Apr 3, 2026

You hit your goal weight. The scale finally cooperates. The appetite suppression feels like second nature. And then the question hits, the one nobody talks about until it is staring you in the face. Do you take this forever?
It is a fair question. GLP-1 receptor agonists like semaglutide and tirzepatide have changed the weight loss conversation entirely. They work. The clinical data is overwhelming. But the treatment duration question sits in a gray zone that frustrates patients and providers alike, because the honest answer is not a simple number. It depends on your goals, your biology, your willingness to change habits, and how your body responds when the medication stops.
This guide covers everything. Treatment timelines backed by clinical trials. What actually happens when you stop. Maintenance dosing strategies that let you keep results on less medication. Tapering protocols that reduce the shock of discontinuation. And the lifestyle factors that determine whether you keep the weight off or watch it creep back. SeekPeptides built this resource because the answer matters more than the question, and getting it wrong means months of progress disappearing in weeks.
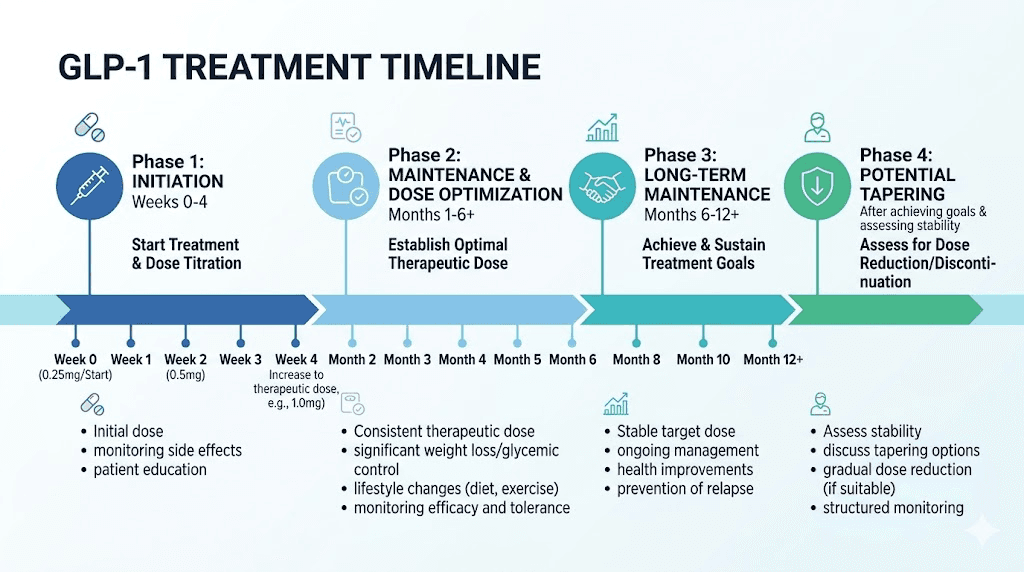
Why GLP-1 treatment duration is not a simple answer
Ask ten doctors how long you should take a GLP-1 and you will get ten different answers. That is not because they do not know. It is because the right duration depends on factors that vary wildly between patients.
Obesity is classified as a chronic disease. This matters.
When you treat hypertension, nobody asks how long they need blood pressure medication. They understand the medication manages an ongoing condition. GLP-1 therapy works the same way for many patients, managing a metabolic condition rather than curing it. The biological mechanisms that drive weight regain do not disappear because you lost weight. Hunger hormones rebound. Metabolic rate adjusts downward. The body fights to return to its previous set point with remarkable persistence.
But that does not mean everyone needs lifelong treatment. Some patients successfully transition off GLP-1 medications after reaching their goals. Others maintain results on reduced doses. And a significant group needs ongoing therapy to sustain their progress. Understanding which category you fall into requires looking at the evidence, your personal history, and your metabolic response.
The clinical trial evidence on duration
The landmark STEP trials for semaglutide and SURMOUNT trials for tirzepatide provide the clearest data on what happens at different treatment durations. In STEP-1, participants taking semaglutide 2.4mg weekly lost an average of 14.9% of body weight over 68 weeks. That is roughly 16 months of treatment. The SURMOUNT-1 trial showed tirzepatide at its highest dose produced 22.5% weight loss over 72 weeks.
Here is the critical finding. Weight loss continued throughout the entire study period. Patients had not plateaued. They were still losing at week 68. This suggests that for maximum results, treatment durations of at least 12 to 18 months produce the best outcomes. Shorter courses produce less total weight loss and may not allow enough time for metabolic adaptation.
The average American takes GLP-1 medications for about six months before discontinuing. That is often driven by cost and access rather than clinical optimization. Six months produces meaningful weight loss, typically 8 to 12%, but falls short of what longer treatment could achieve. For patients with significant weight to lose, staying on treatment longer consistently produces better outcomes.
What the guidelines actually say
Clinical guidelines vary by region and organization. The European Medicines Agency recommends evaluating treatment response at 12 weeks and continuing only if the patient achieves at least 5% body weight reduction. NICE in the UK recommends a maximum of two years for semaglutide (Wegovy), though no such limit exists for tirzepatide (Mounjaro). American guidelines generally treat obesity as a chronic condition requiring ongoing management, without specifying a maximum duration.
The practical takeaway? Most clinical guidance supports treatment for at least 12 months, with ongoing therapy recommended for patients who respond well and have no significant side effects. The decision to continue, taper, or stop should be individualized, not driven by arbitrary timelines.
What actually happens when you stop taking GLP-1
This is the section nobody wants to read. But ignoring it leads to worse outcomes than understanding it.
Weight regain after stopping GLP-1 medications is the rule, not the exception. The degree and speed of regain varies, but the pattern is consistent across clinical trials and real-world data.
Clinical trial data on weight regain
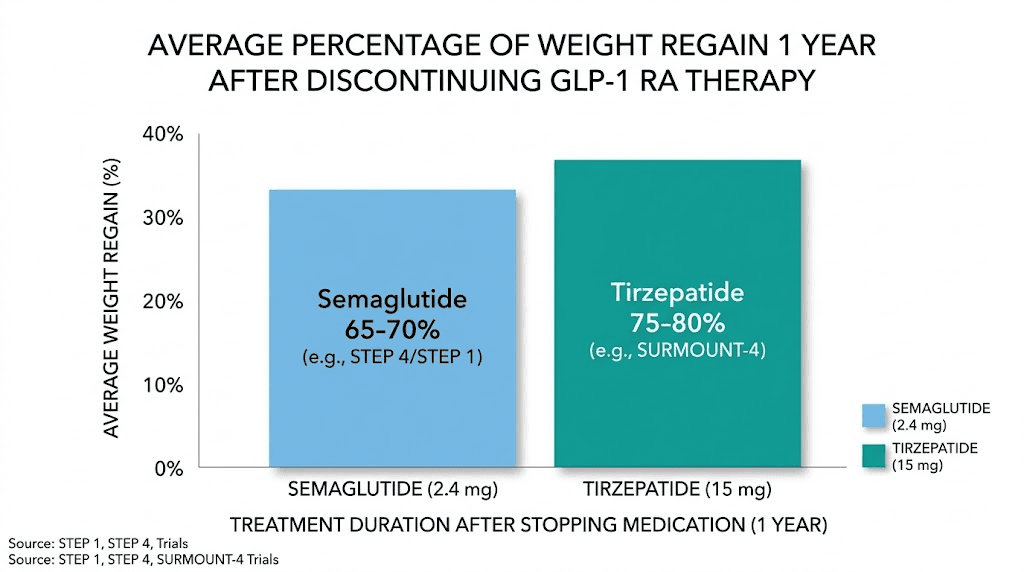
The STEP-10 extension trial revealed that participants who stopped semaglutide regained over 40% of their lost weight within just 28 weeks. That is roughly seven months to lose almost half your progress. The SURMOUNT-4 trial showed even steeper rebounds with tirzepatide, with participants regaining more than 50% of their weight loss over 52 weeks after stopping.
A systematic review and meta-analysis published in eClinicalMedicine found that discontinuing GLP-1 receptor agonist treatment led to an average weight regain of 5.63 kg and 5.81% of body weight across all patients studied. The proportion of weight regained was directly proportional to the amount originally lost. Patients who lost the most weight regained the most in absolute terms.
University of Oxford research paints an even clearer picture. On average, patients return to their pre-treatment weight within 1.7 years of stopping any weight management medication, including semaglutide and tirzepatide. That timeline gives context to the urgency of having a post-treatment plan.
Why weight comes back
Weight regain is not a willpower failure. It is biology.
GLP-1 medications work through multiple mechanisms. They slow gastric emptying, reduce appetite signals in the brain, improve insulin sensitivity, and alter food reward pathways. When you stop the medication, all of these effects reverse. Appetite rebounds, often exceeding pre-treatment levels in the short term. Gastric emptying speeds up. The brain is reward response to food intensifies. Your body essentially overcorrects in the opposite direction.
Metabolic adaptation compounds the problem. After significant weight loss, your body burns fewer calories than someone who naturally weighs the same amount. This metabolic penalty persists for years and makes maintaining weight loss without pharmacological support extraordinarily difficult. Research on contestants from The Biggest Loser showed metabolic suppression lasting at least six years after their weight loss.
Hunger hormone changes add another layer. Ghrelin, the hunger hormone, increases after weight loss. Leptin, the satiety hormone, decreases. These hormonal shifts persist long after the weight comes off and create a constant biological drive to regain. GLP-1 medications counteract this hormonal pressure. Without them, the pressure returns.
Real-world data tells a different story
Interestingly, real-world outcomes look somewhat better than clinical trial results. A Cleveland Clinic analysis of nearly 8,000 patients found that discontinuing semaglutide and tirzepatide did not lead to significant immediate weight regain in clinical practice. The key difference? Many patients later restarted the original medication or switched to an alternative obesity treatment. Real-world patients also tend to have more lifestyle support, ongoing medical relationships, and gradual dose reductions compared to the abrupt cessation in clinical trials.
An Epic Research study found that two years after stopping GLP-1 medications, most patients sustained at least some weight loss. Not all of it, but meaningful retention. The patients who maintained the most weight loss shared common traits: they had made significant dietary changes, increased physical activity, and often used a structured tapering approach rather than stopping abruptly.
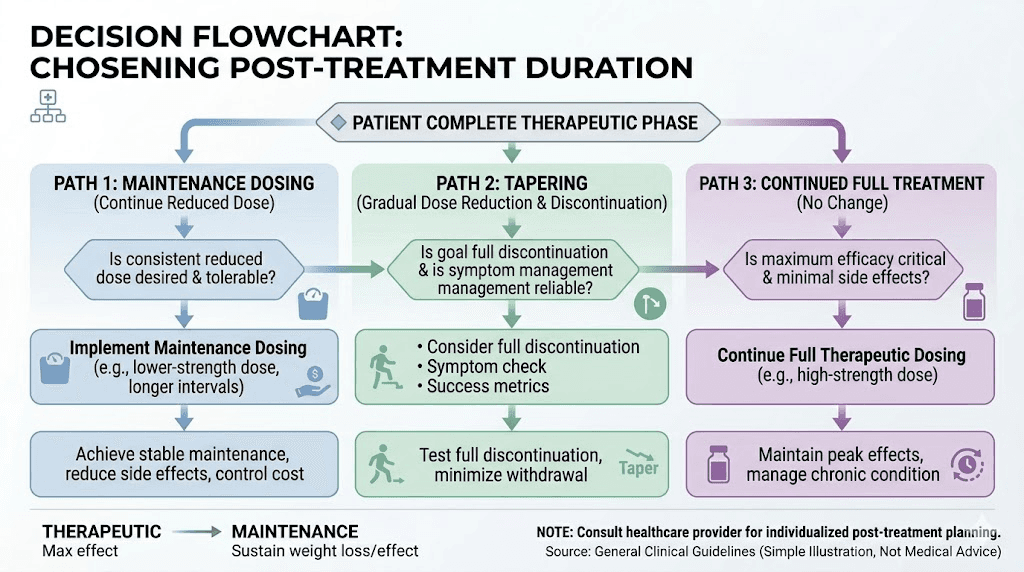
The three paths forward after reaching your goal
Once you reach your target weight or a satisfactory point, you face three options. Each has its place. None is universally right.
Path 1: Continue at maintenance dose
This is the most conservative approach and, for many patients, the most effective. Rather than stopping entirely, you reduce to a lower maintenance dose that preserves the appetite-suppressing and metabolic benefits while minimizing side effects and cost.
A patient on 2.4 mg of semaglutide weekly might reduce to 1.0 mg or even 0.5 mg for maintenance. Someone on 15 mg of tirzepatide might drop to 5 mg or 7.5 mg. The goal is finding the minimum effective dose that keeps weight stable without the full therapeutic load.
Benefits of the maintenance approach include sustained appetite regulation, continued metabolic support, fewer side effects at lower doses, and easier dose escalation if weight begins to creep. The trade-off is ongoing cost and the commitment to continued medication use. For patients with BMIs that started above 35, significant metabolic syndrome, or a history of yo-yo dieting, maintenance dosing often produces the best long-term outcomes.
The semaglutide dosage calculator can help you understand how different maintenance doses translate to actual injection volumes, particularly for compounded formulations where concentrations vary between pharmacies.
Path 2: Gradual taper and discontinuation
For patients who have made substantial lifestyle changes and want to attempt life without medication, a structured taper offers the best chance of success. The key word is structured. Abrupt discontinuation produces the worst outcomes in every study that has examined it.
A typical tapering protocol reduces the dose by 25 to 50% every four weeks, mirroring the same step-down intervals used during dose escalation. This allows appetite regulation and metabolic patterns to adjust gradually rather than experiencing the sudden rebound that comes with cold-turkey cessation.
A sample semaglutide taper from a therapeutic dose might look like this. From 2.4 mg weekly to 1.7 mg for four weeks. Then to 1.0 mg for four weeks. Then to 0.5 mg for four weeks. Then to 0.25 mg for four weeks. Then to 0.25 mg every other week for four weeks. Then discontinuation. That is a five-month taper. It feels slow. But stopping cold turkey typically produces appetite rebounds that are far harder to manage.
For tirzepatide, the approach is similar. A patient on 15 mg weekly might step down to 10 mg, then 7.5 mg, then 5 mg, then 2.5 mg, each for four weeks before the next reduction. Weaning off tirzepatide requires the same patience and monitoring as the initial dose escalation phase.
During the taper, close monitoring is essential. The first three to six months after dose reduction carry the highest risk for metabolic relapse. Follow-up visits every one to three months should track weight trends, waist circumference, metabolic markers, and subjective appetite levels. If weight begins trending upward by more than 3 to 5%, increasing the dose back one step is far easier than trying to reverse a 15-pound regain.
Path 3: Cycling on and off
Some patients adopt a cycling approach, using GLP-1 medications for defined periods, taking breaks, and resuming if weight begins to climb. This is not well-studied in clinical trials, but real-world data suggests it is common. The Cleveland Clinic analysis found that many patients who stopped GLP-1 therapy later restarted, sometimes with the same medication, sometimes switching to an alternative.
Cycling has practical advantages. It reduces total medication exposure and cost. It allows patients to test their ability to maintain weight independently. And it provides psychological breaks from injectable medication routines. The disadvantage is the metabolic disruption of stopping and starting, which may reduce the efficiency of subsequent treatment courses. Some patients report needing longer to achieve the same appetite suppression when restarting after a break.
If you choose cycling, plan for three to six months of active treatment followed by monitored breaks of similar duration. Keep all the habits you built during treatment, because they are the only thing standing between you and regain during the off periods.
Treatment duration by specific GLP-1 medication
Not all GLP-1 medications behave identically, and duration considerations vary by compound.
Semaglutide (Wegovy, Ozempic, compounded)
Semaglutide has the longest treatment duration data among current GLP-1 options. The STEP trials provide 68-week data, and extension studies push that to three to four years. Key findings for semaglutide duration include continued weight loss through at least 68 weeks with no plateau in many patients, weight regain of approximately 40% of lost weight within seven months of stopping, and withdrawal effects including increased appetite, food cravings, and in some cases gastrointestinal changes.
For compounded semaglutide specifically, treatment duration considerations are similar, but access and cost dynamics differ. Compounded formulations often cost significantly less than brand-name versions, making longer treatment courses more financially feasible for many patients. This accessibility factor often translates to longer treatment durations in practice, which may partly explain why week-by-week results in real-world settings sometimes exceed clinical trial averages.
Initial response to semaglutide typically begins within two to four weeks, with meaningful weight loss appearing by weeks four to eight. Most patients reach their maximum tolerated dose by week 16. From that point, the question of duration becomes one of goal achievement and maintenance strategy.
Tirzepatide (Mounjaro, Zepbound, compounded)
Tirzepatide is a dual GIP/GLP-1 receptor agonist, which means its mechanism differs slightly from pure GLP-1 medications. The SURMOUNT trials show consistently greater weight loss with tirzepatide compared to semaglutide, but also steeper regain after discontinuation. In SURMOUNT-4, participants who switched from tirzepatide to placebo regained more than 50% of their lost weight over 52 weeks.
This steeper regain may reflect tirzepatide is more potent metabolic effects. When those effects are removed, the metabolic rebound is proportionally larger. For tirzepatide users, this means the case for longer treatment duration or maintenance dosing is potentially even stronger than for semaglutide.
Tirzepatide weight loss timelines show continued progress through at least 72 weeks. Many patients lose 20% or more of their starting body weight, which represents a transformative result. Protecting that investment with appropriate treatment duration planning makes the extended course worthwhile for most patients.
Dose escalation for tirzepatide follows a specific schedule moving from 2.5 mg through 5 mg, 7.5 mg, 10 mg, 12.5 mg, and up to 15 mg weekly. Most patients reach their target dose by week 20. The dosing guide in units helps translate these milligram doses into practical injection volumes.
Retatrutide (triple agonist, investigational)
Retatrutide is a triple hormone receptor agonist targeting GLP-1, GIP, and glucagon receptors. Phase 2 trial data shows weight loss of up to 24.2% at 48 weeks, the highest of any obesity medication studied to date. While retatrutide is still investigational, its treatment duration implications are worth noting.
The triple mechanism suggests that discontinuation effects could be even more pronounced than with tirzepatide. The glucagon receptor component adds metabolic effects beyond appetite suppression, including enhanced fat oxidation and energy expenditure. When those effects stop, the metabolic adjustment could be substantial. Retatrutide dosing protocols are still being refined, but early data supports extended treatment courses with careful dose optimization.
For patients currently using retatrutide through research channels, transitioning from other GLP-1 medications requires careful dose mapping and expectation management regarding treatment duration.
Factors that determine YOUR ideal treatment duration
Generic timelines help set expectations, but your personal duration depends on specific, measurable factors. Understanding them turns the vague question of how long into a concrete, personalized plan.
Starting BMI and total weight loss goal
Patients starting at higher BMIs generally need longer treatment courses. Someone with a BMI of 40 who needs to lose 80 pounds will need more time than someone at BMI 32 targeting 30 pounds. The dose escalation phase alone takes 16 to 20 weeks for most GLP-1 medications, and meaningful weight loss really begins at therapeutic doses.
As a rough framework, expect one to two pounds of weight loss per week once you reach your target dose. For a 50-pound goal, that translates to 25 to 50 weeks of treatment at therapeutic dose, plus the escalation period. Adding maintenance time pushes total treatment to 12 to 18 months minimum for significant weight loss goals.
Patients with higher starting BMIs also tend to experience more pronounced metabolic adaptation after weight loss, making maintenance dosing or longer treatment courses more important for sustained results.
Metabolic health markers
GLP-1 medications do more than reduce weight. They improve insulin sensitivity, lower HbA1c, reduce blood pressure, and improve lipid profiles. For patients with type 2 diabetes or metabolic syndrome, these metabolic benefits may justify continued treatment even after weight loss goals are met.
Monitoring these markers during and after treatment helps determine readiness for dose reduction. If your metabolic markers remain stable on a lower dose, that supports tapering. If they begin deteriorating as the dose decreases, that signals your body needs ongoing pharmacological support. Your provider should track fasting glucose, HbA1c, blood pressure, lipid panels, and inflammatory markers throughout the treatment course.
Lifestyle changes made during treatment
This is the single most important predictor of success after stopping GLP-1 medications. Patients who use the appetite suppression window to build sustainable dietary habits, establish regular exercise routines, and address the psychological drivers of overeating have dramatically better outcomes when medication stops or decreases.
Think of GLP-1 therapy as a bridge. The medication reduces the biological barriers to weight loss while you build the behavioral and lifestyle foundation that will support your new weight long-term. Patients who rely solely on the medication without changing habits are nearly guaranteed to regain when they stop. Those who use the treatment period strategically have a fighting chance.
Key lifestyle investments during treatment include developing a sustainable eating pattern that does not rely on appetite suppression, building strength training habits that preserve and build lean mass, addressing emotional eating patterns through therapy or structured programs, establishing sleep hygiene that supports metabolic health, and managing stress through sustainable practices.
Previous weight loss history
Your track record matters. Patients with a history of yo-yo dieting, where they repeatedly lose and regain significant weight, tend to experience more aggressive regain after stopping any weight loss intervention. Each cycle of loss and regain may worsen metabolic adaptation, making subsequent weight loss harder and regain faster.
If you have a history of weight cycling, consider this when planning your GLP-1 treatment duration. You may benefit from a longer initial treatment course, a more gradual taper, or a commitment to maintenance dosing rather than complete discontinuation. The cost of ongoing medication may be significantly less than the physical and psychological cost of another regain cycle.
Age and hormonal factors
Metabolic rate declines naturally with age, and hormonal changes during perimenopause, menopause, and andropause compound the challenge. Older patients and those experiencing hormonal transitions may need longer treatment courses or ongoing maintenance therapy because their underlying metabolic environment is less forgiving of medication withdrawal.
Women navigating menopause while on GLP-1 therapy face a dual challenge, as hormonal changes independently promote weight gain. Hormonal effects of GLP-1 medications add another consideration to the treatment duration discussion. Coordinating with endocrinology or gynecology can help optimize the overall approach.
The maintenance dose strategy explained
Maintenance dosing deserves its own deep dive because it represents the middle ground that works for the largest number of patients. Not stopping completely. Not staying at full therapeutic dose forever. Finding the sweet spot.
How maintenance dosing works
The concept is straightforward. After reaching your weight loss goal on a therapeutic dose, you reduce to the lowest dose that maintains your results. This reduces side effects, lowers cost, and decreases total medication exposure while preserving the metabolic and appetite benefits that keep weight stable.
For semaglutide, common maintenance doses range from 0.25 mg to 1.0 mg weekly, down from the therapeutic 1.7 to 2.4 mg. For tirzepatide, maintenance doses typically fall between 2.5 mg and 7.5 mg, down from the therapeutic 10 to 15 mg. The exact dose depends on individual response, and finding it requires careful downward titration with monitoring.
The complete maintenance dose guide covers the specifics of dose reduction strategies, monitoring protocols, and adjustment criteria. The key principle is to reduce slowly and monitor continuously, treating any upward weight trend as a signal to pause the reduction or increase one step.
Finding your minimum effective dose
Start by reducing your therapeutic dose by one step (typically 25 to 30%) and maintaining that level for four to six weeks. Monitor weight weekly. If weight remains stable, reduce another step and repeat. Continue until either your weight begins to creep up or you reach the lowest available dose.
When weight begins to increase, step back up one dose level. That is likely your minimum effective maintenance dose. Some patients find their maintenance dose is only one step below their therapeutic dose. Others maintain successfully on the lowest available dose. There is no way to predict without testing.
During this process, track more than just weight. Monitor how your appetite feels, how your energy levels respond, whether fatigue patterns change, and how your relationship with food shifts. These subjective markers often predict weight trends before the scale reflects them.
Maintenance dose vs. full discontinuation outcomes
The European Congress on Obesity presented data suggesting that maintenance dosing produces significantly better long-term outcomes than complete discontinuation. Patients on maintenance doses retained 70 to 85% of their peak weight loss at two years, compared to 40 to 60% retention for those who stopped completely but implemented lifestyle changes, and less than 30% for those who stopped without significant lifestyle modification.
The cost calculation often favors maintenance dosing as well. A maintenance dose of compounded semaglutide at 0.5 mg weekly costs roughly half to one-third of a therapeutic dose. When weighed against the financial and health costs of weight regain, including renewed medication courses, worsening metabolic disease, and associated healthcare utilization, the maintenance approach frequently wins on pure economics.
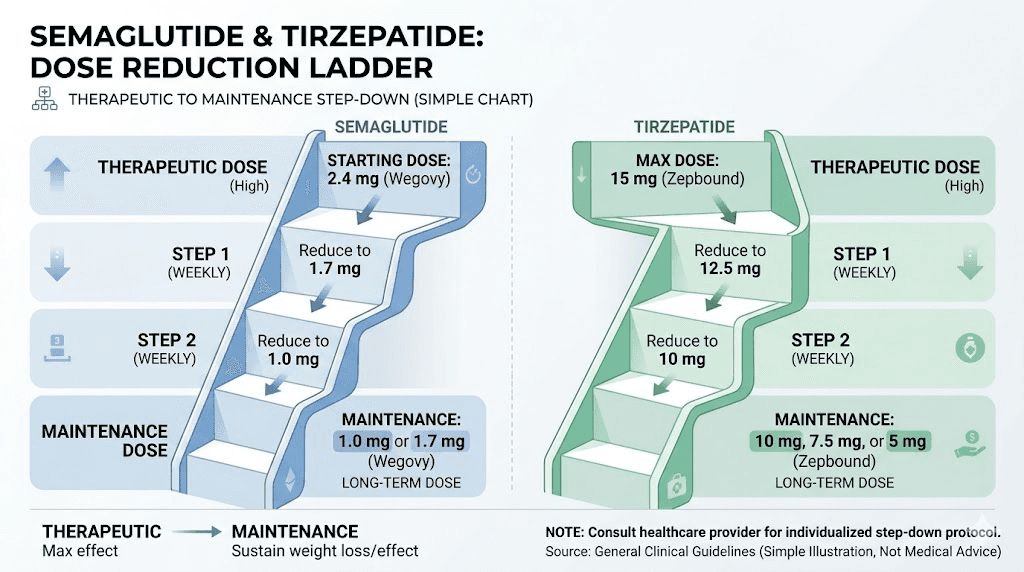
The tapering protocol: step by step
If you and your provider decide that discontinuation is the goal, doing it right dramatically improves your chances of maintaining results. A structured taper is not optional. It is the difference between managed transition and metabolic chaos.
Semaglutide tapering schedule
The following schedule applies to patients coming off a therapeutic dose of 2.4 mg weekly. Adjust proportionally if your therapeutic dose is lower.
Weeks 1 to 4: Reduce from 2.4 mg to 1.7 mg weekly. Most patients notice minimal change in appetite or weight at this step. Converting this to injection units depends on your vial concentration, so verify with your pharmacy or use the dosage calculator.
Weeks 5 to 8: Reduce from 1.7 mg to 1.0 mg weekly. This is where many patients first notice appetite returning. Increase protein intake to compensate. Focus on adequate protein consumption to preserve lean mass and maintain satiety.
Weeks 9 to 12: Reduce from 1.0 mg to 0.5 mg weekly. Appetite suppression diminishes noticeably. Food reward responses strengthen. This is the critical period where lifestyle habits either carry you or fail you. Double down on nutrient-dense food choices and meal timing strategies.
Weeks 13 to 16: Reduce from 0.5 mg to 0.25 mg weekly. Many patients describe this as feeling like the training wheels are coming off. Hunger is present but manageable if habits are strong. Monitor weight closely.
Weeks 17 to 20: Take 0.25 mg every other week. This extended interval tests your ability to function without frequent GLP-1 support while maintaining a safety net.
Week 21+: Discontinuation. Continue weekly weigh-ins and monthly metabolic marker checks for at least six months. If weight increases by more than 5% from your lowest point, consider restarting at a low maintenance dose rather than waiting for larger regain.
Tirzepatide tapering schedule
Tirzepatide tapering follows the same principles but uses different dose steps. Starting from a therapeutic dose of 15 mg weekly, the schedule uses the same four-week intervals between each dose reduction.
Weeks 1 to 4: 15 mg to 12.5 mg. Weeks 5 to 8: 12.5 mg to 10 mg. Weeks 9 to 12: 10 mg to 7.5 mg. Weeks 13 to 16: 7.5 mg to 5 mg. Weeks 17 to 20: 5 mg to 2.5 mg weekly. Weeks 21 to 24: 2.5 mg every other week. Week 25+: Discontinuation with close monitoring.
The tirzepatide unit conversion changes at each dose step, so recalculate your injection volume at each reduction. Errors in unit conversion during tapering can lead to inadvertent dose changes that confuse the tapering process.
Some patients find that weaning off tirzepatide is harder than semaglutide due to the dual receptor mechanism. If appetite rebounds significantly at any step, pause the taper for an additional four weeks before continuing. There is no penalty for going slower.
When to pause or reverse the taper
Red flags that should pause your taper include weight gain exceeding 3% of your lowest weight, significant increase in hunger that interferes with daily functioning, worsening metabolic markers such as rising blood sugar, blood pressure, or lipid levels, return of binge eating patterns or loss-of-control eating episodes, and significant emotional distress related to appetite changes.
If any of these occur, step back up one dose level and stabilize for four to eight weeks before attempting the reduction again. If the same step fails twice, that dose level may be your minimum effective dose, and continuing the taper may not be appropriate. This is not failure. This is data. Your body is telling you what it needs.
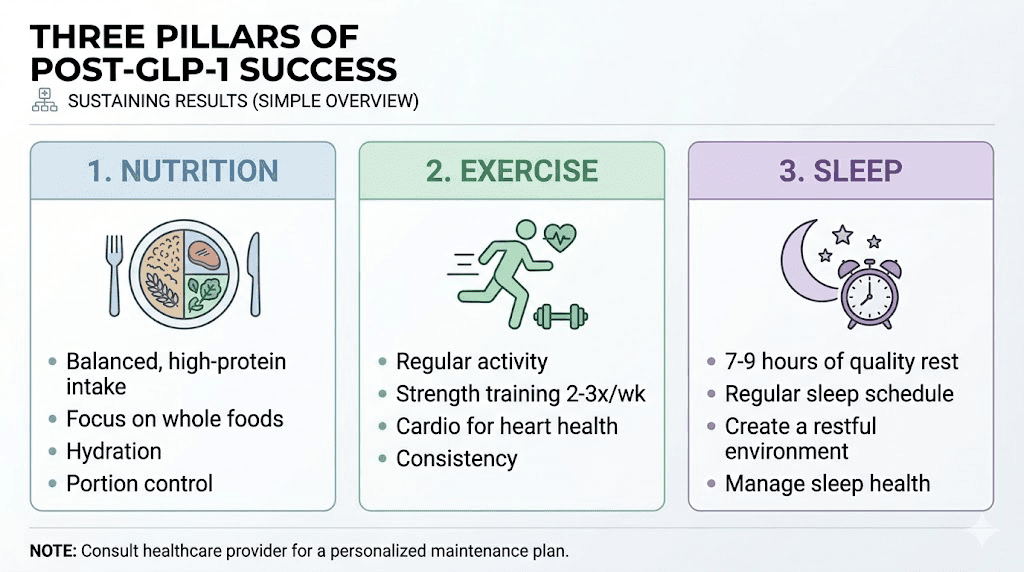
Building the lifestyle foundation during treatment
The single most important thing you can do during GLP-1 treatment is prepare for life after it. Whether you plan to stay on a maintenance dose, taper off completely, or cycle on and off, the habits you build during treatment determine your long-term outcome.
Nutrition strategies that outlast the medication
While on GLP-1 medications, you have a unique advantage. Reduced appetite makes it easier to make better food choices without fighting constant hunger. Use this window to establish eating patterns you can sustain when appetite normalizes.
Protein becomes even more important during and after GLP-1 treatment. The rapid weight loss associated with these medications can include significant lean mass loss if protein intake is inadequate. Aim for 1.0 to 1.2 grams of protein per pound of goal body weight daily. This preserves muscle, supports metabolic rate, and enhances satiety through protein is thermic effect. Our guides on GLP-1 friendly meals and breakfast ideas focus on practical, protein-forward meal strategies.
Meal timing and structure matter. Establish regular eating patterns with three meals and planned snacks rather than grazing. The reduced appetite from GLP-1 medications can lead to meal skipping, which undermines the habit-building process. Even if you are not hungry, eating structured meals teaches your body and brain the patterns that will support you post-treatment.
Foods to avoid during treatment remain important after treatment. Ultra-processed foods, excessive added sugars, and calorie-dense beverages undermine weight maintenance regardless of medication status. Building aversion to these foods during the appetite-suppressed period creates neural pathways that persist after medication stops.
Exercise: the metabolic insurance policy
Regular exercise, particularly resistance training, is the single most effective strategy for mitigating metabolic adaptation after weight loss. Strength training preserves and builds lean mass, which directly supports metabolic rate. Patients who maintain or build muscle during GLP-1 treatment experience less metabolic slowdown and better weight maintenance after stopping.
The question of building muscle on GLP-1 is important because the caloric deficit created by these medications can work against muscle growth. The solution is prioritizing protein intake, following a progressive resistance training program, and potentially adjusting your caloric deficit to preserve lean mass. Creatine supplementation may support muscle preservation during GLP-1 therapy, with evidence suggesting modest benefits for strength and lean mass retention.
Cardiovascular exercise provides additional metabolic and cardiovascular benefits, but should complement rather than replace resistance training. A balanced program might include three to four days of resistance training and two to three days of moderate cardiovascular activity weekly.
Sleep and stress management
Poor sleep and chronic stress both promote weight regain through hormonal mechanisms. Sleep deprivation increases ghrelin (hunger hormone) and decreases leptin (satiety hormone), mimicking the exact hormonal environment that drives post-medication weight regain. Chronic stress elevates cortisol, which promotes visceral fat storage and undermines insulin sensitivity.
During GLP-1 treatment, some patients experience sleep disruption or insomnia as side effects. Addressing these proactively improves both treatment outcomes and post-treatment maintenance. Prioritize seven to nine hours of sleep, maintain consistent sleep and wake times, limit caffeine after noon, and create a sleep environment that supports quality rest.
The supplement stack for transition
Certain supplements can support the transition off GLP-1 medications. Fiber supplements increase satiety and support digestive health as gastric emptying speeds up post-medication. Probiotics may help with the gastrointestinal adjustment that occurs when stopping GLP-1 therapy. Electrolytes support hydration and metabolic function, particularly important if dietary changes accompany the medication transition. Electrolyte supplementation can help manage the transition period.
These supplements support the process but do not replace the medication is core effects. No supplement replicates the appetite suppression and metabolic benefits of GLP-1 receptor agonists. They are adjuncts, not replacements.
Common treatment duration mistakes
Understanding what not to do is sometimes more valuable than knowing what to do. These patterns consistently lead to poor outcomes.
Stopping too early
The most common mistake. Patients feel good after three to four months, see meaningful weight loss, and decide they no longer need the medication. At that point, metabolic adaptation is just beginning, lifestyle habits are still forming, and the biological drivers of regain are at their strongest. Early treatment challenges sometimes cause patients to stop prematurely because they mistake slow initial progress for treatment failure.
Minimum treatment duration for meaningful, sustainable results should be 12 months in most cases. Stopping at six months leaves significant weight loss potential unrealized and insufficient time for metabolic and behavioral adaptation.
Stopping abruptly
Running out of medication and not refilling. Losing insurance coverage and stopping overnight. Deciding on Monday that Friday is your last dose. All of these produce the worst possible outcomes. Abrupt cessation creates a hormonal whiplash that makes weight regain almost inevitable.
Stopping tirzepatide cold turkey and stopping semaglutide abruptly both carry significant risks including appetite rebound, gastrointestinal changes, mood shifts, and rapid weight regain. If cost or access issues force a stop, even a partial taper over two to three weeks is better than nothing.
Not making lifestyle changes during treatment
Using GLP-1 medications as the sole weight loss strategy without modifying diet, exercise, sleep, or stress management is like building a house without a foundation. It looks great while the scaffolding (medication) is up. Remove the scaffolding, and everything collapses.
Every month on GLP-1 therapy should include deliberate lifestyle investment. Track your caloric intake, build exercise habits, address emotional eating, and develop stress management practices. These are not optional add-ons. They are the primary investment that determines whether your GLP-1 treatment produces lasting results or temporary ones.
Ignoring metabolic markers
Weight is one data point. Metabolic health is the full picture. Patients who monitor only weight and ignore blood sugar, blood pressure, lipid levels, and inflammatory markers miss important signals about when to continue, taper, or adjust treatment.
Request comprehensive metabolic panels every three to six months during treatment and every one to three months during and after tapering. Worsening metabolic markers during dose reduction are a clear signal that your body needs more pharmacological support, regardless of what the scale says.
Special considerations for different populations
Treatment duration recommendations vary based on patient characteristics that influence metabolic response and risk.
Patients with type 2 diabetes
For patients using GLP-1 medications for both weight loss and glycemic control, treatment duration extends to address both conditions. Diabetes is a chronic disease requiring ongoing management. Stopping GLP-1 therapy in diabetic patients risks not only weight regain but also deterioration of blood sugar control, potentially requiring additional diabetes medications.
In these patients, maintenance dosing is strongly preferred over complete discontinuation. The metabolic benefits of GLP-1 therapy extend well beyond weight loss, including beta-cell preservation, improved insulin sensitivity, and cardiovascular risk reduction. The case for indefinite treatment is strongest in this population.
Patients with PCOS
Polycystic ovary syndrome creates metabolic challenges that make weight management particularly difficult. Insulin resistance, hormonal imbalances, and inflammatory pathways all contribute to weight gain and resist weight loss. GLP-1 medications address several of these pathways simultaneously, and stopping treatment may allow the underlying PCOS-driven metabolic dysfunction to reassert itself.
Longer treatment courses and maintenance dosing are generally appropriate for PCOS patients. Coordinating with endocrinology to address the full spectrum of PCOS symptoms while using GLP-1 therapy produces better outcomes than treating weight in isolation.
Patients planning pregnancy
GLP-1 medications must be discontinued before conception. Current guidelines recommend stopping semaglutide at least two months before planned conception and tirzepatide at least one month before. This creates a specific treatment duration consideration for patients of reproductive age.
The strategy for these patients involves achieving target weight, stabilizing on a maintenance dose, then implementing a structured taper timed to allow the recommended washout period before conception. Planning pregnancy while on semaglutide and understanding the implications of tirzepatide and pregnancy are critical for this population.
Patients over 65
Older adults face unique considerations. Sarcopenia (age-related muscle loss) makes lean mass preservation during GLP-1 therapy even more critical. The weight loss from these medications includes both fat and muscle, and older patients have less metabolic reserve to recover lost muscle mass.
Treatment duration in older patients should balance weight loss benefits against the risk of excessive lean mass depletion. More moderate dose escalation, higher protein targets, mandatory resistance training, and potentially shorter treatment courses with emphasis on maintenance dosing may be appropriate. Fall risk assessment should be included in the monitoring protocol, as rapid weight loss can affect balance and bone density.
Patients with autoimmune conditions
Emerging research suggests GLP-1 receptor agonists may have anti-inflammatory and immunomodulatory effects beyond their metabolic actions. Patients with conditions like lipedema, Hashimoto is thyroiditis, or endometriosis may experience benefits that extend beyond weight management. For these patients, treatment duration decisions should account for the broader therapeutic effects and the potential for symptom return upon discontinuation.
Microdosing GLP-1 for autoimmune conditions is an emerging area of interest, with some practitioners maintaining patients on very low doses primarily for anti-inflammatory benefits rather than weight management.
Monitoring your progress and making the right call
The decision to continue, reduce, or stop GLP-1 therapy should be driven by data, not arbitrary timelines or gut feelings. A structured monitoring approach transforms the treatment duration question from anxiety-inducing uncertainty into evidence-based decision-making.
Weekly tracking during active treatment
Weigh yourself at the same time each day, preferably morning before eating. Weekly averages matter more than daily fluctuations. Track your weight trend over four-week periods to identify genuine progress versus normal variation. Plotting your weight on a graph makes patterns visible that raw numbers obscure.
Monitor appetite on a simple 1-to-10 scale daily. This subjective measure correlates surprisingly well with medication effectiveness and helps identify when dose adjustments might be needed. If appetite consistently rates below 3, you may be able to reduce your dose. If it climbs above 6 during tapering, that signals your body is resisting the reduction.
Tracking your injections systematically helps maintain consistency and provides data for your provider. Note the date, dose, injection site, and any side effects for each administration.
Quarterly metabolic assessments
Every three months, request a comprehensive metabolic panel including fasting glucose, HbA1c, fasting insulin, complete lipid panel including triglycerides, liver enzymes, kidney function markers, inflammatory markers such as CRP and ESR, and thyroid function. These numbers tell the metabolic story that weight alone cannot.
Compare each quarterly panel to your baseline values and previous results. Improving markers support continued treatment at current doses. Stable markers during dose reduction support continued tapering. Worsening markers at any point should prompt reconsideration of the current approach.
Body composition beyond the scale
Weight does not distinguish between fat loss and muscle loss. During GLP-1 therapy, monitoring body composition provides crucial information about the quality of your weight loss. DEXA scans provide the most accurate body composition data and should be performed at baseline, at peak weight loss, and during any tapering protocol.
If DEXA scans are not accessible, waist circumference and body measurements provide useful proxies. Decreasing waist circumference with stable or increasing limb measurements suggests favorable fat loss with lean mass preservation. The opposite pattern, losing limb circumference while waist remains stable, suggests muscle loss that requires intervention.
The financial equation of treatment duration
Cost influences treatment duration decisions more than most medical discussions acknowledge. Being realistic about the financial dimension helps you plan treatment that is both clinically optimal and financially sustainable.
Brand-name vs. compounded cost comparison
Brand-name semaglutide (Wegovy) costs approximately $1,000 to $1,350 per month without insurance. Brand-name tirzepatide (Zepbound) runs similarly. Compounded versions of both medications cost significantly less, often $100 to $400 per month depending on the pharmacy and dose.
This cost differential has enormous implications for treatment duration. A 12-month course of brand-name semaglutide costs $12,000 to $16,000 out of pocket. The same duration with compounded semaglutide might cost $1,200 to $4,800. For many patients, the compounded route makes clinically appropriate treatment durations financially feasible where brand-name versions would not.
Insurance coverage remains inconsistent and unpredictable. Patients whose coverage changes mid-treatment face difficult decisions about continuing at higher out-of-pocket cost, switching to compounded versions, or stopping prematurely. Planning for potential coverage disruptions by identifying compounded alternatives in advance prevents forced abrupt discontinuation.
The cost of not treating long enough
Weight regain carries its own financial burden. Restarting medication after regain means paying for another full course of treatment, including the dose escalation phase where medication cost accrues without maximum benefit. Worsening metabolic disease from regain increases healthcare utilization, medication costs for diabetes and cardiovascular conditions, and potential surgical interventions.
A conservative estimate suggests that a 50-pound weight regain costs $5,000 to $15,000 in direct healthcare costs over the following five years through increased medications, more frequent provider visits, and management of weight-related complications. Spending $2,000 to $4,000 on a maintenance dose to prevent that regain represents a clear return on investment.
Frequently asked questions
Can you take GLP-1 medications forever?
Yes. Current evidence supports indefinite use for patients who tolerate the medication well and continue to benefit from it. Obesity is a chronic condition, and long-term pharmacological management is appropriate for many patients. Long-term safety data extends to four or more years for semaglutide and continues to accumulate for tirzepatide.
What is the shortest effective treatment duration?
Six months represents the minimum for meaningful results, but 12 months or longer produces significantly better outcomes. Patients who stop before 12 months typically achieve less total weight loss and experience faster regain than those who continue longer. The week-by-week results show that weight loss continues well past the six-month mark for most patients.
Will I regain all the weight if I stop?
Not necessarily all of it, but most patients regain a significant portion without lifestyle changes and ongoing monitoring. Patients who implement structured maintenance strategies, including gradual tapering, exercise, dietary habits, and monitoring, retain more of their weight loss than those who stop abruptly.
Is a maintenance dose the same as a therapeutic dose?
No. A maintenance dose is typically 25 to 50% of the therapeutic dose. The GLP-1 maintenance dose guide explains how to find your minimum effective dose for weight maintenance.
Can I switch between GLP-1 medications during treatment?
Yes. Patients who experience side effects, plateaus, or inadequate response on one medication can switch to another. Switching between tirzepatide and semaglutide requires dose mapping using conversion charts, but the transition can be managed smoothly with provider guidance.
Do GLP-1 medications lose effectiveness over time?
Weight loss typically plateaus after 12 to 18 months, but this reflects reaching a new metabolic equilibrium rather than medication failure. The appetite-suppressing and metabolic benefits continue at plateau. Some patients experience plateaus earlier, which may respond to dose adjustments or medication switches.
Should I increase my dose if I plateau?
Not necessarily. A plateau at a given dose may represent your body is new set point at that medication level. Increasing the dose may produce additional weight loss, but it also increases side effects and cost. Discuss with your provider whether the additional weight loss justifies the higher dose, or whether maintaining at the plateau weight with the current dose is the more appropriate goal.
What happens to my metabolism after stopping GLP-1 medications?
Metabolic rate decreases after weight loss, a phenomenon called metabolic adaptation. This adaptation persists after stopping GLP-1 medications and contributes to weight regain. Resistance training and adequate protein intake are the most effective strategies for minimizing metabolic adaptation.
External resources
PubMed - Weight regain after GLP-1 RA interruption: narrative review
eClinicalMedicine - Metabolic rebound after GLP-1 RA discontinuation: meta-analysis
For researchers navigating the complexities of GLP-1 treatment duration, SeekPeptides provides comprehensive protocol guides, dosing calculators, and evidence-based resources designed to help you make informed decisions about your treatment timeline. Members access detailed tapering protocols, maintenance dosing strategies, and a community of experienced users who have navigated these exact decisions.
In case I do not see you, good afternoon, good evening, and good night. May your treatment timeline stay optimized, your maintenance dose stay effective, and your results stay permanent.