retatrutide is better than tirzepatide for their goals should weigh the total cardiovascular package, not just one parameter.
The TRIUMPH trials and long-term cardiovascular outlook
Understanding the long-term cardiovascular safety of retatrutide requires looking at the broader clinical trial program being conducted by Eli Lilly. The TRIUMPH program includes multiple phase 3 trials, and one trial, TRIUMPH-Outcomes, is specifically designed to answer the question of whether retatrutide reduces major adverse cardiovascular events.
What we know from phase 2
The 48-week phase 2 trial provided reassuring safety signals. There was no increase in serious cardiovascular events during 68 weeks of treatment (including follow-up). No participant experienced a heart attack, stroke, or cardiovascular death attributed to retatrutide. The cardiac arrhythmias that were observed were predominantly mild and supraventricular, not the dangerous ventricular arrhythmias that pose immediate life-threatening risk.
However, phase 2 trials are not powered to detect rare cardiovascular events. With only 338 participants, a cardiovascular event rate of even 1 to 2% could easily be missed statistically. This is why the larger phase 3 and cardiovascular outcomes trials are essential.
What TRIUMPH-Outcomes will tell us
The TRIUMPH-Outcomes trial is designed to determine whether retatrutide significantly lowers the incidence of major adverse cardiovascular events, defined as cardiovascular death, non-fatal heart attack, and non-fatal stroke. This trial enrolls thousands of participants with established cardiovascular disease or high cardiovascular risk and follows them for several years. Results are expected in the 2027 to 2028 timeframe.
If the results mirror what semaglutide achieved in the SELECT trial (a 20% reduction in MACE), retatrutide heart rate effects will likely be viewed as a minor footnote in an overwhelmingly positive cardiovascular profile. If the results are neutral or negative, the heart rate and arrhythmia signals from phase 2 will receive much more scrutiny. For now, researchers are making decisions based on incomplete but generally encouraging data.
The GLP-1 class cardiovascular track record
Context from the broader GLP-1 receptor agonist class is relevant. Multiple GLP-1 drugs have demonstrated cardiovascular benefits in large outcomes trials. Semaglutide reduced MACE by 20% in SELECT. Liraglutide reduced MACE by 13% in LEADER. Dulaglutide reduced MACE by 12% in REWIND. Every GLP-1 agonist that has been tested in a cardiovascular outcomes trial has shown either benefit or neutrality, never harm.
Retatrutide includes GLP-1 receptor activation as one of its three mechanisms. There is reasonable scientific basis to expect that the GLP-1 component will confer similar cardiovascular protection. The question is whether the added glucagon receptor effects, including the heart rate increase, will enhance, diminish, or have no effect on that protection. The comparison between Ozempic and retatrutide often comes down to this exact question of risk-benefit tradeoff.
Heart rate effects during different phases of retatrutide use
The cardiovascular response to retatrutide is not static. It evolves through distinct phases that correspond to the dosing trajectory and physiological adaptation.
Phase 1: initiation and early escalation (weeks 1 to 8)
During the first weeks, heart rate changes are generally minimal at starting doses. Most protocols begin at 1 to 2 mg, which produces modest glucagon receptor activation. Initial effects of retatrutide during this phase are primarily gastrointestinal, with appetite suppression developing before cardiovascular effects become noticeable.
This is the ideal time to establish your baseline measurements and monitoring routine. Your heart rate data from weeks 1 to 4 provides the reference point for tracking changes as doses increase.
Phase 2: active escalation (weeks 8 to 24)
As doses increase toward the therapeutic range of 8 to 12 mg, heart rate effects become more apparent. This is the phase where most people who are going to notice cardiovascular changes will notice them. The heart rate increase is progressive, typically adding 1 to 2 beats per minute with each dose escalation step.
Some researchers report increased awareness of their heartbeat during this phase, particularly during the first few days after a dose increase. This is usually the positive inotropic effect (stronger contractions) rather than an arrhythmia, but it can be unsettling if you are not expecting it. Knowing that this is a documented, transient effect helps manage anxiety about the sensation.
Phase 3: peak effect (weeks 20 to 28)
Heart rate effects peak around week 24 in the clinical data. This is typically when participants have reached their target dose and the full pharmacological effect has developed. If you are going to experience a problem with heart rate on retatrutide, it will most likely manifest during this window.
Researchers who sail through the week 24 period without concerning heart rate changes can generally be reassured that their cardiovascular response is within acceptable parameters for continued use.
Phase 4: adaptation and stabilization (weeks 28 and beyond)
The decline in heart rate effects after week 24 reflects genuine physiological adaptation. The body adjusts glucagon receptor sensitivity, autonomic balance shifts, and the cardiovascular system reaches a new equilibrium. Heart rate typically settles to a level modestly above baseline but well below the week 24 peak.
Long-term users of retatrutide who also exercise regularly and have lost significant weight may find their resting heart rate actually lower than pre-treatment levels, because the cardiovascular benefits of weight loss and improved fitness outweigh the modest pharmacological heart rate increase.
Special populations and extra considerations
Certain groups of researchers may need to approach retatrutide heart rate effects with additional caution or modified monitoring.
Older adults
Age-related changes in cardiovascular function can amplify the impact of retatrutide heart rate effects. Older adults tend to have reduced cardiovascular reserve, stiffer arteries, and less robust autonomic adaptation. A 5 to 10 beat per minute increase that a 35-year-old barely notices might cause fatigue, exercise intolerance, or shortness of breath in a 65-year-old.
The flip side is that older adults often have the most to gain from retatrutide cardiovascular benefits, particularly blood pressure reduction and weight loss. An individualized approach with slower dose escalation and more frequent monitoring is appropriate for this population.
Athletes and highly active individuals
Endurance athletes and highly trained individuals typically have low resting heart rates (40 to 55 beats per minute) due to high vagal tone. Retatrutide heart rate effects are additive to this low baseline, meaning an athlete at 50 BPM might go to 55 to 60 BPM. This is still well within normal range and unlikely to affect performance or health.
However, athletes are often more attuned to subtle changes in cardiovascular function. They may notice the heart rate increase during training sessions, particularly during recovery intervals when heart rate should normally drop quickly. Heart rate variability (HRV), a metric many athletes track, may also be affected by retatrutide. Reduced HRV during the early treatment phase is expected and typically normalizes during the adaptation phase.
For those combining retatrutide with resistance training for body recomposition, heart rate monitoring during heavy compound lifts provides useful safety data. Elevated resting heart rate combined with the cardiovascular demand of heavy squats or deadlifts increases cardiac workload. Taking slightly longer rest periods between sets during the early weeks of retatrutide treatment is a prudent modification.
People on cardiovascular medications
Researchers already taking medications that affect heart rate or rhythm should consult their physician before starting retatrutide. Specific considerations include:
Beta blockers (metoprolol, atenolol, propranolol) lower heart rate by blocking sympathetic stimulation. Since retatrutide heart rate effects are mediated through the glucagon receptor cAMP-PKA pathway rather than the sympathetic system, beta blockers may not fully counteract retatrutide-induced increases. The result could be unpredictable, with beta blockers controlling the sympathetic component while the glucagon-mediated component persists.
Calcium channel blockers (diltiazem, verapamil) that slow heart rate work through a different mechanism than retatrutide.
Their effects are more likely to counteract retatrutide heart rate increases because nifedipine (an L-type calcium channel blocker) was shown to partially reduce retatrutide cardiac effects in laboratory studies.
Stimulant medications (ADHD medications, certain weight loss drugs) add to heart rate through sympathetic activation. Combining stimulants with retatrutide means two independent mechanisms are both pushing heart rate upward, potentially producing larger-than-expected increases. The question of whether to combine phentermine with GLP-1 compounds is particularly relevant here.
Transitioning from other compounds
Researchers switching from tirzepatide to retatrutide should be aware that they may experience a noticeable increase in heart rate effects due to the addition of glucagon receptor activation. This is expected and does not necessarily indicate a problem, but monitoring during the transition is important.
The transition period from tirzepatide to retatrutide involves pharmacological overlap as the previous compound washes out and the new one reaches steady state. Heart rate effects during this window may not be representative of the eventual stable-state response. Allow at least 4 to 6 weeks at a stable retatrutide dose before drawing conclusions about your cardiovascular response.
The inotropic effect: retatrutide makes your heart beat harder, not just faster
Most discussions of retatrutide heart rate focus on the chronotropic effect, the increase in beats per minute. But the research reveals an equally important phenomenon: the positive inotropic effect, meaning retatrutide increases the force of each heartbeat.
The study in isolated human atrial preparations demonstrated that retatrutide increases the rate of tension development and the peak tension of cardiac contractions. In simpler terms, each heartbeat pushes more blood with more force. This is the same effect you get from exercise, from adrenaline, and from drugs like digoxin that are used to treat heart failure.
What this means for researchers
The positive inotropic effect has several practical implications:
Awareness of heartbeat. A stronger contraction is easier to feel, especially at rest when there are fewer competing sensations. Many researchers who report "palpitations" on retatrutide are actually experiencing normal rhythm with increased contractile force. The heart is not beating abnormally, it is beating more forcefully. This is disconcerting but generally not dangerous.
Blood pressure effects. Increased contractile force should theoretically increase blood pressure, but retatrutide actually decreases blood pressure. This is because the vasodilatory effects of GLP-1 receptor activation and the blood volume reduction from weight loss more than compensate for the increased cardiac output. The net result is lower blood pressure despite a harder-working heart.
Exercise performance. Some researchers report improved exercise tolerance on retatrutide, possibly related to increased cardiac output. A heart that beats faster AND harder delivers more oxygen to working muscles. While this is not a reason to take retatrutide, it is a potential secondary benefit that athletically inclined researchers may notice.
The question of whether the increased cardiac workload (both rate and force) could cause long-term cardiac remodeling remains open. In heart failure, chronic increased workload leads to pathological cardiac hypertrophy. But in exercise, similar increases lead to beneficial athletic cardiac remodeling. The difference is context, intensity, and duration. The TRIUMPH-Outcomes trial will eventually provide data on long-term cardiac structural effects. For now, the absence of cardiac damage signals in the phase 2 trial is reassuring but not definitive.
Practical protocols for heart rate management
Based on the clinical data and pharmacological understanding of retatrutide, here are specific protocols for managing heart rate effects at different stages of use.
Protocol 1: standard approach for healthy researchers
Who this is for: Adults without cardiovascular disease, normal baseline heart rate, no cardiac medications.
Pre-treatment:
Measure morning resting heart rate for 5 days
Calculate average baseline
Record any existing caffeine or stimulant use
During treatment:
Measure resting heart rate 3 times weekly during dose escalation
Continue at each dose for minimum 4 weeks before increasing
If resting HR exceeds baseline by more than 15 BPM, hold dose for an additional 2 weeks
If resting HR exceeds 100 BPM at rest on two or more measurements, consult physician
Expected timeline:
Weeks 1-8: minimal heart rate change at starting doses
Weeks 8-24: gradual increase of 5-10 BPM at therapeutic doses
Weeks 24+: stabilization and gradual decline toward baseline
This is the approach most researchers follow successfully, and it aligns with the standard retatrutide dosing protocol used in clinical trials.
Protocol 2: conservative approach for higher-risk researchers
Who this is for: Older adults, those with borderline high resting heart rate, anyone on cardiovascular medications, history of anxiety about cardiac symptoms.
Pre-treatment:
Measure morning resting heart rate for 7 days
Consider baseline ECG if not done recently
Review all medications for cardiac interactions
Discuss with physician specifically about the glucagon receptor heart rate effect
During treatment:
Measure resting heart rate daily during dose escalation
Continue at each dose for minimum 6 to 8 weeks before increasing
If resting HR exceeds baseline by more than 10 BPM, hold dose
If resting HR exceeds 90 BPM at rest consistently, consult physician before any further increases
Consider microdosing approach for ultra-gradual escalation
Expected timeline:
Weeks 1-12: very gradual heart rate change at low doses
Weeks 12-30: moderate increase as dose slowly climbs
Weeks 30+: stabilization (may occur later than standard protocol due to slower escalation)
Protocol 3: monitoring-enhanced approach for data-driven researchers
Who this is for: Researchers who want comprehensive data, athletes tracking performance metrics, anyone who finds data reassuring.
Equipment needed:
Chest strap heart rate monitor (more accurate than wrist-based devices)
Heart rate variability (HRV) app
Blood pressure cuff
Spreadsheet or tracking app
Daily measurements:
Morning resting heart rate (before getting up)
Morning HRV (using chest strap, 5-minute measurement)
Blood pressure (morning, before caffeine)
What to track:
7-day rolling average of resting HR (smooths daily variation)
HRV trend (declining HRV indicates autonomic stress)
Blood pressure trend (should improve or stay stable)
Subjective cardiac awareness (scale of 0-5)
This level of monitoring is not necessary for most people, but for those who want complete data, it provides early warning of adverse trends and concrete evidence of cardiovascular adaptation over time. SeekPeptides members who track these metrics systematically develop the most complete understanding of how their individual cardiovascular system responds to treatment.
Common questions and misconceptions about retatrutide and heart rate
Several misunderstandings circulate about retatrutide cardiovascular effects. Addressing them directly prevents unnecessary anxiety and helps researchers make informed decisions.
Misconception: retatrutide causes dangerous heart problems
The clinical data does not support this. No heart attacks, strokes, or cardiovascular deaths have been attributed to retatrutide in published trial data. The heart rate increase is a pharmacological effect, not a pathological one. It is comparable in magnitude to what happens when you drink a large cup of coffee or walk up a flight of stairs. The key word in the safety data is "no increase in serious cardiovascular events."
That said, the absence of evidence is not evidence of absence. The phase 2 trial was too small to detect rare events. The ongoing phase 3 program and TRIUMPH-Outcomes trial will provide definitive safety data. Until those results are available, a reasonable approach is cautious optimism informed by monitoring.
Misconception: the heart rate increase means retatrutide is harder on your heart than semaglutide
This assumes that heart rate is the only relevant cardiac parameter. It is not. Retatrutide produces greater blood pressure reduction than semaglutide. It produces greater weight loss. It produces greater improvements in lipid profile and inflammatory markers. The net cardiovascular effect, considering all parameters, may actually be superior to semaglutide despite the higher heart rate. The full comparison of these three compounds should weigh all cardiovascular parameters together.
Misconception: you need a heart monitor to take retatrutide
While monitoring is recommended and provides useful data, it is not strictly required for healthy individuals at standard doses. The clinical trial participants did not use continuous heart rate monitoring at home. Regular vital sign checks at follow-up appointments were sufficient to identify concerning trends. That said, the wide availability of inexpensive fitness trackers makes monitoring easy enough that there is little reason not to do it.
Misconception: heart rate effects mean you should use a lower dose
Not necessarily. Heart rate effects should be evaluated in the context of overall tolerability and treatment goals. A researcher who experiences a 7 BPM resting heart rate increase but is losing weight steadily, feeling well, and has normal blood pressure is not necessarily best served by reducing their dose. The decision to adjust dose based on heart rate should involve the overall clinical picture, not just one number in isolation.
However, if heart rate effects are accompanied by palpitations, chest discomfort, or significant exercise intolerance, dose reduction is appropriate. The relationship between dose and heart rate is clear enough that reducing the dose will reduce the cardiovascular effect proportionally.
How retatrutide heart rate effects compare to everyday activities
Context helps calibrate the significance of a 5 to 10 BPM heart rate increase. Here is how the retatrutide effect compares to other common heart rate influences that most people experience daily without concern:
Activity or factor | Typical heart rate increase |
|---|---|
Standing up from sitting | 10-20 BPM |
Walking at moderate pace | 40-60 BPM above resting |
One cup of coffee (200mg caffeine) | 3-15 BPM |
Mild stress or anxiety | 5-20 BPM |
Eating a large meal | 5-15 BPM |
Poor night of sleep | 5-10 BPM next morning |
Dehydration (mild) | 5-15 BPM |
Retatrutide (therapeutic dose) | 5-10 BPM |
This table demonstrates that the retatrutide heart rate effect falls squarely within the range of everyday physiological variations that healthy hearts handle without difficulty. The difference is that the retatrutide effect is sustained rather than transient. Your heart rate drops back to resting when you sit down or when your coffee wears off, but the retatrutide effect persists (though it does diminish after week 24 as discussed earlier).
For most healthy individuals, a sustained 5 to 10 BPM increase is the cardiovascular equivalent of mild background stress, significant enough to measure, but not significant enough to cause harm in a healthy heart. The timeline for retatrutide effects including cardiovascular adaptation makes this a temporary concern during the treatment initiation period rather than a permanent cardiovascular burden.
What the research community is watching for next
Several ongoing investigations will significantly clarify the retatrutide-heart rate relationship over the coming years. Understanding what data is coming helps researchers make informed decisions about starting, continuing, or modifying their protocols.
TRIUMPH-Outcomes results
This is the most important upcoming dataset. If retatrutide reduces major cardiovascular events (like semaglutide did in SELECT), the heart rate increase will be viewed as a pharmacologically interesting but clinically irrelevant side effect. If the results are neutral, the conversation becomes more nuanced. Expected timeline for results is 2027 to 2028.
NCT06383390: dedicated cardiac effects study
A clinical trial specifically designed to study the cardiac effects of retatrutide is registered on ClinicalTrials.gov. This focused investigation should provide detailed electrocardiographic data, ambulatory monitoring results, and potentially echocardiographic measurements that will clarify whether the heart rate and contractility changes translate into any structural or functional cardiac concerns.
Real-world cardiovascular data
As retatrutide moves toward wider availability, real-world data from clinical practice will emerge. This data is valuable because it captures effects in broader populations than clinical trials, including people with comorbidities, those taking multiple medications, and those using doses or schedules that differ from trial protocols. Post-marketing surveillance will be particularly important for detecting rare cardiovascular events that trials were not powered to find.
For researchers currently using retatrutide, the best approach is informed engagement. Understand the mechanism. Monitor your response. Contextualize heart rate changes within the broader cardiovascular picture. And stay current with emerging data that will continue to refine our understanding of this compound unique cardiovascular profile.
Combining retatrutide with other compounds: cardiovascular considerations
Researchers who combine retatrutide with other peptides or supplements should consider cardiovascular interactions, particularly those that affect heart rate.
Retatrutide and cagrilintide
Combining cagrilintide with retatrutide is gaining interest for enhanced weight loss. Cagrilintide (an amylin analog) does not have significant direct cardiac effects, making it a relatively safe cardiovascular combination. However, the enhanced weight loss from the combination could lead to more pronounced fluid and electrolyte shifts that indirectly affect heart rate and blood pressure. Adequate hydration and electrolyte monitoring are important.
Retatrutide and tesamorelin
The combination of tesamorelin and retatrutide targets both visceral fat reduction and overall weight loss. Tesamorelin (a growth hormone releasing hormone analog) can cause mild fluid retention and transient increases in blood pressure in some users. When combined with retatrutide, researchers should monitor both heart rate and blood pressure more frequently during the initial adjustment period.
Retatrutide and stimulant-containing supplements
Pre-workout supplements, fat burners, and caffeine-containing thermogenic products all increase heart rate through sympathetic nervous system activation. Adding these on top of retatrutide glucagon-mediated heart rate increase can produce cumulative effects that exceed what either would cause alone. This does not mean they cannot be combined, but it means researchers should be aware of the additive potential and monitor accordingly.
Products containing synephrine, yohimbine, or high-dose caffeine (over 300 mg) warrant particular caution. These compounds can individually increase heart rate by 10 to 20 BPM. Adding retatrutide 5 to 10 BPM on top of that pushes the total increase into a range where healthy individuals might notice uncomfortable cardiovascular symptoms.
Retatrutide and other GLP-1 compounds
Some researchers ask about combining retatrutide with semaglutide or tirzepatide. There is no clinical data on this combination, and it is generally not recommended because the GLP-1 receptor activation from multiple compounds would overlap, potentially increasing gastrointestinal side effects without proportional benefit. The question of using semaglutide and tirzepatide together has similar concerns. From a cardiovascular perspective, stacking GLP-1 agonists would likely produce additive heart rate effects, which is another reason to avoid the combination.
The bottom line on retatrutide and heart rate
Retatrutide increases resting heart rate by an average of 5 to 10 beats per minute. This effect is dose-dependent, peaks around week 24, and diminishes thereafter even with continued treatment. It is driven primarily by glucagon receptor activation of the cAMP-PKA signaling pathway in cardiac tissue, which is a direct pharmacological effect rather than an indirect autonomic change.
For the vast majority of healthy researchers, this heart rate increase is clinically insignificant. It falls within the range of normal daily heart rate variation. It is accompanied by cardiovascular benefits, including significant blood pressure reduction, lipid improvements, and dramatic weight loss, that almost certainly produce a net positive cardiovascular effect.
For researchers with pre-existing cardiovascular conditions, cardiac medications, or other risk factors, the heart rate increase warrants closer monitoring and slower dose escalation. A conversation with a physician about the specific pharmacology of retatrutide cardiovascular effects is prudent before starting treatment.
The data we have is reassuring but not complete. No serious cardiovascular events have been attributed to retatrutide in published trials. But larger, longer studies are needed to provide definitive safety confirmation. The TRIUMPH-Outcomes trial will be the most important dataset for resolving remaining questions.
In the meantime, the evidence supports a straightforward approach: start low, escalate slowly, monitor regularly, and evaluate heart rate changes in the context of overall cardiovascular health rather than as an isolated number. For researchers managing their own protocols, understanding the mechanism behind the heart rate effect removes the anxiety of the unknown and replaces it with informed, measured decision-making.
SeekPeptides members access comprehensive retatrutide protocols that include cardiovascular monitoring guidance, dose escalation schedules tailored to individual risk profiles, and dosage calculators that account for tolerability factors beyond just weight. For researchers serious about optimizing their peptide protocols safely, SeekPeptides provides the most thorough evidence-based resource available, with protocols, community support, and expert guidance for navigating exactly these kinds of nuanced clinical questions.
Frequently asked questions
Does retatrutide increase heart rate in everyone?
Clinical trial data shows heart rate increases across most participants at therapeutic doses, but the magnitude varies significantly. Some people experience increases under 5 BPM while others see increases above 10 BPM. Factors including baseline heart rate, dose level, cardiovascular fitness, and individual receptor sensitivity all influence the response.
How long does the heart rate increase last on retatrutide?
Heart rate effects peak around week 24 of treatment and then decline at weeks 36 and 48 even with continued dosing. This suggests the cardiovascular system adapts over time. The effect is most noticeable during the active dose-escalation phase and the first few weeks at target dose. Most researchers report that cardiac awareness diminishes significantly after 6 months of stable dosing. See the full retatrutide timeline guide for more detail.
Is retatrutide heart rate increase dangerous?
For most healthy individuals, a 5 to 10 BPM increase in resting heart rate is not clinically dangerous. It is comparable to the effect of a cup of coffee or mild stress. No serious cardiovascular events have been attributed to retatrutide in published trial data. However, individuals with pre-existing arrhythmias, heart failure, or other cardiovascular conditions should consult their physician before starting retatrutide.
Can I take beta blockers with retatrutide?
Beta blockers can be taken concurrently with retatrutide, but they may not fully control the heart rate increase because retatrutide works through the glucagon receptor cAMP-PKA pathway rather than the sympathetic nervous system that beta blockers target. Discuss specific medication interactions with your physician, as dose adjustments to either medication may be needed.
Does the heart rate effect go away after stopping retatrutide?
Yes. Heart rate changes observed during treatment return to pre-treatment baseline after discontinuation. The effect is pharmacological (dependent on the presence of the drug) rather than structural (permanent changes to the heart). Once retatrutide is cleared from the body, glucagon receptor stimulation ceases and heart rate normalizes. This is also true for other GLP-1 compounds after discontinuation.
How does retatrutide heart rate compare to semaglutide heart rate?
Retatrutide produces approximately double the heart rate increase of semaglutide. Semaglutide typically increases resting heart rate by 2 to 3 BPM, while retatrutide increases it by 5 to 10 BPM. The difference is driven by retatrutide glucagon receptor activation, which directly stimulates cardiac tissue through a mechanism semaglutide does not engage. Both compounds produce cardiovascular benefits that appear to outweigh the heart rate effect.
Should I lower my retatrutide dose if my heart rate increases?
Not automatically. A heart rate increase of 5 to 10 BPM without symptoms is expected and generally acceptable. Consider dose reduction if your resting heart rate consistently exceeds 100 BPM, if you experience persistent palpitations or chest discomfort, or if the heart rate increase significantly affects your exercise tolerance. The appropriate retatrutide dose balances efficacy with tolerability across all side effect domains, not just heart rate.
Does exercise help manage retatrutide heart rate effects?
Yes. Regular aerobic exercise improves vagal tone and lowers resting heart rate over time, which can offset retatrutide pharmacological heart rate increase. Researchers who maintain consistent exercise routines while on retatrutide often see their resting heart rate stabilize or decrease below baseline over months, despite the drug effect. Exercise during retatrutide treatment serves as both a weight loss enhancer and a cardiovascular management strategy.
External resources
PMC - Inotropic effects of retatrutide in isolated human atrial preparations
ClinicalTrials.gov - Effect of Retatrutide on Cardiac Function
PMC - Contractile effects of retatrutide in isolated mouse atrial preparations
AHA Circulation - GLP-1 Receptor Agonists for Cardiovascular Risk Reduction
In case I do not see you, good afternoon, good evening, and good night. May your heart rate stay steady, your protocols stay informed, and your cardiovascular health stay strong.