Mar 23, 2026

Tired of conflicting information about oral tirzepatide drops? You are not alone. Some providers claim sublingual tirzepatide works just as well as injections. Others call it a waste of money. Reddit threads swing wildly between enthusiastic five-star endorsements and frustrated warnings. And the science? It paints a very different picture than any of these voices suggest.
The truth about tirzepatide drops sits somewhere in the middle of this noise, buried under marketing claims, genuine user confusion, and a regulatory landscape that shifted dramatically when the FDA ended its temporary allowance for compounded tirzepatide formulations. Understanding what real users experience, what the data actually supports, and where the genuine risks hide requires cutting through layers of hype and fear alike. This guide does exactly that. Every claim gets measured against published research, every user report gets context, and every provider gets evaluated on the merits rather than the marketing.
SeekPeptides has tracked the evolution of oral tirzepatide formulations from their earliest compounding pharmacy offerings through the current regulatory environment, analyzing hundreds of user reports and cross-referencing them with the available scientific literature. What follows is the most comprehensive review of oral tirzepatide drops available anywhere, built on evidence rather than advertising.
What oral tirzepatide drops actually are
Before diving into reviews, you need to understand what these products actually contain. Oral tirzepatide drops are compounded sublingual formulations that use the same active ingredient found in FDA-approved injectable tirzepatide (sold as Mounjaro and Zepbound). The key difference is the delivery method. Instead of a weekly subcutaneous injection, these drops go under the tongue daily.
That sounds simple enough. It is not.
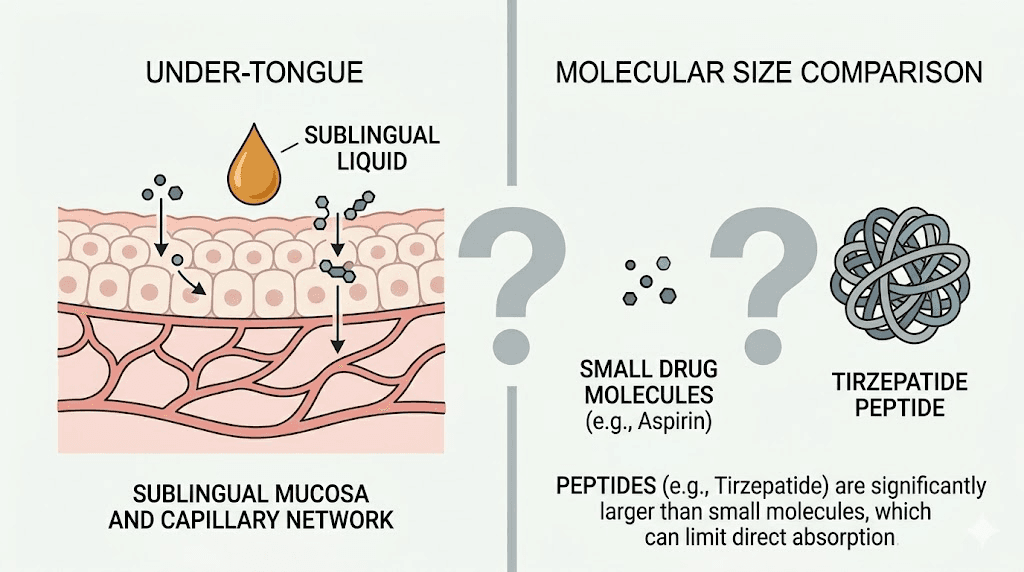
Tirzepatide is a dual GIP/GLP-1 receptor agonist, a large peptide molecule weighing approximately 4,810 daltons. This molecular weight matters enormously because it determines how well the peptide can cross biological membranes. For context, most drugs that absorb well through sublingual tissue weigh under 500 daltons. Tirzepatide is nearly ten times that threshold, and that single fact shapes everything about the oral drops debate.
Compounding pharmacies create these formulations by dissolving tirzepatide powder into liquid solutions designed for sublingual administration. Users place drops under the tongue, hold them for two to five minutes, and then swallow the remainder. The theory is that blood vessels beneath the tongue absorb the peptide directly into the bloodstream, bypassing the digestive system that would otherwise destroy the molecule.
The formulation landscape
Not all oral tirzepatide drops are created equal. Different providers use different formulations, concentrations, and additional ingredients. Some common variations include:
Basic sublingual drops contain tirzepatide dissolved in a liquid carrier. These are the most straightforward formulations and typically the least expensive option available from compounding providers.
Enhanced sublingual drops add absorption-enhancing compounds designed to improve bioavailability. Some pharmacies include permeation enhancers, surfactants, or other excipients that theoretically help the peptide cross the sublingual membrane more effectively. Whether these additions meaningfully improve absorption remains unproven in clinical settings.
Combination drops blend tirzepatide with other compounds like vitamin B12, glycine, or levocarnitine. Providers market these combinations as offering additional benefits beyond weight management, though the evidence base for sublingual delivery of these combinations is similarly limited.
Orally disintegrating tablets (ODTs) represent a slightly different approach. These are not liquid drops but rather solid tablets that dissolve under the tongue. The tirzepatide ODT format has gained some traction as an alternative to both liquid drops and injections, though it faces the same fundamental bioavailability challenges.
How sublingual absorption is supposed to work
The sublingual region beneath the tongue contains a dense network of capillaries. When a drug dissolves here, it can enter the bloodstream directly without passing through the gastrointestinal tract or undergoing first-pass metabolism in the liver. This works beautifully for small molecules like nitroglycerin (molecular weight: 227 daltons) and buprenorphine (molecular weight: 467 daltons).
The problem is scale. Tirzepatide is a peptide, not a small molecule. At 4,810 daltons, it faces significant barriers to crossing the sublingual epithelium. The tight junctions between epithelial cells in the mouth are designed to keep large molecules out, which is exactly what a peptide this size qualifies as. Research on sublingual peptide delivery consistently shows that without specialized absorption technology, bioavailability for large peptides ranges from negligible to nonexistent.
For comparison, FDA-approved oral semaglutide (Rybelsus) required the development of SNAC (sodium N-[8-(2-hydroxybenzoyl)amino] caprylate), a specialized absorption enhancer that took years of research and billions of dollars to develop. Even with this advanced technology, oral semaglutide achieves only about 1% bioavailability, meaning 99% of the dose never reaches the bloodstream. Compounded tirzepatide drops do not use SNAC or any equivalent technology.
The bioavailability problem nobody wants to discuss
This is where oral tirzepatide drops reviews get complicated. And where honest analysis diverges sharply from provider marketing.
Injectable tirzepatide achieves approximately 80% bioavailability. That means if you inject 5mg, roughly 4mg reaches your bloodstream and produces therapeutic effects. The absorption is consistent, predictable, and well-documented across thousands of clinical trial participants.
Sublingual tirzepatide drops? The bioavailability is unknown. Not "somewhat lower." Not "slightly reduced." Unknown. No published pharmacokinetic studies have measured sublingual tirzepatide absorption in humans. Zero peer-reviewed data exists on this specific question.
What we do know comes from the physics of sublingual absorption and research on similar-sized peptides. Even optimistic estimates, and these are truly generous projections, suggest sublingual bioavailability for a peptide of this molecular weight would fall well below 1%. Some researchers put the figure closer to 0.1% or less. That means a 5mg sublingual dose might deliver the equivalent of 0.005mg to the bloodstream, a dose so small it may produce no meaningful physiological effect whatsoever.
Why some users still report results
If the bioavailability math looks this bad, why do some reviews claim the drops work? Several explanations exist, and they are not mutually exclusive.
Swallowed absorption. After holding drops under the tongue, users swallow whatever remains. Some tirzepatide may survive the journey through the stomach (particularly if taken on an empty stomach) and achieve limited absorption in the intestines. This would be inconsistent and unpredictable, but it could produce some effect in some users some of the time.
Placebo response. Weight loss interventions carry notoriously strong placebo effects. Studies consistently show that participants in placebo arms of weight loss trials lose 2-5% of their body weight simply from the behavioral changes that come with enrolling in a treatment program, including dietary awareness, increased motivation, and regular check-ins with healthcare providers.
Concurrent lifestyle changes. Many people who start oral tirzepatide drops simultaneously improve their diet, increase exercise, and adopt other weight management strategies. The dietary changes recommended alongside tirzepatide alone can produce meaningful weight loss, making it difficult to attribute results specifically to the drops.
Variable formulation quality. Not all compounding pharmacies use the same techniques, concentrations, or quality control measures. Some formulations may achieve slightly better absorption than others, though none have demonstrated anything approaching injectable bioavailability in controlled testing.
Higher dosing. Because providers understand the absorption limitations, many prescribe significantly higher daily doses of oral tirzepatide compared to the weekly injectable dose. If even a small fraction absorbs, the cumulative daily exposure might approach a minimally effective level for some individuals. This strategy introduces its own risks, including cost inefficiency and unpredictable dose-to-dose variation.
What real users actually report
With that scientific context established, let us examine what users actually experience when taking oral tirzepatide drops. These reports come from online forums, telehealth provider review pages, social media discussions, and community groups dedicated to weight loss peptide research.
Positive experiences
Users who report positive outcomes with oral tirzepatide drops typically describe a consistent pattern. Appetite suppression appears first, usually within the first one to two weeks. It tends to be milder than what injection users report, which makes pharmacological sense given the likely lower blood levels. Weight loss follows, with most positive reviewers citing 5 to 15 pounds over the first two to three months.
The convenience factor dominates positive reviews. People who have needle phobia or travel frequently appreciate not having to store and inject a medication weekly. The daily ritual of drops feels more manageable to some users than the weekly injection anxiety they describe experiencing with injectable tirzepatide.
Some users specifically praise the gentler side effect profile. Because the effective dose reaching the bloodstream is likely lower than with injections, the gastrointestinal side effects that often accompany injectable tirzepatide, particularly constipation, diarrhea, and nausea, tend to be milder with the oral formulation. Whether this represents a benefit or simply a sign of subtherapeutic dosing is a matter of perspective.
Mixed experiences
A significant portion of reviews fall into the middle ground. These users report some appetite reduction but describe it as inconsistent. Good days alternate with days of normal hunger. Weight loss occurs but plateaus quickly compared to the steady progression typically seen with injectable tirzepatide timelines.
Several recurring themes emerge in mixed reviews:
The absorption variability frustrates many users. Taking the drops at the same time, holding them for the same duration, and following the same fasting protocol does not produce the same appetite suppression from day to day. This inconsistency aligns perfectly with what the bioavailability science predicts. Sublingual absorption of large peptides is inherently variable because it depends on factors like sublingual blood flow, saliva production, mucosal integrity, and even how much the user moves their tongue during the holding period.
Cost concerns appear frequently. Oral tirzepatide drops typically cost $200 to $400 per month depending on the provider and dosage. While this is often less than brand-name injectable tirzepatide, users note that the results frequently do not match the investment when compared to more affordable injectable options that deliver more predictable outcomes.
Some users describe a frustrating cycle: the drops seem to work initially, then effectiveness fades. They increase their dose, experience a brief resurgence of appetite suppression, then plateau again. This pattern is consistent with the body reaching a steady state where even the small amount of absorbed tirzepatide no longer provides enough stimulation to overcome natural appetite regulatory mechanisms.
Negative experiences
Negative reviews cluster around a few key complaints. The most common is simply that the drops do not work. No appetite suppression. No weight loss. No discernible effect whatsoever. These users often report having switched from injectable tirzepatide or semaglutide sublingual formulations to drops and immediately noticing the difference in effectiveness.
Some users report unpleasant taste. Tirzepatide in liquid form can have a chemical, bitter, or metallic flavor that makes the two-to-five-minute sublingual hold uncomfortable. This is a minor issue but one that affects daily compliance, and daily compliance matters significantly more for a sublingual formulation than for a weekly injection.
The regulatory uncertainty bothers many reviewers. Multiple users describe anxiety about the legal status of their medication, especially following the FDA decision to end the temporary shortage designation for tirzepatide that had previously allowed broader compounding. Questions about grey market tirzepatide sources and their legitimacy appear repeatedly in negative reviews.
A smaller but notable group reports unexpected side effects. While oral tirzepatide generally causes fewer GI issues than injections, some users experience unusual symptoms including mouth irritation, tongue numbness, burning sensation under the tongue, and persistent dry mouth. These local side effects are specific to the sublingual delivery method and do not occur with injectable administration.
Provider-by-provider review breakdown
Multiple telehealth companies now offer oral tirzepatide drops, each with different formulations, pricing, and patient support. Understanding the differences matters because provider quality directly affects user experience.
Shed
Shed offers tirzepatide GLP-1 drops through a telehealth platform with physician oversight. Their standard titration protocol starts at 3mg daily for the first four weeks, then increases to 4mg daily for maintenance. Shed combines tirzepatide with additional ingredients in their formulation and provides ongoing virtual support including messaging access between visits.
User reviews for Shed tend to be moderately positive, with most praise directed at customer service and the convenience of the platform rather than dramatic weight loss results. The monthly cost starts around $229, positioning Shed in the mid-range of the oral tirzepatide market. Users consistently mention that the onboarding process is straightforward and that response times from their care team are reasonable.
The limitations users identify include relatively modest appetite suppression compared to injectable tirzepatide experiences and the daily dosing requirement that some find burdensome despite initially choosing drops specifically for convenience.
Willow
Willow markets tirzepatide oral drops as part of a broader weight management program. Their approach emphasizes the needle-free advantage and positions the drops as appropriate for patients who are new to GLP-1 therapy or who have anxiety about injections.
Willow tirzepatide pricing tends to be competitive, and their reviews generally reflect satisfaction with the customer experience. However, a pattern emerges in longer-term reviews: users who stay on drops for three months or more frequently express disappointment in the total weight lost compared to expectations set by injectable tirzepatide clinical trial data.
Strive Pharmacy
Strive Pharmacy compounds tirzepatide as SubMagna sublingual drops, a formulation they describe as designed for enhanced sublingual absorption. Strive is one of the few providers that directly addresses the bioavailability challenge in their marketing, acknowledging that sublingual delivery differs from injectable and claiming their formulation technology improves upon basic sublingual solutions.
Reviews for Strive are mixed. Some users report better results than they experienced with other providers of oral drops, potentially suggesting that formulation differences do matter. Others see no meaningful difference. The pharmacy has a following among users who appreciate the transparency about sublingual limitations while still wanting a needle-free option.
Daytryp
Daytryp combines tirzepatide drops with vitamin B12 and glycine in a sublingual troche formulation. Their approach positions the product as both a weight management tool and a general wellness supplement. The tirzepatide glycine B12 combination is common among compounding pharmacies, though the evidence for synergistic benefits of this specific combination in sublingual form remains limited.
User reviews for Daytryp frequently mention the daily dosing schedule as both a pro and a con. Proponents argue that daily microdosing provides more stable blood levels (though this assumes meaningful absorption occurs). Critics counter that remembering to take drops every morning, holding them under the tongue for one to two minutes, and then fasting for ten to fifteen minutes afterward transforms a once-weekly task into a daily ritual.
Other providers
Numerous other telehealth platforms and compounding pharmacies offer oral tirzepatide drops. Elevate Health, Ivim Health, Prime Health, and Belle Health all offer variations of sublingual tirzepatide with varying prices, formulations, and support levels. The common thread across provider reviews is that customer service quality often correlates more strongly with satisfaction than the actual drops do with weight loss outcomes.
Drops versus injections: the head-to-head comparison users actually want
Every person researching oral tirzepatide drops reviews wants to know the same thing. How do they compare to injections? The honest answer is not encouraging for drops enthusiasts, but it is nuanced enough to deserve a thorough examination.
Effectiveness
Injectable tirzepatide, studied in the SURMOUNT clinical trial program involving thousands of participants, produced average weight loss of 15 to 22.5% of body weight over 72 weeks at the highest doses. These results are supported by rigorous, peer-reviewed research published in leading medical journals. The before and after results from injectable tirzepatide are among the most impressive in weight management pharmacotherapy history.
Oral tirzepatide drops have zero published clinical trial data. None. Not a single randomized controlled trial. Not a single pharmacokinetic study in humans. Not even a small pilot study published in a peer-reviewed journal. Every claim about effectiveness comes from anecdotal user reports, provider marketing materials, or extrapolation from injectable data.
This is not a small gap. This is a canyon.
Among user reports, the general consensus suggests oral drops produce less appetite suppression, slower weight loss, and less total weight lost compared to properly dosed injectable tirzepatide. Users who have tried both overwhelmingly prefer injections for effectiveness, even when they prefer drops for convenience.
Consistency
Injectable tirzepatide delivers consistent blood levels when administered as directed. The weekly injection produces a predictable pharmacokinetic curve that maintains therapeutic concentrations throughout the dosing interval. Users know what to expect from week to week.
Oral drops deliver inconsistent blood levels by nature. Two people taking the same oral formulation from the same pharmacy might achieve dramatically different blood concentrations. The same person taking their drops on different days might absorb different amounts depending on sublingual blood flow, hydration status, food timing, and a dozen other variables. This inconsistency shows up clearly in user reviews, where day-to-day variation in appetite suppression is the single most common complaint.
Convenience
This is where drops genuinely win, and it matters more than some dismiss.
No needles. No refrigeration in most cases. No injection site reactions, no redness or itching at injection sites, no bruising. Easy to travel with (no cold chain required), easy to store, and easy to take without anyone noticing. For people with severe needle phobia, the convenience advantage is not trivial.
The daily dosing cuts both ways though. Weekly injections require remembering once per week. Daily drops require remembering every single morning, plus the two-to-five-minute sublingual hold, plus the post-dose fasting period. Over time, many users find that daily compliance is harder to maintain than weekly compliance.
Side effects
Injectable tirzepatide commonly causes nausea (affecting roughly 25-30% of users at higher doses), diarrhea, constipation, and decreased appetite beyond what is therapeutically intended. These side effects are dose-dependent and typically improve with time and proper dose titration.
Oral tirzepatide drops generally produce fewer systemic side effects, which is consistent with lower systemic absorption. The trade-off is local side effects that injections do not cause: mouth irritation, taste disturbances, tongue numbness, and throat irritation. Users must weigh fewer GI side effects against these oral-specific discomforts.
It is worth noting that fatigue, body aches, and headaches are reported with both delivery methods, though typically at lower rates with oral formulations. Again, this likely reflects lower systemic drug exposure rather than a genuine safety advantage of the oral route.
Cost comparison
Brand-name injectable tirzepatide (Mounjaro/Zepbound) costs $1,000+ per month without insurance. Compounded injectable tirzepatide (when available through legal channels) ranges from $150 to $400 per month depending on the provider and pharmacy. Oral tirzepatide drops typically cost $200 to $450 per month.
On a pure cost basis, oral drops often cost the same or more than compounded injections while delivering less predictable results. When you factor in cost per unit of actual weight lost, injections almost certainly provide better value. Affordable injectable tirzepatide options exist that outperform oral drops on both effectiveness and price.

The FDA and regulatory reality
Understanding the regulatory environment is essential for anyone reading oral tirzepatide drops reviews, because it directly affects product availability, quality, and legality.
Current FDA status
As of early 2026, no oral tirzepatide formulation has received FDA approval. The only FDA-approved tirzepatide products are the injectable formulations Mounjaro (for type 2 diabetes) and Zepbound (for weight management). Any oral tirzepatide drops, tablets, troches, or sublingual formulations are compounded products that have not undergone FDA review for safety, efficacy, or manufacturing quality.
The FDA removed tirzepatide from its drug shortage list, which ended the temporary regulatory flexibility that had allowed compounding pharmacies to produce tirzepatide formulations. Under the Federal Food, Drug, and Cosmetic Act, pharmacies can compound medications that are on the drug shortage list. Once tirzepatide came off that list, the legal basis for compounding shifted significantly.
The timeline matters:
For 503A pharmacies (traditional compounding pharmacies), the FDA temporary leniency ended in February 2025. For 503B outsourcing facilities (larger-scale compounding operations), the deadline was March 2025. After these dates, the legal landscape for compounded tirzepatide became considerably more complex.
What this means for oral drops users
Users reviewing oral tirzepatide drops should understand that the products they are evaluating exist in a regulatory grey area. This does not necessarily mean the products are dangerous, but it does mean they lack several safeguards that FDA-approved products provide.
No FDA review of efficacy means no one has verified that these drops work as claimed. No FDA review of manufacturing means no one has verified that the drops contain what the label says in the amounts the label claims. No post-market surveillance means adverse events may go unreported and untracked.
The FDA had collected 480 reports of adverse events involving compounded tirzepatide products as of April 2025. While this number must be contextualized against the millions of doses administered, it underscores that quality control issues in compounded products are not hypothetical.
Impact on user reviews
The regulatory situation creates a selection bias in online reviews that readers should recognize. Users who obtained oral tirzepatide drops from high-quality compounding pharmacies with rigorous quality control may have genuinely different experiences than users who received products from less scrupulous sources. Reviews do not always distinguish between these scenarios, making it difficult to evaluate whether positive or negative experiences reflect the oral delivery method itself or the quality of the specific product received.
The science behind user experiences
Moving beyond individual reviews to examine what the scientific literature tells us about why users report what they report.
Why the absence of evidence matters
The phrase "absence of evidence is not evidence of absence" gets invoked frequently in oral tirzepatide discussions. While technically true in a philosophical sense, this argument misses the practical point entirely.
For injectable tirzepatide, we have evidence from multiple Phase III clinical trials involving over 10,000 participants, published in the New England Journal of Medicine, The Lancet, and JAMA. We know exactly how much weight people lose, how quickly, at what doses, with what side effects, and with what consistency.
For oral tirzepatide drops, we have nothing comparable. Not because no one thought to study it, but because the basic pharmacological principles suggest the approach faces fundamental absorption barriers that existing formulation technology has not overcome.
The existing research on tirzepatide oral versus injection delivery consistently points to injectable administration as the only clinically validated route. This does not mean oral delivery is impossible in principle. It means the current products have not been demonstrated to work.
Peptide absorption research
Oral peptide delivery is one of the most active areas of pharmaceutical research. The challenge of getting large peptide molecules across biological barriers has occupied scientists for decades. The success stories, like oral semaglutide (Rybelsus), required:
Specialized absorption enhancers (SNAC technology). Novel formulation science. Years of clinical trials. Billions in research investment. Even then, bioavailability reached only about 1%.
Compounded tirzepatide drops represent formulations created without this level of scientific investment. The absorption-enhancing technologies used in compounding are generations behind what pharmaceutical companies employ. This does not mean zero absorption occurs, but it means expecting comparable results to injectable administration, or even to the modest absorption that Rybelsus achieves, is not scientifically justified.
Research on tirzepatide tablet formulations versus injections consistently finds that without advanced absorption technology, oral tirzepatide cannot match injectable pharmacokinetics. Eli Lilly, the manufacturer of Mounjaro and Zepbound, is actively developing oral tirzepatide formulations using proprietary technology, but these remain in clinical trials and are not yet available.
What the rat study actually showed
One study frequently cited in discussions of sublingual tirzepatide absorption was actually conducted on semaglutide, not tirzepatide, and used rats, not humans. The study demonstrated that sublingual delivery of semaglutide achieved measurable absorption in 24 rats using a single dose. The authors explicitly stated that the study was "not intended to provide guidance on the sublingual dosing of semaglutide in humans."
Extrapolating from a rat study on a different peptide to human use of tirzepatide drops involves multiple leaps of scientific faith that responsible review cannot endorse.
Dosing protocols and what users report about them
Understanding the dosing protocols for oral tirzepatide drops helps contextualize user reviews and expectations.
Standard dosing approaches
Most providers follow a titration schedule that starts low and increases gradually, mirroring the approach used with injectable tirzepatide but adapted for daily oral use.
A typical protocol looks like this:
Weeks 1 through 4: 2 to 3mg daily. This initiation phase allows the body to adjust and helps providers assess tolerance. Users at this stage commonly report minimal appetite suppression and question whether the drops are working. Patient reviewers frequently describe this period as discouraging, especially those who have read about the dramatic appetite reduction that injectable GLP-1 medications produce early in treatment.
Weeks 5 through 8: 3 to 5mg daily. The dose increases, and some users begin noticing appetite changes. Reviews from this period are more mixed, with a subset of users reporting clear appetite suppression while others still feel nothing. The variability is the hallmark feature of sublingual delivery and the most consistent theme across user reports.
Weeks 9 and beyond: 4 to 8mg daily (maintenance). At maintenance doses, the user experience diverges significantly. Positive reviewers describe stable appetite management and steady weight loss. Negative reviewers describe persistent inconsistency or complete lack of effect. There is very little middle ground in long-term reviews.
How to take the drops properly
User reviews consistently show that technique matters with sublingual drops, far more than it does with injections. The proper technique for taking GLP-1 drops involves several steps that, if done incorrectly, may further reduce an already questionable absorption rate.
Timing: Take drops first thing in the morning on an empty stomach. The sublingual tissues absorb more efficiently when not competing with food particles or excess saliva from recent eating.
Placement: Place the prescribed number of drops directly under the tongue, not on top of it or in the cheek. The sublingual area has the highest concentration of blood vessels in the oral cavity.
Hold duration: Keep the liquid under the tongue for at least two minutes. Some providers recommend up to five minutes. Moving the tongue, talking, or swallowing during this period reduces absorption. Users who find this holding period difficult should know that it represents one of the most significant factors in determining how much tirzepatide actually reaches their bloodstream.
Post-dose fasting: Avoid eating or drinking for 10 to 30 minutes after dosing. This prevents food or liquid from washing away any tirzepatide still being absorbed from the sublingual tissues.
Users who follow these steps meticulously report better results than those who rush through the process, though even perfect technique cannot overcome the fundamental bioavailability limitations of sublingual peptide delivery.
Microdosing approaches
Some users have experimented with tirzepatide microdosing protocols adapted for oral drops, splitting their daily dose into two or three smaller doses taken at different times. The theory is that more frequent exposure might maintain steadier blood levels throughout the day.
Reviews of microdosing approaches are inconsistent. Some users believe splitting doses improved their experience. Others noticed no difference. Without pharmacokinetic data on sublingual tirzepatide absorption patterns, there is no scientific basis to recommend one dosing frequency over another.
Side effects reported with oral tirzepatide drops
The side effect profile of oral tirzepatide drops differs from injectable tirzepatide in important ways that user reviews consistently highlight.
Gastrointestinal effects
The most commonly reported GI side effects are nausea and decreased appetite. However, users consistently describe these as milder than what they experienced or expected from injectable tirzepatide. This aligns with the hypothesis that less tirzepatide reaches the bloodstream via sublingual delivery.
Foods to avoid while taking tirzepatide remain relevant for oral drop users, though the dietary recommendations are the same regardless of delivery method. High-fat and greasy foods tend to worsen nausea, while lean proteins and fiber-rich foods are generally better tolerated.
Constipation is reported less frequently with oral drops than with injections. Bloating and gas still occur but at seemingly lower rates. Some users interpret this as a benefit, while others recognize it as a potential sign that insufficient tirzepatide is reaching the GI tract receptors that mediate these effects.
Oral-specific side effects
These side effects are unique to the sublingual delivery method and do not occur with injectable administration:
Taste disturbances. Many users describe a bitter, chemical, or metallic taste during and after the sublingual hold. Some formulations are worse than others, and taste preferences vary, but this is among the most common complaints in oral drops reviews.
Mouth and tongue irritation. The concentrated peptide solution can irritate the delicate sublingual tissues with repeated daily exposure. Users describe numbness, tingling, burning, or soreness under the tongue that sometimes persists for hours after dosing.
Dry mouth. Persistent dry mouth with tirzepatide occurs with both delivery methods but appears more pronounced with sublingual drops, likely due to the direct contact with oral tissues and the post-dose fasting requirement that limits fluid intake.
Throat irritation. When swallowing the remaining liquid after the sublingual hold, some users experience throat burning or irritation. This typically resolves with water intake after the recommended fasting period.
Systemic side effects
Users report the same systemic side effects as injectable tirzepatide, but generally at lower intensity:
Insomnia is mentioned in some reviews, particularly among users taking their dose in the evening (most providers recommend morning dosing). Anxiety has been reported by a small number of users. Energy changes vary widely, with some users reporting increased energy and others reporting fatigue.
The lower incidence of systemic side effects with oral drops is a double-edged sword. Fewer side effects mean a more comfortable experience, but they may also mean less drug is reaching systemic circulation to produce therapeutic effects. For first-time tirzepatide users, the gentle introduction that oral drops provide may be appealing, but they should understand that the subdued side effect profile likely reflects subdued therapeutic action as well.
Who oral tirzepatide drops might work for
Despite the significant limitations, there are specific scenarios where oral tirzepatide drops might represent a reasonable choice. This assessment comes from analyzing user reviews alongside the scientific constraints.
People with severe needle phobia
True needle phobia (trypanophobia) affects an estimated 10% of the population and can be severe enough to prevent medical treatment entirely. For someone whose needle phobia would otherwise prevent them from using any injectable GLP-1 medication, oral drops represent something rather than nothing. Even if the drops deliver a fraction of the therapeutic effect, that fraction exceeds zero.
Users in this category tend to leave the most positive reviews proportional to their expectations. They understand they may get less dramatic results but value having any accessible option at all. These reviewers often mention that they plan to transition to injections eventually, using drops as a bridge while working on their needle anxiety with injection technique education and desensitization.
People seeking a gentle introduction
Some users start with oral drops specifically because they want to experience tirzepatide effects at a lower intensity before committing to injections. The gentler appetite suppression and milder side effects serve as a preview of what the medication does without the full-force impact of injectable delivery.
Reviews from this user segment tend to be positive initially but transition to disappointment as expectations grow. Once they understand what injectable tirzepatide can do, many switch. The oral drops served their purpose as a stepping stone.
People who need extreme portability
Frequent travelers, military personnel, outdoor enthusiasts, and others whose lifestyles make cold-chain storage and injection supplies impractical sometimes choose oral drops for logistical reasons. Traveling with injectable tirzepatide requires cooler packs, syringes, and sometimes documentation. Drops require only a small bottle.
Reviews from this category emphasize convenience over effectiveness and typically acknowledge the trade-off explicitly.
People with mild weight loss goals
Users seeking modest weight loss, perhaps 5 to 10 pounds, sometimes find oral drops adequate for their needs. The partial appetite suppression may provide just enough behavioral nudge to reduce caloric intake sufficiently for small-scale weight management. For these users, the limited effectiveness of drops aligns with their limited ambitions.
Red flags in oral tirzepatide drops reviews
Not all reviews are created equal, and several red flags should prompt skepticism when reading oral tirzepatide drops reviews online.
Claims of equivalent effectiveness to injections
Any review or provider that claims oral tirzepatide drops work "just as well" as injections is either misinformed or dishonest. The pharmacology does not support this claim, and the overwhelming weight of user experience contradicts it. Reviews making this claim should be treated with extreme caution.
Dramatic before-and-after results
When tirzepatide before and after results are attributed specifically to oral drops and show dramatic transformations, question whether those results came solely from the drops or from a comprehensive weight management program that happened to include drops. Context matters enormously. A user who lost 40 pounds on oral tirzepatide drops while simultaneously starting a rigorous exercise program and working with a nutritionist did not lose 40 pounds because of the drops alone.
Reviews that never mention limitations
Genuine reviews typically include both positives and negatives. Reviews that describe nothing but miraculous results with zero downsides are almost certainly promotional content rather than authentic user experiences. Real users complain about taste, inconsistency, cost, or some aspect of their experience.
Provider-hosted reviews without verification
Reviews hosted exclusively on provider websites lack independent verification. While some provider reviews are genuine, the selection bias is obvious. Companies display their best reviews and suppress their worst. Look for reviews on independent platforms, forums, and social media groups where the provider does not control what appears.
Reviews from very early users
Reviews written during the first two to four weeks of oral tirzepatide use carry limited weight. The initiation period is too early for meaningful weight loss assessment, and the novelty effect can create optimistic bias. Reviews from users who have taken drops for three months or longer provide much more useful information about real-world effectiveness.
How to evaluate oral tirzepatide drops for yourself
If you are considering oral tirzepatide drops despite the limitations this review identifies, here is a framework for making an informed decision.
Questions to ask your provider
Before starting oral tirzepatide drops, ask your prescribing provider these specific questions. Their answers will tell you a lot about their honesty and expertise.
What bioavailability data supports this formulation? If they cite clinical trials, ask which specific trials studied sublingual tirzepatide in humans. (The answer is none.) A provider who acknowledges the limitations while explaining their rationale for prescribing is more trustworthy than one who overpromises.
Which compounding pharmacy fills the prescription? Research the pharmacy independently. Is it a 503A or 503B facility? What quality control measures do they employ? Do they test final products for potency and purity? Legitimate pharmacies will answer these questions transparently.
What results should I realistically expect? A responsible provider will not promise that oral drops will match injectable results. They should discuss the likely range of outcomes honestly, including the possibility that drops may not work for some patients.
What is the plan if drops are not effective? Providers who have a clear escalation pathway, typically transitioning to injectable tirzepatide if drops prove inadequate, demonstrate patient-centered care. Those who only push higher doses of drops or additional oral products may be prioritizing their business model over your outcomes.
Setting realistic expectations
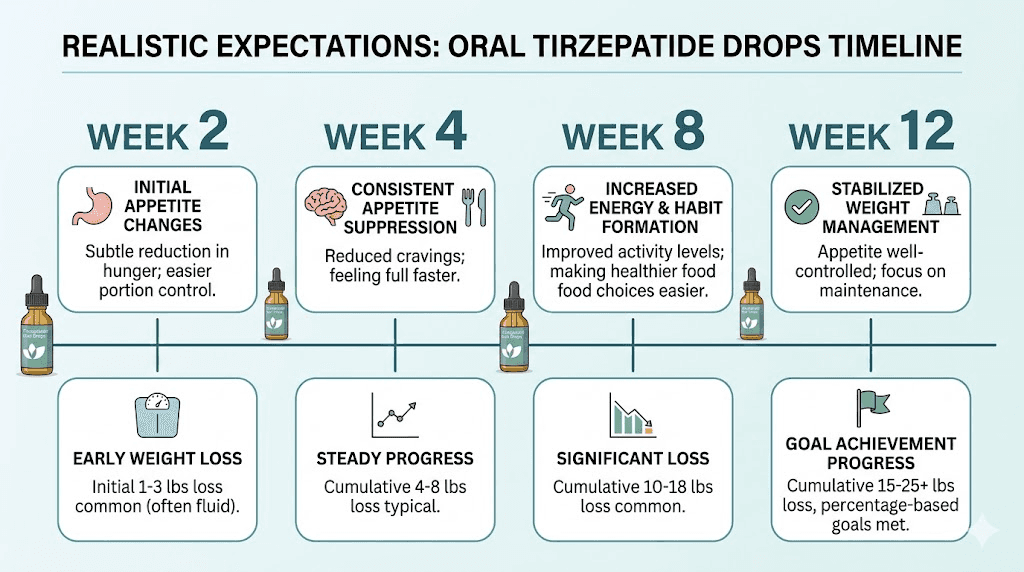
Based on the aggregate of user reviews, realistic expectations for oral tirzepatide drops include:
Appetite suppression: Mild to moderate, inconsistent day to day, typically 30 to 50% of what injectable users report. Some users experience none at all.
Weight loss: 3 to 8% of body weight over 3 to 6 months for users who respond. This compares to 15 to 22.5% for injectable tirzepatide over similar or longer timeframes. Some users will not lose meaningful weight on drops alone.
Timeline: Slower onset than injections. Most users who respond begin noticing effects at weeks 4 to 8, compared to weeks 1 to 3 for injectable users. Full effects, if they materialize, may take 3 to 4 months to develop.
Consistency: Variable. Good days and bad days are normal with sublingual delivery. This differs markedly from the steady, predictable effect of properly administered injectable tirzepatide.
Tracking your results
If you decide to try oral tirzepatide drops, systematic tracking is essential. Without it, you cannot objectively evaluate whether the drops are working for you specifically.
Track these metrics weekly:
Body weight (same time, same conditions). Waist measurement. Appetite level on a 1-to-10 scale. Any side effects and their severity. Compliance (did you take the drops every day with proper technique?).
Give the drops a fair trial of at least 8 to 12 weeks at adequate dosing before concluding they do not work. But also set a deadline. If after 12 weeks of consistent, properly-administered drops you have not lost at least 3% of your starting body weight, the drops are very likely not providing therapeutic benefit for you specifically. At that point, transitioning to injectable tirzepatide would be the evidence-based next step.
SeekPeptides members access detailed tracking tools and protocol guides that help evaluate whether any peptide therapy is producing meaningful results, including frameworks for deciding when to adjust, switch, or escalate treatment approaches.
The future of oral tirzepatide
The story of oral tirzepatide is not over. In fact, the most important chapters have not been written yet.
Eli Lilly oral tirzepatide development
Eli Lilly, the company behind Mounjaro and Zepbound, is actively developing oral tirzepatide formulations using proprietary technology that addresses the bioavailability challenges that compounded drops cannot solve. Their approach involves advanced absorption-enhancing technology similar to what made oral semaglutide (Rybelsus) possible, but optimized for tirzepatide.
Clinical trials for pharmaceutical-grade oral tirzepatide are ongoing. If successful, these products would offer:
Validated bioavailability backed by clinical data. Consistent dosing confirmed through pharmacokinetic studies. FDA approval ensuring manufacturing quality. Insurance coverage potential.
This represents a fundamentally different product than current compounded drops, even though both would be "oral tirzepatide." The difference is billions of dollars of research and development, rigorous clinical testing, and regulatory oversight.
What this means for current drop users
Users currently taking compounded oral tirzepatide drops should recognize that the products they use today are essentially placeholder versions of what pharmaceutical companies are working to develop properly. The concept of oral tirzepatide is not flawed. The execution of current compounded products is limited by the technology available to compounding pharmacies.
For those who find current drops helpful, continue using them while monitoring results objectively. For those who are disappointed, injectable tirzepatide remains the gold standard while waiting for pharmaceutical-grade oral formulations to potentially reach the market.
Emerging delivery technologies
Beyond sublingual drops, researchers are exploring other needle-free tirzepatide delivery methods including:
Transdermal patches using microneedle technology that could deliver peptides through the skin. These are in early research stages and face their own absorption challenges, but the approach is fundamentally different from sublingual delivery.
Oral tablets with absorption enhancers similar to the SNAC technology used in Rybelsus. Multiple pharmaceutical companies are pursuing this approach for various GLP-1 receptor agonists.
Nasal spray formulations that deliver tirzepatide through the nasal mucosa. Some evidence suggests nasal delivery may be more effective than sublingual for certain peptides due to the different characteristics of nasal epithelial tissue.
None of these alternatives are commercially available yet for tirzepatide specifically, but they represent the direction the field is moving. The goal of effective, needle-free tirzepatide delivery is achievable. It simply has not been achieved by current compounded products.
Comparing oral tirzepatide drops with other needle-free options
For users whose primary motivation is avoiding needles, oral tirzepatide drops are not the only option worth considering.
Oral semaglutide (Rybelsus)
Oral semaglutide is the only FDA-approved oral GLP-1 medication. Rybelsus uses SNAC absorption technology and has been studied in multiple clinical trials demonstrating meaningful weight loss. While it may produce somewhat less weight loss than injectable semaglutide or tirzepatide, it represents a scientifically validated needle-free option.
The key advantage over compounded oral tirzepatide drops: Rybelsus actually works with documented, reproducible evidence. The disadvantage: it requires a prescription, may cost more, and some users prefer the dual GIP/GLP-1 action of tirzepatide over the GLP-1-only mechanism of semaglutide.
Compounded semaglutide sublingual drops
Semaglutide sublingual formulations face the same bioavailability limitations as tirzepatide drops but have a slightly longer track record in the compounding market. User reviews for semaglutide drops are remarkably similar to tirzepatide drop reviews: mixed effectiveness, inconsistent absorption, convenience praised, effectiveness questioned.
Some users who find tirzepatide drops ineffective try switching to semaglutide drops or vice versa. The underlying absorption challenges are nearly identical for both peptides, so switching between sublingual formulations rarely solves the core problem.
Tirzepatide troches and ODTs
Tirzepatide orally disintegrating tablets dissolve in the mouth and are intended to be absorbed through the oral mucosa. Troches are similar but use a lozenge format. Both face the same fundamental bioavailability limitations as liquid drops, though some users report slightly different experiences with solid versus liquid sublingual formulations.
Reviews suggest that troches and ODTs may be more convenient than liquid drops (no measuring, no dripping) but do not offer meaningfully different effectiveness. The formulation vehicle matters less than the biological reality of trying to push a large peptide through oral tissues.
What experienced users recommend
After analyzing hundreds of reviews from users who have tried oral tirzepatide drops for three months or longer, several practical recommendations emerge consistently.
Give injections a real chance first
The most common advice from experienced users is straightforward: try injectable tirzepatide first if you can tolerate needles at all. The effectiveness difference is substantial enough that most users who try both prefer injections. Modern insulin syringes used for tirzepatide use extremely thin 30 to 31 gauge needles that most people describe as nearly painless.
Users who dreaded injections but eventually tried them overwhelmingly report that the anticipation was worse than the reality. The weekly injection takes seconds, produces minimal discomfort, and delivers dramatically more consistent results than daily drops.
If choosing drops, optimize your technique
Users who get the best results from oral drops consistently emphasize proper technique. Morning dosing on a completely empty stomach. Full two-to-five-minute sublingual hold without moving the tongue. No food or drink for at least 15 minutes afterward. Consistent daily timing. These details matter more for sublingual delivery than for any other administration route.
Track everything from day one
Experienced drop users recommend starting a detailed log on day one. Record your weight, measurements, appetite ratings, side effects, and dosing details daily. This data eliminates the guesswork about whether the drops are actually working or whether other factors (diet changes, increased activity, stress reduction) deserve the credit.
Set a clear evaluation timeline
Most experienced users recommend giving drops 8 to 12 weeks before making a final judgment. Shorter trials may not capture the full titration effects. Longer trials without measurable results may simply waste time and money. If you have not seen at least 3% body weight loss after 12 weeks of consistent use at an adequate dose, the drops are likely not providing sufficient therapeutic effect for you.
Consider drops as a complement, not a replacement
A few users report success using oral drops alongside a comprehensive weight management program that includes dietary modifications based on tirzepatide-friendly eating strategies, regular exercise, adequate sleep, and stress management. In this context, even partial appetite suppression from drops can provide a meaningful nudge. The drops become one tool among many rather than the sole intervention.
For researchers serious about optimizing their protocols and making evidence-based decisions, SeekPeptides offers the most comprehensive resource available, with detailed protocol guides, tracking tools, and a community of thousands who have navigated these exact decisions.
Cost analysis: what oral tirzepatide drops actually cost per pound lost
Cost matters. And when evaluating oral tirzepatide drops reviews, the cost-effectiveness calculation reveals an important story.
Monthly costs by provider
The typical monthly cost range for oral tirzepatide drops spans from approximately $200 to $450 depending on the provider, dosage, and formulation. Some providers include physician consultations in this price while others charge separately for telehealth visits.
For comparison, compounded injectable tirzepatide costs approximately $150 to $400 per month through similar telehealth channels. Brand-name injectable tirzepatide (Mounjaro/Zepbound) costs $1,000 or more per month without insurance but may be covered by some insurance plans.
Cost per pound of weight loss
This is where the math gets uncomfortable for oral drops advocates.
If a user spends $300 per month on oral tirzepatide drops and loses 8 pounds over 4 months (a realistic positive outcome based on user reviews), that works out to $150 per pound lost.
If that same user spent $250 per month on injectable tirzepatide and lost 25 pounds over 4 months (consistent with clinical trial data for properly dosed injections), that works out to $40 per pound lost.
The cost-per-pound difference is approximately 3 to 4 times higher for oral drops compared to injections, even when the monthly subscription cost is similar. This calculation does not include the potential costs of ineffective treatment, including delayed results, the psychological impact of slow progress, and the eventual cost of switching to injections after months of suboptimal response.
Hidden costs
Beyond the monthly subscription, oral tirzepatide drops users should budget for:
Follow-up consultations if the provider charges separately for dose adjustments. Switching costs if drops prove ineffective and a transition to injectable tirzepatide becomes necessary. Opportunity cost of delayed weight loss while using a less effective delivery method during the months when injectable treatment would have produced better results.
The bottom line on oral tirzepatide drops reviews
After examining hundreds of user reviews, analyzing the available science, comparing providers, and evaluating cost-effectiveness, the conclusions are clear but nuanced.
Oral tirzepatide drops are not a scam. They contain real tirzepatide. Some users genuinely experience appetite suppression and weight loss. The convenience of needle-free daily dosing appeals to real people with real concerns about injections.
But oral tirzepatide drops are also not equivalent to injectable tirzepatide. The scientific evidence for sublingual absorption of a peptide this size is essentially nonexistent. User reviews consistently show less effectiveness, less consistency, and less total weight loss compared to injections. The cost-per-result ratio significantly favors injectable administration.
The right choice depends entirely on individual circumstances. For the person with severe needle phobia who would otherwise forgo GLP-1 therapy entirely, drops offer a meaningful, if imperfect, alternative. For the person comfortable with weekly injections seeking maximum weight loss results, drops represent a significant compromise in effectiveness for a modest gain in convenience.
Read the reviews critically. Ask providers hard questions. Track your own results objectively. And remember that the most important review of oral tirzepatide drops is the one you will write yourself based on your own carefully measured experience.
SeekPeptides provides the tools, protocols, and community support to help you navigate these decisions with confidence, whether you choose oral drops, injectable tirzepatide, or another approach entirely. Evidence-based guidance and personalized protocol support are what set informed researchers apart from those guessing in the dark.
Frequently asked questions
Do oral tirzepatide drops actually work for weight loss?
Some users report appetite suppression and modest weight loss with oral tirzepatide drops, but results are inconsistent and significantly less dramatic than injectable tirzepatide. No clinical trials have studied sublingual tirzepatide in humans, so effectiveness claims rely entirely on anecdotal reports rather than scientific evidence. If you do try drops, track your results objectively and consider switching to injectable tirzepatide if you do not see meaningful results within 8 to 12 weeks.
How long do oral tirzepatide drops take to work?
Users who respond to oral tirzepatide drops typically notice initial appetite changes within 2 to 4 weeks, though meaningful weight loss usually takes 6 to 12 weeks to become apparent. This timeline is notably slower than injectable tirzepatide, where most users notice significant appetite suppression within the first 1 to 2 weeks. The slower onset with drops likely reflects lower and less consistent systemic absorption.
Are oral tirzepatide drops FDA approved?
No. As of early 2026, no oral tirzepatide formulation has received FDA approval. The only FDA-approved tirzepatide products are injectable formulations (Mounjaro for type 2 diabetes and Zepbound for weight management). All oral tirzepatide drops, tablets, and troches are compounded products that have not undergone FDA safety and efficacy review.
Can I switch from oral tirzepatide drops to injections?
Yes, and many users eventually make this transition. The transition from oral to injectable tirzepatide typically involves starting at the lowest injectable dose and titrating up following standard protocols, regardless of what oral dose you were taking previously. Because sublingual absorption is unpredictable, your oral dose does not reliably predict your appropriate injectable dose.
What is the best way to take tirzepatide drops?
For optimal sublingual absorption, take drops first thing in the morning on an empty stomach. Place them directly under the tongue and hold for 2 to 5 minutes without moving your tongue, talking, or swallowing. After the hold period, swallow any remaining liquid and avoid eating or drinking for 10 to 30 minutes. Consistent daily timing and technique are critical for maximizing whatever absorption the sublingual route can achieve. See our guide on how to take GLP-1 drops properly for detailed instructions.
Why do some people say tirzepatide drops work while others say they do not?
The variable results stem from multiple factors: inconsistent sublingual absorption between individuals, differences in formulation quality between compounding pharmacies, variations in user technique, concurrent lifestyle changes that may contribute to weight loss independently, and the well-documented placebo effect in weight loss interventions. Two people taking the same product from the same pharmacy may achieve very different blood levels and therapeutic responses.
How much weight can I lose on oral tirzepatide drops?
Based on aggregate user reviews, realistic expectations for oral tirzepatide drops are 3 to 8% of body weight over 3 to 6 months for users who respond to the treatment. Some users lose more, many lose less, and a significant portion report no meaningful weight loss. For comparison, injectable tirzepatide produces 15 to 22.5% body weight loss in clinical trials over 72 weeks at optimal doses.
Are oral tirzepatide drops safe?
The safety profile of oral tirzepatide drops has not been formally studied. Users report generally milder systemic side effects than injectable tirzepatide (likely due to lower absorption) but may experience oral-specific side effects including taste disturbances, tongue irritation, and dry mouth. The larger safety concern involves product quality, as compounded formulations do not undergo the same manufacturing oversight as FDA-approved products. The FDA collected 480 adverse event reports for compounded tirzepatide products as of April 2025.
External resources
FDA: Medications containing semaglutide or tirzepatide marketed for weight loss
PMC: Efficacy and safety of tirzepatide on weight loss, systematic review and meta-analysis
In case I do not see you, good afternoon, good evening, and good night. May your research stay evidence-based, your absorption stay consistent, and your results stay measurable.