Mar 10, 2026

If you took a GLP-1 medication and something went wrong, seriously wrong, you are not alone. More than 3,300 people have filed lawsuits. The number grows every month. And the legal machinery processing these claims is unlike anything most people have ever encountered.
Finding the right attorney matters more than most realize. This is not a simple slip-and-fall case. GLP-1 litigation involves complex pharmaceutical science, multidistrict litigation procedures, and evolving medical evidence that changes the landscape of every pending claim. The wrong attorney means missed deadlines, undervalued injuries, and years of frustration. The right one means someone who understands the current state of GLP-1 lawsuits, knows the science behind semaglutide and tirzepatide side effects, and has the resources to take on pharmaceutical giants like Novo Nordisk and Eli Lilly.
This guide covers everything. What the lawsuits are actually about. Which injuries qualify. How the MDL process works. What to look for in a GLP-1 attorney. What kind of compensation people are pursuing. And the critical steps you need to take right now to protect your claim, even if you have not decided whether to file.
Whether you were prescribed Ozempic, Wegovy, Mounjaro, Rybelsus, or another GLP-1 receptor agonist for weight loss or diabetes management, understanding your legal options starts here. SeekPeptides has tracked this litigation from the beginning, and the information below reflects the most current data available.
What the GLP-1 lawsuits are actually about
The GLP-1 lawsuits center on one core allegation. Pharmaceutical companies knew their drugs could cause serious, life-altering injuries, and they failed to adequately warn patients and doctors about those risks.
That is the legal foundation. Failure to warn.
But the specifics matter enormously. The litigation targets GLP-1 receptor agonist medications, a class of drugs originally developed for type 2 diabetes that became massively popular for weight loss. The defendants include Novo Nordisk, which manufactures Ozempic, Wegovy, and Rybelsus (all containing semaglutide), and Eli Lilly, which manufactures Mounjaro (containing tirzepatide). These companies generated tens of billions in revenue from these medications while allegedly downplaying severe adverse effects that went far beyond the bloating, constipation, and nausea listed on standard warning labels.
The injuries at the center of these lawsuits are not minor inconveniences. They are severe, sometimes permanent, sometimes fatal. And the evidence connecting them to GLP-1 medications has grown substantially over the past two years.
The two separate MDLs
The federal courts have organized GLP-1 lawsuits into two separate multidistrict litigations (MDLs), both in the Eastern District of Pennsylvania under Judge Karen Spencer Marston.
MDL 3094 handles gastrointestinal injury claims. This is the larger of the two, covering gastroparesis (stomach paralysis), ileus (bowel obstruction), and related severe GI injuries. Of the lawsuits filed in this MDL, 75% list gastroparesis in the complaint. Another 18% cite ileus. Eight percent involve gallbladder complications. And 8% describe other severe gastrointestinal conditions. Some plaintiffs list multiple injuries, which is why the percentages exceed 100%.
The second MDL covers vision loss claims. Specifically, non-arteritic anterior ischemic optic neuropathy, known as NAION. This is a condition where blood flow to the optic nerve is suddenly reduced or cut off, causing permanent vision loss. The connection between GLP-1 medications and vision problems emerged more recently, but the clinical evidence is striking. A study published in JAMA Ophthalmology found that patients prescribed semaglutide experienced NAION at a rate over seven times higher than those on other medications. Research from Harvard found that non-diabetic users, people taking the drug purely for weight loss, faced the highest risk.
As of March 2026, more than 3,363 lawsuits have been filed across both MDLs. Legal analysts estimate total liability could exceed 2 billion dollars.
Why failure to warn is the central claim
The lawsuits do not argue that GLP-1 medications should never have been approved. They argue that the manufacturers knew about serious risks and either concealed them or minimized them in their labeling and marketing materials.
Consider this timeline. For years, semaglutide labels mentioned nausea, vomiting, and diarrhea as common side effects. But gastroparesis, a condition where the stomach loses its ability to empty properly, was not prominently warned about. It was not until January 2025 that the Ozempic label was updated to state that the drug is "not recommended in patients with severe gastroparesis." Even then, the label does not state that the drug itself may cause the condition.
Plaintiffs argue this is insufficient. They claim internal company data showed the risk of severe gastroparesis was known much earlier, and that the manufacturers had a legal obligation to warn both doctors and patients more clearly and more urgently. Every week that passed without adequate warnings meant more patients exposed to serious harm without informed consent.
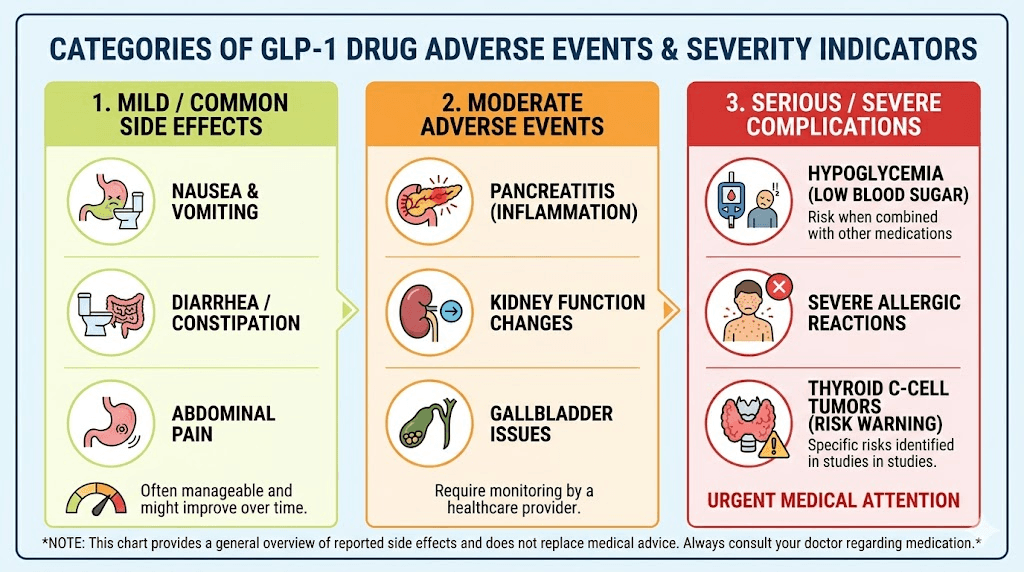
Injuries that qualify for a GLP-1 lawsuit
Not every side effect qualifies for legal action. The litigation focuses on severe, documented injuries that required medical treatment, hospitalization, or resulted in permanent harm. Understanding which injuries are currently being accepted by attorneys is critical before you begin your search for legal representation.
Gastroparesis and severe gastrointestinal injuries
Gastroparesis is the signature injury in GLP-1 litigation. It accounts for three-quarters of all filed claims, and for good reason. GLP-1 receptor agonists work partly by slowing gastric emptying, which helps people feel fuller longer and reduces blood sugar spikes after meals. That mechanism is central to both the therapeutic benefit and the potential harm. In some individuals, the delayed gastric emptying becomes extreme. The stomach essentially stops functioning normally.
Symptoms of gastroparesis include persistent nausea and vomiting that does not resolve, severe abdominal pain and bloating, inability to eat or keep food down, significant weight loss from malnutrition, and in the worst cases, the need for hospitalization, feeding tubes, or surgical intervention. If you experienced severe bloating on tirzepatide or semaglutide bloating that progressed to something far worse, this is the category your claim likely falls into.
Related GI injuries that qualify include ileus (impaired bowel function where the intestines stop moving food through), intestinal obstruction, and severe gallbladder complications including the need for gallbladder removal after tirzepatide use.
Vision loss (NAION)
The vision loss claims represent a newer but rapidly growing segment of GLP-1 litigation. NAION occurs when the optic nerve does not receive enough blood, causing sudden, painless vision loss in one eye. It can progress to both eyes. The damage is often permanent.
The clinical evidence linking GLP-1 drugs to NAION is significant. A Danish and Norwegian cohort study of over 424,000 patients found that semaglutide use more than doubled the risk of NAION compared to patients taking other diabetes medications. Harvard researchers found diabetic users were four times more likely to develop the condition, while non-diabetic users faced a sevenfold increased risk. A meta-analysis of clinical trials, including data from Novo Nordisk itself, found higher incidence of optic ischemic neuropathy in GLP-1 groups compared to placebo.
In August 2024, the European Medicines Agency required updated warning labels for semaglutide drugs to include NAION risk. Several ophthalmologists and public health advocates are now urging the FDA to require a black box warning, the most serious type of drug safety warning available.
Other qualifying injuries
Beyond gastroparesis and NAION, several other injuries are being evaluated in GLP-1 claims. These include necrotizing pancreatitis, a life-threatening inflammation of the pancreas. Pulmonary embolisms and deep vein thrombosis (DVT), which are blood clot conditions potentially linked to semaglutide. And in the most tragic cases, wrongful death claims where a patient died from complications attributable to GLP-1 medication use.
Some attorneys are also evaluating claims involving suicidal ideation linked to GLP-1 medications, though this area of litigation is still developing.
What does NOT currently qualify
It is important to understand the boundaries. Currently, most law firms are unable to accept cases based solely on pancreatitis (without the necrotizing component), kidney issues like semaglutide-related kidney stones, or thyroid cancer. The causal link for these conditions is more difficult to prove in the current litigation framework. That does not mean these injuries are not real or serious. It means the legal standard for proving manufacturer liability is higher for these specific conditions at this stage.
Common GLP-1 side effects like fatigue, headaches, sulfur burps, hair loss, and fatigue from semaglutide also do not meet the threshold for individual lawsuits, though they contribute to the broader picture of inadequate safety warnings.
How to find the right GLP-1 attorney
Choosing an attorney for a pharmaceutical mass tort case is fundamentally different from hiring a lawyer for a car accident or a contract dispute. The stakes are higher. The timeline is longer. And the wrong choice can cost you significantly, both in compensation and in years of your life spent waiting for resolution.
What makes a GLP-1 attorney different
A qualified GLP-1 attorney is not just any personal injury lawyer. Mass tort pharmaceutical litigation requires specific expertise that most attorneys simply do not have. The right attorney understands the science behind GLP-1 receptor agonists, knows how semaglutide works in the body, can interpret clinical trial data, and has experience navigating the MDL process.
They need to understand why gastrointestinal complications from tirzepatide differ from expected side effects. They need to be able to explain to a jury why delayed gastric emptying sometimes crosses the line from therapeutic mechanism to permanent injury. And they need the financial resources to fund years of litigation against some of the wealthiest pharmaceutical companies on the planet.
This is not a field where you want someone learning on the job.
Key qualifications to look for
When evaluating a GLP-1 attorney, look for these specific qualifications.
Mass tort experience. Has the attorney or firm handled pharmaceutical mass tort cases before? Previous experience with cases like Vioxx, Zantac, talcum powder, or opioid litigation means they understand the unique challenges of suing major pharmaceutical companies. Ask how many mass tort cases they have handled and what the outcomes were.
MDL involvement. Are they actively involved in the current GLP-1 MDL? In February 2026, Judge Marston appointed attorneys to the Plaintiffs Executive Committee, the leadership group coordinating litigation strategy for all plaintiffs. Firms connected to this leadership structure typically have the best access to discovery materials, expert witnesses, and strategic decisions that affect every case in the MDL.
Medical expertise. Does the firm have access to medical experts who can review your records and establish the causal link between your GLP-1 medication use and your injuries? Understanding the mechanism behind when semaglutide side effects appear and how they progress is crucial for building a strong case.
Financial resources. Pharmaceutical litigation is expensive. Expert witnesses, medical record reviews, depositions, and trial preparation cost hundreds of thousands of dollars. A firm that works on contingency (meaning they only get paid if you win) must have the financial capacity to front these costs for years before seeing any return. Smaller firms sometimes partner with larger firms for this reason, which can be perfectly fine as long as the arrangement is transparent.
Communication track record. Mass tort cases move slowly. You want an attorney who communicates proactively, provides regular updates even when nothing dramatic is happening, and responds to your questions within a reasonable timeframe. Ask current or former clients about their experience if possible.
Red flags to watch for
The GLP-1 litigation has attracted legitimate attorneys and opportunists alike. Be cautious of these warning signs.
Guaranteed outcomes. No attorney can guarantee a settlement amount or a specific result. Anyone who promises you a certain payout is being dishonest. The litigation has produced zero settlements so far. Anyone claiming otherwise is misrepresenting the current state of affairs.
Pressure to sign immediately. A reputable firm will give you time to review the retainer agreement, ask questions, and consult with family or other advisors. High-pressure tactics suggest the attorney is more interested in case volume than case quality.
Excessive advertising with no substance. You have probably seen the television commercials and online ads. "Did you take Ozempic? You may be entitled to compensation." These ads are not necessarily from bad firms, but they are from firms that are investing heavily in client acquisition. Make sure the firm behind the ad actually has the litigation experience to back up the marketing.
Unclear fee structures. Most GLP-1 attorneys work on contingency, typically taking 33-40% of any settlement or verdict. This should be clearly explained in writing. If there are additional fees, costs, or deductions beyond the contingency percentage, you should know about them upfront.
Questions to ask during your consultation
Most GLP-1 attorneys offer free initial consultations. Make them count. Ask these questions.
How many GLP-1 cases has your firm filed? This tells you their commitment to the litigation. A firm with 5 cases is in a very different position than one with 500.
Are you involved in the MDL leadership or working with firms that are? This affects how much influence the firm has over litigation strategy and settlement negotiations.
What is your assessment of my specific case? A good attorney will give you an honest evaluation, including potential weaknesses, rather than telling you exactly what you want to hear.
What is the expected timeline? Honest answers here will include phrases like "years, not months" and "bellwether trials expected in late 2026 at the earliest."
How will you communicate with me? Will you receive regular updates? Who is your primary point of contact? Will it be the attorney you are speaking with or a paralegal?
What costs will I be responsible for if we lose? In most contingency arrangements, the client owes nothing if the case is unsuccessful. But confirm this explicitly.

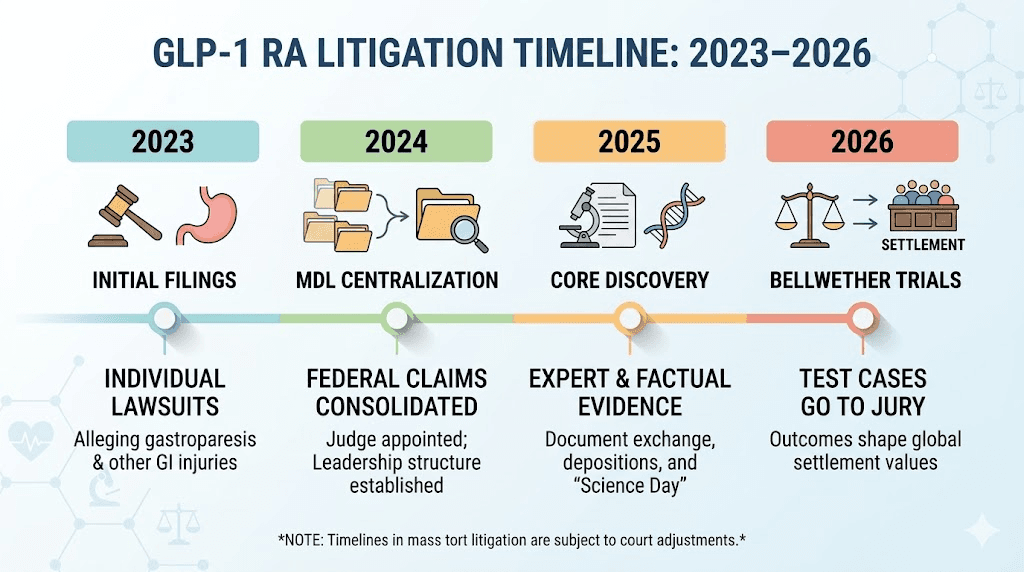
Understanding the MDL process
If you have never been involved in a mass tort lawsuit, the MDL process can feel confusing and painfully slow. Understanding how it works will help you set realistic expectations and evaluate whether your attorney is doing their job.
What is an MDL?
MDL stands for multidistrict litigation. When hundreds or thousands of similar lawsuits are filed across the country, all targeting the same defendant for the same type of injury, federal courts consolidate them before a single judge for pretrial proceedings. This is not a class action. Each plaintiff maintains their own individual case with their own specific injuries and damages. But the pretrial work, discovery, expert testimony challenges, and procedural motions, happens once for everyone rather than being repeated thousands of times in thousands of courts.
Think of it as efficiency without uniformity. Your case is still your case. But the groundwork that supports it gets done collectively.
Where the GLP-1 MDL stands now
As of March 2026, the GLP-1 MDL is in the expert discovery phase. This is where both sides retain medical and scientific experts, those experts prepare their reports and opinions, and the opposing side gets to challenge those opinions through a legal process called Daubert review. The court decides which expert testimony is reliable enough to be presented at trial.
Key upcoming milestones include expert discovery completion, scheduled for March 27, 2026. Motions for summary judgment are due April 16, 2026. After that, the court will begin preparing for bellwether trials.
What are bellwether trials?
Bellwether trials are test cases selected from the larger pool of lawsuits. A small number of representative cases go to trial first. The outcomes of these trials, both the verdicts and the reasoning behind them, inform settlement negotiations for the remaining thousands of cases.
No bellwether trial will occur until at least late 2026. This means that even in the most optimistic scenario, the first GLP-1 trial verdicts are months away. And broader settlements, if they happen, typically follow bellwether outcomes by months or years.
This timeline frustrates many plaintiffs. People who are dealing with ongoing medical issues from semaglutide complications or persistent tirzepatide problems want resolution now. But the MDL process, while slow, is designed to produce the best possible outcome for the largest number of people. Bellwether trials create leverage. Strong plaintiff verdicts push defendants toward settlement. And settlements in mass torts tend to be substantially larger than what individual plaintiffs could negotiate on their own.
Your role during the MDL
While the MDL proceeds, your primary responsibilities are straightforward. Provide your attorney with complete medical records. Document ongoing symptoms and treatments. Respond to requests for information promptly. And be patient.
Your attorney handles everything else. But you should expect regular updates on MDL progress, even when the news is simply that things are proceeding on schedule. If months go by without any communication from your legal team, that is a problem worth addressing.
Which GLP-1 drugs are involved in the litigation
The lawsuits cover multiple medications across two manufacturers. Understanding which drugs are involved helps you assess whether your specific situation falls within the litigation.
Novo Nordisk medications
Ozempic (semaglutide injection for type 2 diabetes) is the most prominent drug in the litigation. It was originally approved for diabetes management but became widely prescribed off-label for weight loss before Wegovy received its separate approval. Many of the earliest semaglutide injury claims involve Ozempic users.
Wegovy (semaglutide injection for weight management) contains the same active ingredient as Ozempic at higher doses. Its FDA approval specifically for weight loss in 2021 opened the floodgates for prescriptions to people without diabetes, a population that the studies suggest may face even higher risks for certain complications like NAION.
Rybelsus (oral semaglutide) is included in the litigation as well. While the oral form of semaglutide has different pharmacokinetics than injectable versions, it targets the same GLP-1 receptors and carries similar risks.
Eli Lilly medications
Mounjaro (tirzepatide) is the primary Eli Lilly drug in the litigation. Tirzepatide is a dual GIP/GLP-1 receptor agonist, meaning it targets two receptors rather than one. It has become one of the most prescribed weight loss medications in the world, and the comparison between semaglutide and tirzepatide is one of the most searched topics in the GLP-1 space. The dual mechanism does not appear to reduce the gastroparesis risk.
Zepbound (tirzepatide for weight management) and any compounded versions of these medications are also within the scope of the litigation, though claims involving compounded semaglutide or compounded tirzepatide may involve additional legal considerations regarding the compounding pharmacy.
What about newer GLP-1 drugs?
The litigation currently focuses on semaglutide and tirzepatide products. Newer medications like retatrutide, cagrilintide, survodutide, and mazdutide are still in clinical trials or early market stages. As they become more widely used, they could potentially face similar legal scrutiny if severe adverse events emerge. The mechanism of action, slowing gastric emptying via GLP-1 receptor stimulation, is shared across the entire drug class.
Eligibility requirements for filing a claim
Before contacting a GLP-1 attorney, understanding the basic eligibility criteria saves time for both you and the legal team. While every case is evaluated individually, certain baseline requirements apply across the litigation.
Basic eligibility criteria
You may qualify for a GLP-1 lawsuit if you meet these general conditions. You were prescribed a GLP-1 receptor agonist medication (Ozempic, Wegovy, Rybelsus, Mounjaro, Zepbound, or their compounded equivalents). You used the medication for at least 30 days. And you developed a qualifying injury that required medical treatment or hospitalization.
The 30-day minimum is a general guideline, not a hard cutoff. Some injuries can develop quickly. But most attorneys look for a meaningful period of use to establish the causal connection between the drug and the injury.
Documentation you need
Strong cases are built on strong documentation. Start gathering these materials now, even before you contact an attorney.
Prescription records. Proof that you were prescribed a GLP-1 medication, including the prescribing doctor, dates, dosage information, and the specific drug. If you were on a specific semaglutide dosage or following a tirzepatide dosing schedule, document that clearly.
Pharmacy records. Fill dates, refill history, and quantities dispensed. These create a clear timeline of your medication use.
Medical records. Emergency room visits, hospitalizations, diagnostic tests (especially gastric emptying studies for gastroparesis claims or eye examinations for NAION claims), surgical records, and ongoing treatment documentation.
Symptom timeline. A written record of when symptoms started, how they progressed, and how they affected your daily life. Include dates when you first noticed side effects after starting semaglutide and when those effects became severe enough to seek medical attention.
Financial records. Medical bills, lost wages documentation, out-of-pocket expenses for treatment, and any other financial impact from your injuries.
Factors that could complicate your claim
Certain pre-existing conditions or circumstances can make a GLP-1 claim more complex, though not necessarily impossible. If you had a history of gastroparesis before starting GLP-1 medication, the defense will argue the drug did not cause the condition. If you were using the medication off-label or obtained it through unofficial channels, that introduces additional questions. And if there was a significant time gap between stopping the medication and developing symptoms, establishing causation becomes more challenging.
None of these factors automatically disqualify you. But they make it more important to work with an experienced attorney who can address these complications in your specific case.
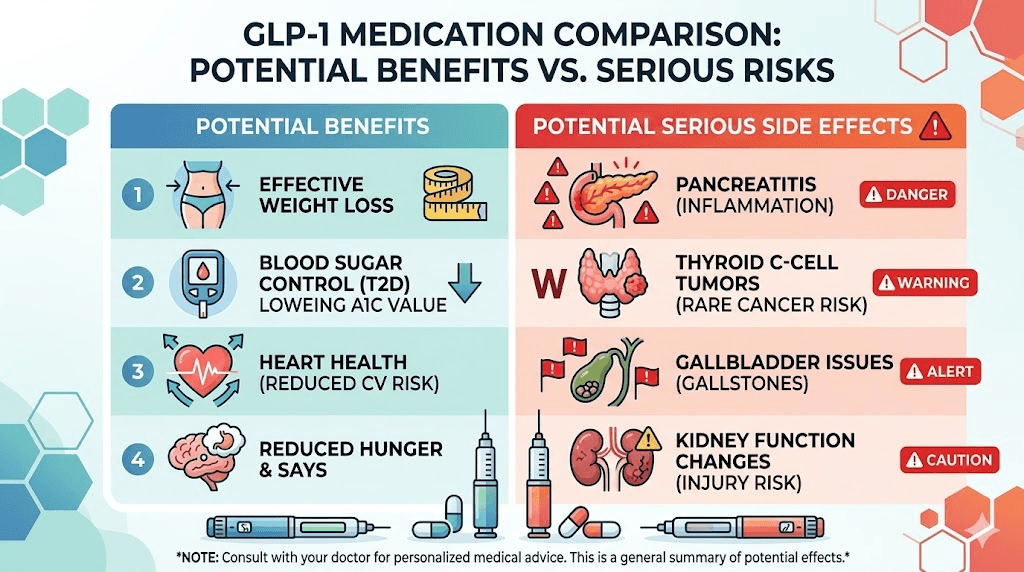
What compensation looks like in GLP-1 cases
Let us be direct about expectations. No settlements have been reached in GLP-1 litigation as of March 2026. Zero. Any specific dollar figures you see are projections based on comparable pharmaceutical mass torts, not actual outcomes from these cases.
That said, legal analysts have provided estimates based on the severity of injuries and precedent from similar litigation.
Projected settlement ranges
For severe gastroparesis cases requiring hospitalization, feeding tubes, or surgery, projections range from 350,000 to over 500,000 dollars. Cases involving permanent disability or ongoing medical needs may exceed those estimates. Wrongful death claims, where a patient died from complications attributable to GLP-1 medication, are projected at over 1 million dollars.
NAION vision loss cases are harder to project because the litigation track is newer. But permanent vision loss is a catastrophic injury, and historically, catastrophic injury claims in pharmaceutical litigation produce higher individual settlements.
These numbers are educated guesses. The actual settlement values will depend on bellwether trial outcomes, the strength of the scientific evidence as evaluated by the court, and the willingness of Novo Nordisk and Eli Lilly to negotiate rather than fight each case individually.
How settlements typically work in mass torts
If you are expecting a quick check in the mail, recalibrate. Mass tort settlements follow a predictable but slow process.
First, bellwether trials establish the legal viability of claims and give both sides data on what juries think. Strong plaintiff verdicts push defendants toward settlement. Then, the parties negotiate a global settlement framework that establishes different compensation tiers based on injury severity, duration of medication use, quality of documentation, and other factors. Individual plaintiffs are assigned to tiers, and payouts are calculated accordingly.
The entire process, from first filing to final payout, often takes 3 to 7 years in major pharmaceutical mass torts. The GLP-1 litigation is roughly 2.5 years in, with bellwether trials still ahead. Patience is not optional. It is required.
Contingency fee structure
Most GLP-1 attorneys work on contingency, meaning they take a percentage of your recovery (typically 33-40%) rather than charging hourly fees. If your case is unsuccessful, you owe nothing in attorney fees. However, some firms may require you to cover certain costs (like medical record retrieval fees) regardless of outcome. Clarify this before signing.
The contingency model means your attorney is financially invested in your success. They do not get paid unless you do. This aligns incentives and makes legal representation accessible to people who could not afford to pay hundreds of dollars per hour out of pocket.
Steps to take right now to protect your claim
Even if you have not decided whether to file a lawsuit, taking certain steps now protects your options for the future. Statutes of limitations, the legal deadlines for filing claims, vary by state and can be as short as one to two years from when you discovered (or should have discovered) the injury. Do not assume you have unlimited time.
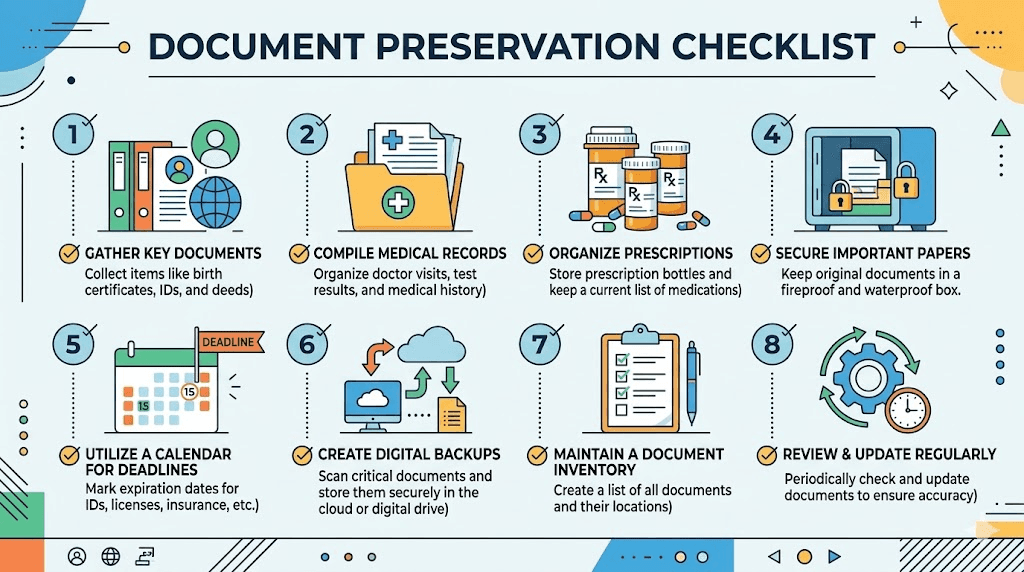
Preserve your evidence
Stop throwing away anything related to your GLP-1 medication use. Prescription bottles, pharmacy printouts, insurance statements, medical bills, appointment records, and even the medication packaging itself. All of it could be relevant.
If you kept a journal or notes about your experience, even informal ones on your phone, preserve those. Timestamped contemporaneous notes are powerful evidence because they were created in real time, not reconstructed later from memory.
Get a medical evaluation
If you suspect your GLP-1 medication caused an injury, get a formal medical evaluation as soon as possible. For gastroparesis claims, a gastric emptying study is the gold standard diagnostic test. For NAION claims, a comprehensive eye examination with documentation from an ophthalmologist is essential.
Medical documentation created close to the time of injury is far more valuable than records generated years later. The defense will scrutinize every gap in your medical timeline.
Document your impact
Beyond medical records, document how the injury has affected your life. Can you work? Can you care for your family? Can you eat normally? Can you participate in activities you enjoyed before? Keep a daily or weekly log of symptoms, limitations, and emotional impact. This becomes the foundation for calculating damages beyond just medical bills.
Consult an attorney sooner rather than later
Initial consultations with GLP-1 attorneys are almost always free. There is no downside to having your case evaluated, even if you decide not to proceed. What you do not want is to wait past the statute of limitations and lose your right to file entirely.
Most firms can do an initial case evaluation over the phone in 15 to 30 minutes. They will ask about your medication history, your injuries, your medical treatment, and your documentation. Based on that conversation, they will tell you whether they think you have a viable claim.
The science behind GLP-1 injury claims
Understanding the scientific basis for these lawsuits helps you evaluate the strength of your own potential claim and have more informed conversations with attorneys. The science is not simple. But the core concepts are accessible.
How GLP-1 medications cause gastroparesis
GLP-1 receptors exist throughout the body, including in the stomach and intestines. When GLP-1 receptor agonists like semaglutide or tirzepatide bind to these receptors, one of their effects is slowing the rate at which food moves from the stomach to the small intestine. This delayed gastric emptying contributes to appetite suppression and blood sugar control, which is why these drugs work for weight loss and diabetes management.
But the exact mechanism by which this therapeutic effect sometimes becomes a pathological one, where delayed emptying crosses the line into permanent gastroparesis, remains unclear. Research published in the Journal of Clinical Endocrinology and Metabolism describes the clinical consequences of delayed gastric emptying with both GLP-1 receptor agonists and tirzepatide, noting that the dual GIP/GLP-1 receptor agonist tirzepatide transiently delays gastric emptying similarly to selective long-acting GLP-1 receptor agonists.
Some researchers believe the effect may involve the vagus nerve, which plays a central role in gut motility. Others point to the prolonged half-life of newer GLP-1 drugs, which means the receptors are being stimulated continuously rather than in the pulsatile pattern that natural GLP-1 produces. Whatever the exact mechanism, the clinical reality is documented. A small but significant percentage of users, estimated at less than 2% in clinical trials but potentially higher in real-world use, develop gastroparesis symptoms that persist even after stopping the medication.
The NAION connection
The link between GLP-1 medications and NAION is newer and still being actively researched. NAION occurs when blood flow to the optic nerve is compromised, leading to ischemic damage. The proposed mechanisms include GLP-1 mediated changes in blood pressure regulation, effects on retinal and optic nerve blood flow, and potential interactions with cardiovascular risk factors that are common in the populations taking these drugs.
What makes the evidence compelling for legal purposes is the consistency across multiple independent studies, the dose-response relationship (higher risk with longer use), and the fact that non-diabetic users taking the drugs solely for fat loss show the highest risk increase. This last point is legally significant because it undermines the argument that the vision loss is caused by underlying diabetes rather than the medication itself.
Why the science matters for your case
In pharmaceutical litigation, the science is the battlefield. The defense will argue that the injuries are caused by other factors, pre-existing conditions, obesity itself, diabetes complications, or simple coincidence. The plaintiff must demonstrate, through expert testimony supported by clinical evidence, that the medication more likely than not caused the injury.
This is where the expert discovery phase currently underway in the MDL becomes so important. Both sides are marshaling their scientific evidence. The court will evaluate which expert opinions meet the reliability standards required for trial. The outcome of this process shapes the entire litigation, including settlement prospects.
State-by-state considerations
While the federal MDL consolidates pretrial proceedings, several state-specific factors can affect your claim. The most important is the statute of limitations.
Statute of limitations
Every state has a deadline for filing personal injury claims, typically ranging from one to six years depending on the state and the type of injury. For pharmaceutical injury cases, many states apply a "discovery rule," meaning the clock starts when you discovered (or reasonably should have discovered) that the medication caused your injury, not necessarily when the injury first occurred.
This distinction matters enormously. If you developed gastroparesis in 2024 but did not learn until 2025 that it could be connected to your GLP-1 medication, the discovery rule may extend your filing deadline. However, the specifics vary dramatically by state. Some states have strict cutoffs. Others are more flexible. An experienced GLP-1 attorney will know how your state handles these deadlines.
Do not gamble with timing. If you think you might have a claim, consult an attorney before the deadline becomes an issue.
State court vs. federal court
While most GLP-1 cases are consolidated in the federal MDL, some plaintiffs file in state court instead. State court filings can sometimes move faster or offer procedural advantages depending on the jurisdiction. Your attorney will recommend the best forum for your specific case based on the injury, the defendant, your state laws, and strategic considerations.
What happens after you hire an attorney
Once you sign a retainer agreement with a GLP-1 attorney, the process follows a fairly predictable path. Knowing what to expect reduces anxiety and helps you stay engaged with your case.
Case investigation and filing
Your attorney will conduct a thorough review of your medical records, prescription history, and injury documentation. They may request additional medical evaluations to strengthen the causal connection between your GLP-1 use and your injuries. Once they are satisfied that the case meets the legal standard, they will file your complaint in the appropriate court.
If your case joins the federal MDL, it will be assigned a case number and become part of the consolidated pretrial proceedings. Your individual case information is cataloged, but the pretrial work happens collectively.
Discovery phase
During discovery, both sides exchange information. The defendants produce internal documents, clinical trial data, safety reports, marketing materials, and communications about their GLP-1 drugs. Plaintiffs provide medical records, testimony about their injuries, and documentation of damages.
This phase is where the most important evidence emerges. Internal emails showing the manufacturer knew about risks earlier than disclosed. Safety data that was downplayed in FDA submissions. Marketing strategies that promoted off-label weight loss use despite known risks. Discovery is where cases are won or lost, long before anyone steps into a courtroom.
The waiting period
Between filing and resolution, there is waiting. Significant waiting. The MDL process handles motions, expert challenges, bellwether selection, and procedural disputes. Your attorney should keep you informed, but the honest truth is that many months will pass with no dramatic developments in your individual case.
Use this time productively. Continue medical treatment and documentation. Keep your attorney updated on any changes in your condition. And resist the temptation to settle prematurely for less than your case is worth just because you are tired of waiting.
Protecting yourself while using GLP-1 medications
Not everyone reading this guide has been injured. Some are currently taking GLP-1 medications and want to understand their rights proactively. Here is what you should know.
Monitoring for warning signs
If you are currently using a GLP-1 medication, awareness of serious side effects is your first line of defense. Gastroparesis warning signs include nausea and vomiting that persists beyond the initial adjustment period, severe bloating that does not improve with dietary changes, inability to eat normal portions even weeks after dose stabilization, and unexplained weight loss that goes beyond your target.
For vision concerns, watch for sudden painless vision loss in one eye, blurred or dimmed vision, and loss of peripheral vision. Any of these symptoms warrant immediate medical attention.
The first week on semaglutide and the first dose of tirzepatide often come with expected side effects like mild nausea and reduced appetite. These are normal. But symptoms that escalate rather than improve over the first few weeks deserve medical evaluation.
Informed consent matters
The legal concept at the heart of these lawsuits is informed consent. You have the right to know the risks of any medication before you take it. Ask your prescribing doctor specifically about gastroparesis risk and vision loss risk associated with your GLP-1 medication. Document that conversation. If your doctor was not aware of these risks, that itself becomes part of the larger narrative about inadequate manufacturer warnings.
Resources like SeekPeptides exist to help you understand the full picture of GLP-1 medications, including side effect comparisons between semaglutide and tirzepatide, general peptide safety information, and the latest research on complications. Being an informed patient is always your best protection, whether or not you ever need an attorney.
Dosing and risk reduction
Some researchers believe that slower dose escalation and lower maintenance doses may reduce the risk of severe GI complications. If you are starting a GLP-1 medication, following the prescribed semaglutide dosage schedule or tirzepatide dosing guide carefully matters. Skipping dose escalation steps or starting at higher doses increases the burden on your digestive system.
Using tools like the semaglutide dosage calculator or compounded tirzepatide dosage calculator to ensure accurate dosing, proper semaglutide reconstitution and tirzepatide reconstitution technique, and appropriate storage after reconstitution all contribute to safer use. Incorrect dosing from poor preparation could compound the risks.
Understanding the best time to take your GLP-1 shot, proper injection technique, and injection site selection are all part of responsible use. And knowing what foods to avoid on semaglutide or tirzepatide can help manage the GI effects that, if left unaddressed, could potentially escalate.
Common misconceptions about GLP-1 lawsuits
Misinformation about pharmaceutical litigation spreads quickly online. Let us clear up the most common misconceptions.
"The lawsuits mean GLP-1 drugs are being recalled"
False. Ozempic, Wegovy, Mounjaro, and other GLP-1 medications remain FDA-approved and on the market. The lawsuits allege inadequate warnings, not that the drugs should never have been approved. Many effective medications carry serious risks. The issue is whether patients were properly informed of those risks before making the decision to use them.
"I can file a lawsuit for any side effect"
Not currently. The litigation focuses on severe injuries, primarily gastroparesis, severe GI complications, and NAION vision loss. Common side effects like constipation, diarrhea, fatigue, headaches, and dry mouth, while unpleasant, do not meet the legal threshold for individual claims in the current litigation framework.
"Settlements are already being paid out"
False. As of March 2026, there have been zero settlements and zero trial verdicts in GLP-1 litigation. Anyone claiming to have received a settlement payout is either confused about the facts or being dishonest. The litigation is still in pretrial proceedings, with bellwether trials expected in late 2026 at the earliest.
"I missed my chance to file"
Possibly not. Statutes of limitations vary by state, and the discovery rule may extend your deadline. The only way to know for certain is to consult with an attorney who can evaluate your specific timeline and jurisdiction. Free consultations make this a zero-risk step.
"All GLP-1 attorneys are the same"
Far from it. The difference between a mass tort specialist with pharmaceutical litigation experience and a general practice attorney who started taking GLP-1 cases after seeing a business opportunity is enormous. That difference can affect your case outcome, your communication experience, and the timeline to resolution. Choose carefully.
The role of SeekPeptides in keeping you informed
While SeekPeptides is not a law firm and does not provide legal advice, the platform serves a critical role in helping GLP-1 users make informed decisions. Understanding the medications you take, their mechanisms, their risks, and the evolving research landscape is essential whether you are a current user, a former user considering legal action, or someone weighing whether to start GLP-1 therapy.
The GLP-1 lawsuit news tracker provides ongoing coverage of litigation developments. Comprehensive guides on topics like semaglutide results week by week, tirzepatide weight loss timelines, and how semaglutide makes you feel help users understand what normal looks like versus what warrants concern.
For researchers serious about understanding their GLP-1 protocols fully, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, detailed safety profiles, calculators for accurate dosing, and a community of thousands who have navigated these exact questions.
Frequently asked questions
How much does it cost to hire a GLP-1 attorney?
Most GLP-1 attorneys work on contingency, meaning there is no upfront cost. The attorney takes a percentage (typically 33-40%) of your settlement or verdict. If your case is unsuccessful, you typically owe nothing in attorney fees. Clarify any potential costs for medical records or administrative fees before signing.
How long will a GLP-1 lawsuit take?
Mass tort pharmaceutical cases typically take 3 to 7 years from initial filing to final resolution. The GLP-1 MDL began in 2023, with bellwether trials expected in late 2026. Broader settlements, if reached, would likely follow in 2027 or beyond. This timeline requires patience, but the collective process typically produces better outcomes than individual litigation.
Can I still take my GLP-1 medication while suing the manufacturer?
Yes. Filing a lawsuit does not require you to stop taking your medication. That decision should be made with your doctor based on your medical needs and risk factors, not legal considerations. However, document any ongoing side effects carefully, as they may be relevant to your claim.
What if I used compounded semaglutide or tirzepatide?
Claims involving compounded versions of GLP-1 medications are more complex because liability may extend to the compounding pharmacy in addition to the original manufacturer. An experienced attorney can evaluate whether your claim against the brand-name manufacturer remains viable and whether the compounder bears separate liability.
Do I qualify if my gastroparesis resolved after stopping the medication?
Potentially. Even temporary gastroparesis that required medical treatment, hospitalization, or caused significant suffering may qualify for a claim. The severity of the injury and the extent of medical treatment are more important factors than whether the condition is permanent. Discuss your specific situation with an attorney.
What if my doctor did not tell me about these risks?
This actually strengthens the failure-to-warn argument. If the manufacturer did not adequately communicate risks to healthcare providers, doctors could not pass that information along to patients. Your doctor not knowing about gastroparesis or NAION risks is evidence of the manufacturer failure that these lawsuits target.
Can I join the lawsuit if I live outside the United States?
The current MDLs are in U.S. federal court and primarily cover injuries occurring within the United States. International claims face additional jurisdictional challenges. However, if you were prescribed the medication in the U.S. or suffered injuries here, you may still qualify regardless of your current residence. Consult an attorney familiar with cross-border pharmaceutical litigation.
Is there a class action I can join?
The GLP-1 litigation is NOT a class action. It is a mass tort MDL, which means each plaintiff has their own individual case with their own specific injuries and damages. This is actually better for plaintiffs because compensation is based on your individual circumstances rather than being divided equally among all class members.
External resources
U.S. District Court, Eastern District of Pennsylvania - MDL 3094 Official Page
Journal of Clinical Endocrinology and Metabolism - GLP-1 and Delayed Gastric Emptying
In case I do not see you, good afternoon, good evening, and good night. May your rights stay protected, your documentation stay thorough, and your recovery stay steady.