Feb 28, 2026

Over 3,000 lawsuits. Two pharmaceutical giants. And a growing list of patients who say nobody warned them. The GLP-1 litigation wave has grown from a single filing in August 2023 into one of the largest pharmaceutical legal battles in recent memory, and the numbers keep climbing every month.
This is not a small dispute. It is not a handful of disgruntled patients chasing settlement checks. The GLP-1 receptor agonist litigation now encompasses claims against eight separate medications, involves allegations of severe gastroparesis, permanent vision loss, bowel obstruction, pancreatitis, and kidney injury, and targets both Novo Nordisk and Eli Lilly with accusations that they knew about these risks and chose profits over patient safety. Legal analysts project total liability exceeding $2 billion.
But here is what makes this situation uniquely complicated for anyone researching peptides. The same compounds at the center of these lawsuits, semaglutide and tirzepatide, are also the most widely discussed peptides in the research community. Understanding the legal landscape is not just about following courtroom drama. It is about understanding the actual risk data, the specific side effects driving claims, and what this means for anyone involved in peptide research. Whether you are tracking your own protocol or simply staying informed, this guide covers everything that matters.
The scope of GLP-1 litigation
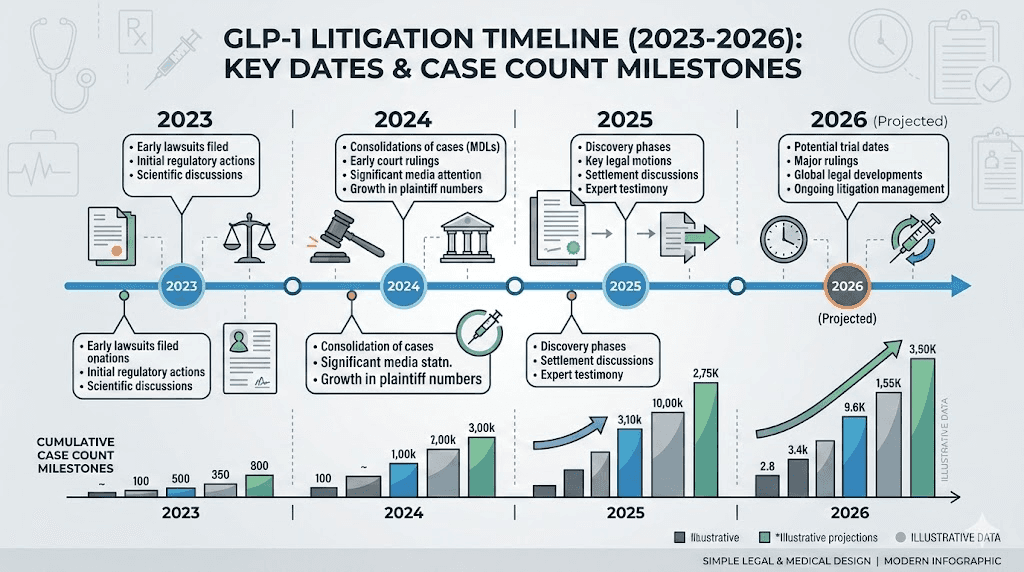
The numbers tell a story that keeps accelerating. In August 2023, Jaclyn Bjorklund filed the first lawsuit against Novo Nordisk and Eli Lilly after experiencing severe vomiting on Ozempic and Mounjaro that was so violent it caused tooth loss and required multiple emergency room visits. By September 2025, the federal multidistrict litigation had grown to 2,914 cases. By January 2026, that number hit 3,097. As of February 2026, it stands at 3,191 and counting, with approximately 200 new cases being filed each month.
That is not slowing down.
The litigation is consolidated under MDL 3094, officially titled "In Re: Glucagon-like Peptide-1 Receptor Agonists Products Liability Litigation," in the Eastern District of Pennsylvania. Judge Karen Spencer Marston presides over both the original gastroparesis-focused cases and a newer set of vision loss claims. In December 2025, the U.S. Judicial Panel on Multidistrict Litigation created an additional MDL (No. 3163) specifically for NAION (non-arteritic anterior ischemic optic neuropathy) vision loss cases, which already includes more than 30 federal filings with new ones arriving regularly.
Which drugs are named in the lawsuits
Eight GLP-1 receptor agonist medications are named across the litigation. These span two manufacturers and cover both diabetes management and weight loss indications. Understanding which drugs are involved helps contextualize the scope, because this is not just about one medication or one company. It is a class-wide issue that touches every major GLP-1 agonist on the market.
Novo Nordisk products:
Ozempic (semaglutide injection for type 2 diabetes)
Wegovy (semaglutide injection for weight management)
Rybelsus (oral semaglutide for type 2 diabetes)
Saxenda (liraglutide for weight management)
Victoza (liraglutide for type 2 diabetes)
Eli Lilly products:
Mounjaro (tirzepatide for type 2 diabetes)
Zepbound (tirzepatide for weight management)
Trulicity (dulaglutide for type 2 diabetes)
For researchers tracking semaglutide protocols or tirzepatide timelines, these lawsuits carry direct relevance. The side effects being litigated are not theoretical. They are documented in medical records, emergency room visits, and hospitalizations across thousands of patients. Anyone working with these compounds needs to understand the specific risks that drove this litigation.
The core legal allegations
At the heart of every GLP-1 lawsuit sits a simple accusation. The manufacturers knew. They knew about the risks. They chose not to warn adequately. And patients paid the price with their health.
More specifically, the lawsuits allege that Novo Nordisk and Eli Lilly failed to warn patients and prescribing physicians about the full spectrum and severity of potential side effects, made false, misleading, or incomplete representations regarding the safety of their GLP-1 products, aggressively marketed these drugs for weight loss while concealing known risks, downplayed the nature, duration, extent, and seriousness of gastrointestinal events, and failed to update warning labels even as adverse event reports accumulated.
U.S. District Judge Karen Marston allowed most of the plaintiffs' claims to proceed, including accusations of failure to warn and misrepresentation of safety and effectiveness for weight management. This was a significant procedural win for plaintiffs, signaling that the court found sufficient evidence to move the litigation forward.
Gastroparesis: the primary injury claim
Stomach paralysis. That is the non-medical translation of gastroparesis, and it is the injury driving the majority of GLP-1 lawsuits. When the nerves and muscles of the stomach stop pushing food into the small intestine, food sits there. For hours. Sometimes days. The consequences range from debilitating nausea to life-threatening complications.
GLP-1 receptor agonists work partly by slowing gastric emptying. That is by design. It is one of the mechanisms that produces appetite suppression and helps with dietary changes. But the lawsuits allege that for some patients, this slowing goes far beyond therapeutic benefit and crosses into dangerous territory where the stomach essentially stops functioning.
The JAMA study that changed everything
On October 5, 2023, a study published in JAMA and authored by Mohit Sodhi and colleagues at the University of British Columbia provided the first large-scale epidemiological evidence linking GLP-1 agonists to serious gastrointestinal events in weight loss patients. This was not a small case report. The researchers analyzed a random sample of 16 million patients from the PharMetrics Plus database, which captures 93% of all outpatient prescriptions and physician diagnoses in the United States, covering data from 2006 to 2020.
The findings were stark.
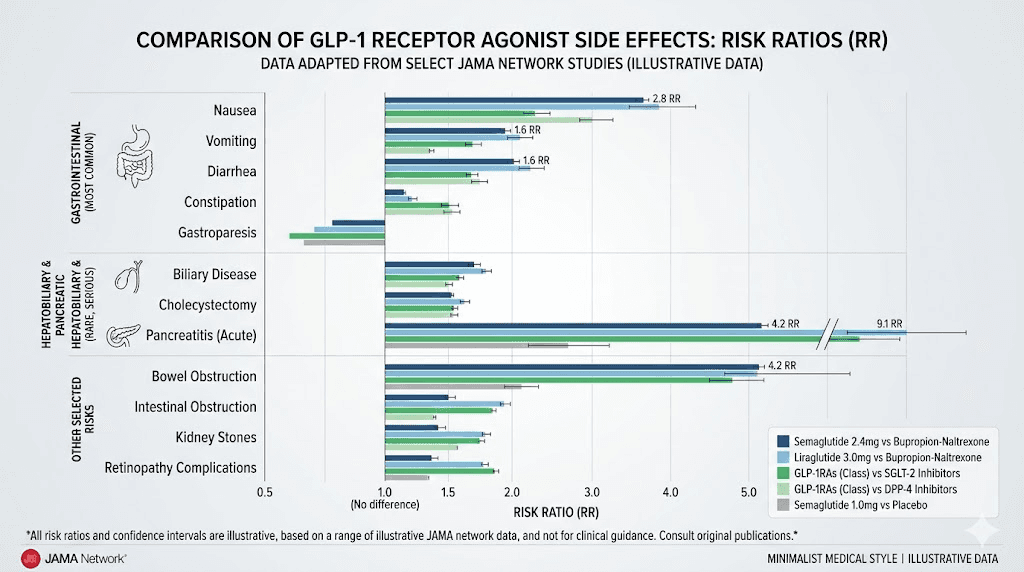
Compared to patients taking bupropion-naltrexone (another weight loss medication), GLP-1 agonist users had a 3.67 times higher risk of developing gastroparesis, a 9.09 times higher risk of pancreatitis, and a 4.22 times higher risk of bowel obstruction. These were not marginal differences. A nearly four-fold increase in stomach paralysis risk and a nine-fold increase in pancreatic inflammation risk represented exactly the kind of data that plaintiffs' attorneys could build cases around.
What made this study particularly significant was its focus on non-diabetic patients using GLP-1 agonists specifically for weight loss. Previous safety data had primarily come from diabetes populations, where pre-existing conditions like gastrointestinal issues could confound results. By isolating weight loss users, the Sodhi study eliminated that variable and showed that the drugs themselves, not underlying diabetes, were associated with these adverse events.
What gastroparesis looks like in practice
Court filings describe experiences that go far beyond typical medication side effects. Laura Marrero used Ozempic and experienced persistent vomiting from May through October 2023, was hospitalized in August with abdominal pain and rectal bleeding, and continues to suffer complications. Jaclyn Bjorklund, the first plaintiff, reported vomiting so severe it caused dental damage, required multiple ER visits, and left her dependent on additional medications to manage the condition.
Untreated gastroparesis can lead to malnutrition from the inability to absorb nutrients, severe dehydration from persistent vomiting, bezoar formation (solid masses in the stomach), bacterial overgrowth in the stagnant food, and dangerous blood sugar fluctuations. These are not temporary inconveniences. For many plaintiffs, the condition persists long after discontinuing the medication. Some may never fully recover normal gastric function.
For researchers monitoring their own GLP-1 protocols, understanding the difference between normal gastric slowing and pathological gastroparesis is critical. Symptoms like constipation, persistent bloating, and excessive burping may signal more than typical adjustment effects. If food sits in the stomach for extended periods, it demands medical attention, not just a wait-and-see approach.
The objective evidence requirement
Judge Marston issued an important ruling that will shape how gastroparesis cases proceed. Plaintiffs claiming gastroparesis must support their diagnosis with objective medical evidence, specifically a properly conducted gastric emptying study using scintigraphy, a breath test, or a wireless motility capsule. Self-reported symptoms alone are not sufficient.
This ruling may narrow the pool of viable claims, particularly for individuals diagnosed based solely on clinical judgment without formal testing. But it also strengthens the remaining cases, because plaintiffs who can produce objective test results demonstrating delayed gastric emptying have powerful evidence that their condition is measurable and verifiable, not subjective.
Vision loss: the emerging battlefront
If gastroparesis cases formed the first wave of GLP-1 litigation, vision loss claims represent the second, and potentially more legally powerful. The shift began after research linked semaglutide use to a rare but devastating eye condition called non-arteritic anterior ischemic optic neuropathy, or NAION.
NAION occurs when blood flow to the optic nerve is disrupted, causing sudden, painless vision loss in one or both eyes. There is no effective treatment. The damage is typically permanent. And the studies linking GLP-1 drugs to this condition show risk increases that are difficult to dismiss.
The JAMA Ophthalmology findings
A study published in JAMA Ophthalmology reported findings that sent shockwaves through both the medical and legal communities. Researchers found that patients with type 2 diabetes who took semaglutide-containing medications had a 4.28 times higher risk of developing NAION compared to patients not on GLP-1 agonists. That is a more than four-fold increase.
But the numbers were even more striking for weight loss patients. Overweight or obese individuals who took semaglutide had a 7.64 times higher risk, nearly an eight-fold increase. Over a 36-month period, the cumulative incidence of NAION in the semaglutide group reached 6.7%, compared to just 0.8% in the non-GLP-1 group. In the overweight population specifically, 20 NAION events occurred in the semaglutide cohort versus only 3 in the comparison group.
Additional research reinforced these findings. A 2024 study from the Netherlands analyzing over 400,000 patients found that type 2 diabetes users of semaglutide had twice the NAION risk. A study on liraglutide (the active ingredient in Saxenda and Victoza) found a 179% higher NAION risk compared to non-users. And a Swedish study from July 2025 found that GLP-1 users had a 1.6 times higher risk of retinal vein occlusion, suggesting the eye damage concerns extend beyond NAION alone.
A separate study from the University of Toronto in June 2024 found that semaglutide users were twice as likely to develop neovascular age-related macular degeneration, adding yet another eye condition to the growing list of concerns.
The legal significance of vision loss claims
Vision loss cases carry unique legal weight for several reasons. First, the injuries are objectively verifiable. Unlike gastroparesis, which requires specific testing to confirm, vision loss is measurable and undeniable. Second, the damage is permanent. There is no treatment that reverses NAION. Third, the impact on quality of life is profound and easily communicable to a jury.
In December 2025, the creation of a separate MDL (No. 3163) for vision loss cases demonstrated that courts view these claims as distinct from the gastrointestinal litigation. Judge Marston issued an order on January 29, 2026, establishing a streamlined direct filing process for vision loss lawsuits, making it easier for new plaintiffs to join the litigation.
Settlement projections for NAION cases run significantly higher than gastroparesis claims. Legal analysts estimate unilateral (one eye) blindness cases at $500,000 to over $1,000,000, while bilateral vision loss cases are expected to exceed $1,000,000. Compare that to gastroparesis settlements projected at $200,000 to $500,000 for severe cases and $50,000 to $150,000 for temporary conditions.
For anyone using semaglutide or tirzepatide, monitoring vision changes is something these studies suggest should not be ignored. Any sudden changes in visual acuity, particularly painless loss of vision in one eye, warrant immediate ophthalmological evaluation.
Beyond gastroparesis and vision loss: other injury claims
While gastroparesis and NAION dominate the headlines, the GLP-1 litigation encompasses a broader spectrum of injuries. Understanding the full range of claimed side effects provides a more complete picture of the safety concerns being litigated.
Pancreatitis
The Sodhi JAMA study found GLP-1 agonist users had a 9.09 times higher risk of pancreatitis, making it the highest relative risk increase among the gastrointestinal conditions studied. Pancreatitis, inflammation of the pancreas, can range from acute episodes requiring hospitalization to chronic conditions causing permanent organ damage. The FDA drug information label for these medications now includes severe pancreatitis as a recognized risk.
Pancreatitis symptoms include severe upper abdominal pain that may radiate to the back, nausea and vomiting, fever, and elevated pancreatic enzymes in blood tests. For researchers on GLP-1 protocols, any severe abdominal pain, especially after eating, should not be attributed to typical dietary adjustment without ruling out pancreatic involvement.
Bowel obstruction and ileus
Bowel obstruction, where the intestines become partially or fully blocked, appeared at 4.22 times higher risk in GLP-1 users according to the JAMA data. Ileus, a condition where normal bowel motility stops without physical obstruction, was significant enough that the FDA added ileus warnings to the Ozempic label in September 2023.
One plaintiff described the experience bluntly: "My colon blew up." These are not subtle side effects. Bowel obstruction can require surgical intervention and carries risks of tissue death, perforation, and sepsis if not treated promptly.
Kidney injury
Acute kidney injury claims stem from the cascade effect of GLP-1 side effects. The medications can cause severe diarrhea and vomiting, which leads to dehydration, which in turn damages the kidneys. While the kidney injury may be secondary to the gastrointestinal effects rather than a direct drug action, the lawsuits argue that inadequate warnings about the severity and persistence of GI side effects prevented patients from taking steps to protect their renal function.
Approximately 25,000 emergency room visits between 2022 and 2023 involved semaglutide, with the vast majority related to gastrointestinal complaints, according to CDC data. Each of those ER visits represents a potential dehydration event that could impact kidney function.
Gallbladder disease
Gallstones and gallbladder inflammation are additional claimed injuries in the litigation. Rapid weight loss itself is a known risk factor for gallstone formation, complicating the question of whether GLP-1 drugs directly cause gallbladder problems or whether the weight loss they produce is the intermediate cause. Regardless, plaintiffs argue that manufacturers should have warned more explicitly about this risk and recommended monitoring.
Thyroid cancer concerns: what the evidence actually shows
GLP-1 medications carry an FDA boxed warning about thyroid C-cell tumors based on rodent studies. However, recent research has provided some reassurance on this front. A study concluded that GLP-1 drugs such as Ozempic, Wegovy, Mounjaro, and Zepbound do not appear to increase the risk of thyroid cancer in humans. While the boxed warning remains based on the animal data, the absence of human evidence has meant that thyroid cancer claims have not become a major component of the current litigation.
This is an important distinction for researchers to understand. Not every theoretical risk translates to documented human harm, and the legal system reflects that distinction. The strongest claims are those backed by human epidemiological data, which currently points most powerfully at gastroparesis and NAION.
Key procedural developments and timeline
Understanding where the litigation stands procedurally helps predict what happens next and when outcomes might emerge. Pharmaceutical litigation moves slowly, and the GLP-1 cases are no exception.
Critical dates in the litigation timeline
August 2, 2023: Jaclyn Bjorklund files the first Ozempic gastroparesis lawsuit against Novo Nordisk and Eli Lilly. This single filing opens the floodgates.
September 2023: The FDA adds ileus warnings to the Ozempic label, acknowledging at least one of the gastrointestinal risks plaintiffs are alleging.
October 5, 2023: The Sodhi study publishes in JAMA, providing the epidemiological evidence that dramatically accelerates lawsuit filings.
September 2024: All federal Ozempic-related cases are consolidated into MDL 3094 in the Eastern District of Pennsylvania under Judge Karen Spencer Marston.
January 2025: Novo Nordisk updates the Ozempic label to warn against use in patients with severe gastroparesis, an implicit acknowledgment of the link between the drug and the condition.
June 2025: The World Health Organization warns that semaglutide may rarely cause NAION, adding international regulatory weight to the vision loss claims. European regulators add NAION warnings to the Ozempic label the same month.
July 2025: Fact discovery closes in the MDL.
August 2025: Judge Marston rules that gastroparesis plaintiffs must demonstrate objective diagnostic evidence, shaping case viability standards.
December 2025: A separate MDL (No. 3163) is created for GLP-1 vision loss cases, with initial 29 cases consolidated.
January 29, 2026: Judge Marston establishes a streamlined direct filing process for vision loss lawsuits.
February 2026: Case count reaches 3,191 and continues to grow. Over 100 new lawsuits filed in recent weeks alone.
What comes next: bellwether trials and settlements
The MDL is currently in the expert discovery phase. Expert depositions were scheduled through early 2026, with summary judgment motions due in April 2026. Bellwether case selections, the small group of representative cases that go to trial first to test legal theories and gauge potential outcomes, are expected soon after.
Some legal analysts project bellwether trials could begin in late 2026, though others suggest they may not arrive until 2027. Either way, no global settlements have been announced as of February 2026, and most legal experts agree that meaningful settlements are unlikely before late 2026 at the earliest.
This timeline matters for several reasons. First, it means the litigation will continue to grow as new cases are filed faster than existing ones are resolved. Second, the bellwether trial outcomes will dramatically influence settlement negotiations, as both sides assess how juries respond to the evidence. Third, individual settlement values will vary widely based on injury severity, documentation quality, and the strength of each plaintiff's causation evidence.
Projected settlement ranges
While no settlements have been finalized, legal analysts have published projections based on comparable pharmaceutical litigation and the specific injuries involved.
Injury type | Projected settlement range | Key factors |
|---|---|---|
Severe/permanent gastroparesis | $200,000 to $500,000 | Objective testing, duration, impact on daily life |
Temporary gastroparesis | $50,000 to $150,000 | Recovery timeline, medical documentation |
NAION (unilateral) | $500,000 to $1,000,000+ | Degree of vision loss, impact on employment |
NAION (bilateral) | Expected to exceed $1,000,000 | Total disability, permanent impact |
Bowel obstruction (surgical) | $250,000 to $700,000 | Surgical records, complications, recovery |
Pancreatitis (acute) | $100,000 to $400,000 | Hospitalization records, ongoing damage |
These projections suggest total litigation liability could exceed $2 billion, though final numbers will depend heavily on bellwether trial outcomes and the ultimate scope of proven causation.
The regulatory response
How have regulators responded to the safety signals that these lawsuits are built upon? The answer is a mix of incremental label changes, formal warnings, and what plaintiffs argue was too little, too late.
FDA actions
The FDA has taken several steps in response to accumulating safety data. In September 2023, ileus was added to the Ozempic warning label. In January 2025, the label was updated to warn against use in patients with severe gastroparesis. These changes represent regulatory acknowledgments that the gastrointestinal risks extend beyond typical side effects.
But plaintiffs argue these changes came years after the drugs reached market, during which millions of patients were exposed to undisclosed risks. The appetite-suppressing effects that made these drugs blockbusters are mechanistically linked to the very gastric slowing that causes gastroparesis. Plaintiffs contend this connection should have been obvious to manufacturers from the earliest clinical trials.
Additionally, approximately 25,000 emergency room visits involving semaglutide occurred between 2022 and 2023 according to CDC data, largely for gastrointestinal complaints. This volume of adverse events, plaintiffs argue, should have triggered faster and more comprehensive label updates.
International regulatory response
International regulators have moved on some fronts ahead of the FDA. In June 2025, the WHO issued a warning that semaglutide may rarely cause NAION, and European regulators added NAION warnings to the Ozempic label. The European Medicines Agency estimated NAION incidence at approximately 1 in 10,000 users.
While 1 in 10,000 might sound rare, the sheer scale of GLP-1 usage transforms this into a significant absolute number. With millions of patients taking these medications worldwide, even a 1-in-10,000 incidence rate translates to thousands of potential vision loss cases globally.
What the label changes tell researchers
For anyone following semaglutide protocols or tirzepatide timelines, the regulatory trajectory provides useful safety information. The fact that labels have been updated, that warnings have been added, and that international agencies have issued alerts does not mean these drugs are uniformly dangerous. It means specific risks require monitoring.
The label changes essentially provide a checklist of what to watch for. Persistent gastrointestinal symptoms beyond typical adjustment periods, any sudden vision changes, signs of pancreatitis (severe abdominal pain), and symptoms of kidney stress (decreased urine output, swelling) all deserve prompt medical evaluation rather than assumption that they are normal side effects.
The compounding pharmacy battle
Running parallel to the patient injury litigation is an entirely different legal war, one that directly impacts the peptide research community. Eli Lilly and Novo Nordisk have turned their legal departments toward compounding pharmacies and telehealth platforms, creating a separate but interconnected battlefield.
Manufacturer lawsuits against compounders
In April 2025, Eli Lilly filed federal lawsuits in northern California against several telehealth companies, including Fella Health and Mochi Health, accusing them of distributing unapproved versions of tirzepatide without complying with FDA requirements. In August 2025, Novo Nordisk followed suit, filing against 14 defendants including pharmacies engaged in compounding or distributing semaglutide products.
The manufacturers argued that these entities were producing non-FDA-approved copies of their drugs, violating trademark and patent protections, and potentially putting patients at risk with unregulated formulations. This legal offensive coincided with the FDA resolving the tirzepatide shortage in December 2024, which removed the regulatory exemption that had allowed compounding pharmacies to produce tirzepatide copies during the shortage period.
Compounders fighting back
The compounding pharmacy response has been aggressive. An Arizona-based compounding pharmacy filed an antitrust lawsuit accusing Eli Lilly and Novo Nordisk of unlawfully restricting competition in the GLP-1 market. The complaint alleged that the manufacturers forged exclusive alliances with telehealth companies that barred those providers from working with compounders, effectively using market power to eliminate competition.
This antitrust angle adds a layer of complexity to the GLP-1 legal landscape. If compounders succeed in their claims, it could reshape access to compounded versions of these medications. If the manufacturers prevail, the compounded semaglutide and compounded tirzepatide markets could shrink significantly.
Regulatory enforcement at the state level
State regulators have also entered the fray. In June 2025, the Ohio Board of Pharmacy issued a formal notice that retatrutide, a newer investigational GLP-1 product, was not eligible for compounding under any circumstances, with the board threatening summary suspension for pharmacies that continued to do so.
California inspectors began flagging compounded GLP-1 products during routine pharmacy surveys over the summer of 2025, citing even minor modifications as potentially creating an "essentially a copy" of commercially available products unless accompanied by clear documentation of medical necessity.
For researchers who have been sourcing through compounding pharmacies or 503b facilities, this legal environment creates uncertainty. The availability, legality, and quality of compounded GLP-1 products are all in flux as courts work through these competing claims.
Who can file a GLP-1 lawsuit
Understanding the eligibility criteria for GLP-1 lawsuits helps separate legitimate claims from general dissatisfaction with medication side effects. Not everyone who experiences discomfort on a GLP-1 drug has a viable legal case.
Basic eligibility requirements
To pursue a GLP-1 lawsuit, an individual generally must have been prescribed a GLP-1 receptor agonist (Ozempic, Wegovy, Rybelsus, Mounjaro, Zepbound, Trulicity, Saxenda, or Victoza) for either diabetes management or weight loss. They must then have developed a serious side effect such as gastroparesis, bowel obstruction, vision loss (NAION), pancreatitis, gallbladder disease, or kidney injury that requires medical documentation.
Medical records are essential. Plaintiffs need documentation showing their GLP-1 usage, the timing of symptom onset relative to drug use, diagnostic testing confirming their condition, and treatment records demonstrating the severity and impact of their injury.
The strength of different claim types
Not all claims carry equal weight. The strongest cases typically involve objective diagnostic evidence (gastric emptying studies for gastroparesis, ophthalmological testing for NAION), clear temporal relationship between drug use and symptom onset, severe outcomes requiring hospitalization or surgery, documented impact on quality of life and ability to work, and absence of pre-existing conditions that could explain the symptoms.
Claims are weaker when diagnosis relies solely on clinical symptoms without objective testing, when pre-existing conditions like diabetes or hypertension could contribute to the alleged injury, or when the side effects, while unpleasant, are temporary and resolve with drug discontinuation.
The defense strategy
Manufacturers are not conceding these cases. Their defense strategy includes blaming underlying conditions (diabetes, hypertension, high cholesterol) for the alleged injuries, challenging expert testimony on gastroparesis that lacks objective testing confirmation, arguing multifactorial causation to reduce drug-specific liability, and pointing to label warnings that they claim were adequate.
This defense approach means that plaintiffs with the cleanest cases, those with no confounding conditions, strong diagnostic evidence, and clear timelines, will likely drive the largest settlements.
What this means for researchers and peptide users
The GLP-1 litigation carries implications that extend well beyond the courtroom. For anyone involved in peptide research, particularly with semaglutide or tirzepatide, several practical takeaways emerge from this legal landscape.
Safety monitoring becomes non-negotiable
The studies underpinning these lawsuits provide specific risk data that should inform any research protocol. A 3.67x increase in gastroparesis risk means gastrointestinal symptoms deserve serious attention, not dismissal as "normal adjustment effects." A 4-8x increase in NAION risk means any vision changes warrant immediate evaluation. A 9x increase in pancreatitis risk means severe abdominal pain is a red flag.
Researchers tracking initial experiences with semaglutide or first doses of tirzepatide should establish baseline monitoring that includes regular assessment of gastrointestinal function, awareness of vision changes, attention to any upper abdominal pain, and hydration status (particularly if experiencing nausea or vomiting).
Dosing protocols and risk management
The litigation data reinforces something the peptide research community already knows. Starting low and titrating slowly is not just about comfort. It is about safety. The risk of gastroparesis and other serious GI events may correlate with dose escalation speed. Protocols that rush through lower doses to reach higher therapeutic levels faster could increase vulnerability to the exact side effects being litigated.
Microdosing approaches and careful titration schedules take on new significance in light of this litigation data. Conservative dosing charts exist for a reason, and the lawsuit evidence underscores why departing from gradual escalation carries measurable risk.
Documentation and self-monitoring
The legal proceedings highlight the importance of documentation for anyone using GLP-1 compounds. Keeping detailed records of when you started, what doses you used, what side effects you experienced, and when those effects appeared or resolved creates exactly the kind of timeline that distinguishes between manageable side effects and potentially serious adverse events.
Tools like the GLP-1 plotter and dosage calculators are not just convenience features. They support the kind of systematic tracking that helps researchers identify problems early, before they become the type of injuries being litigated in federal court.
The compounding question
The parallel litigation against compounding pharmacies raises important questions about product quality and regulatory compliance. Researchers sourcing compounded GLP-1 products should be aware that the legal landscape is shifting rapidly. Sources that were legal during the shortage period may not retain that status, and quality controls may vary significantly between providers.
SeekPeptides has consistently emphasized the importance of understanding the regulatory and safety landscape around these compounds. The current litigation reinforces why thorough research, careful sourcing, and diligent monitoring are not optional steps but essential practices for anyone working with GLP-1 agonists.
The broader peptide regulation landscape
GLP-1 lawsuits do not exist in isolation. They are part of a larger trend in peptide regulation that affects the entire research community. Understanding how this litigation fits into the broader regulatory picture provides crucial context.
How GLP-1 litigation affects peptide regulation
High-profile lawsuits attract regulatory attention. When thousands of patients file claims alleging serious injuries from a class of drugs, regulators respond with increased scrutiny, not just of the drugs themselves, but of the entire compound class and related substances. The GLP-1 litigation has already contributed to stricter enforcement against compounding pharmacies, increased FDA attention to peptide products marketed for weight loss, state-level restrictions on compounding of GLP-1 and related compounds, and greater emphasis on safety documentation for all peptide products.
For the broader peptide research community, this means that regulatory changes driven by GLP-1 litigation could have ripple effects on access to other compounds, the legal status of compounding for non-GLP-1 peptides, and the overall regulatory framework for peptide research.
The impact on GLP-1 alternatives
As the legal cloud over semaglutide and tirzepatide grows, interest in alternative approaches has increased. Researchers are exploring newer GLP-1 agonists like retatrutide, combination approaches with cagrilintide, emerging compounds like mazdutide, and oral alternatives like orforglipron.
However, it is important to note that many of the safety concerns driving the current litigation may apply to the entire GLP-1 receptor agonist class, not just individual drugs. The mechanism of action that slows gastric emptying is fundamental to how all GLP-1 agonists work. If gastroparesis is an inherent risk of this mechanism, newer drugs in the same class may carry similar risks, and the legal precedents set by the current litigation could apply to future products as well.
Implications for non-GLP-1 peptide research
Researchers working with non-GLP-1 peptides like BPC-157, TB-500, GHK-Cu, and other compounds should monitor this litigation for its regulatory implications. The principle established here, that manufacturers can be held liable for inadequate safety warnings, reinforces the importance of thorough research, careful dosing, and awareness of potential risks when working with any peptide compound.
The current litigation also underscores why resources like SeekPeptides matter. Having access to comprehensive, evidence-based safety information, detailed protocols, and a community of experienced researchers helps navigate an increasingly complex regulatory and safety landscape.
Understanding the science behind the claims
To fully grasp the GLP-1 litigation, you need to understand the mechanism that makes these drugs work, and why that same mechanism creates risk. This is not just legal context. It is essential knowledge for anyone researching these compounds.
How GLP-1 agonists affect the gut
GLP-1 receptor agonists mimic the natural hormone glucagon-like peptide-1. Among their many effects, they stimulate insulin secretion (which lowers blood sugar), suppress glucagon release (which further lowers blood sugar), slow gastric emptying (which extends satiety), and act on the brain to reduce appetite.
The third effect, slowed gastric emptying, is both the therapeutic mechanism that drives weight loss and the potential source of the most common litigation injury. The drugs are designed to keep food in the stomach longer, making you feel full. But in some patients, "longer" becomes "indefinitely," and therapeutic slowing crosses into pathological paralysis.
This is why the timeline of appetite suppression matters. If appetite suppression arrives very quickly and very intensely, it may indicate that gastric slowing has gone beyond therapeutic levels. Gradual onset through proper dose titration is designed to find the balance between effective appetite reduction and excessive gastric stalling.
The NAION mechanism question
The connection between GLP-1 agonists and NAION is less mechanistically clear than the gastroparesis link. NAION occurs when blood flow to the optic nerve is disrupted, but the exact pathway by which GLP-1 drugs might cause this disruption is still under investigation. Some researchers hypothesize that rapid weight loss could alter blood pressure dynamics and affect small vessel perfusion. Others suggest that GLP-1 receptor activation might directly affect vascular tone in the optic nerve blood supply.
What the studies show clearly is a statistical association. The risk is elevated. The correlation is consistent across multiple studies and populations. Whether GLP-1 drugs cause NAION directly or through an intermediate mechanism, the practical implication for researchers remains the same: vision monitoring during any GLP-1 protocol is prudent, and any sudden vision changes require immediate medical evaluation.
Risk factors that may increase vulnerability
Not everyone on GLP-1 agonists develops serious side effects. The litigation data and underlying research suggest that certain factors may increase vulnerability. For gastroparesis, pre-existing delayed gastric emptying (even subclinical), rapid dose escalation, higher doses, concurrent use of other medications that slow gut motility, and diabetes-related autonomic neuropathy may all contribute to higher risk.
For NAION, the JAMA Ophthalmology study noted that the risk was even higher in the overweight/obese weight loss population (7.64x) than in the diabetes population (4.28x), suggesting that the weight loss indication may carry a higher eye-related risk profile than the diabetes indication. Risk factors like small optic disc size, hypertension, diabetes, and sleep apnea are known NAION risk factors that may compound the drug-related risk.
This understanding should inform protocol decisions. Researchers with pre-existing GI conditions should approach semaglutide with extra caution. Those with known cardiovascular risk factors should be particularly attentive to visual symptoms.
The manufacturer defense and what it reveals
The defense strategies employed by Novo Nordisk and Eli Lilly provide insight into how the manufacturers view their own products' safety profiles. Understanding their arguments is as informative as understanding the plaintiffs' claims.
The "adequate warning" defense
Manufacturers argue that their labels did warn about gastrointestinal side effects, including nausea, vomiting, diarrhea, and abdominal pain. They contend that these warnings were adequate to inform prescribing physicians, who are expected to monitor patients for adverse effects.
Plaintiffs counter that mentioning common GI side effects is fundamentally different from warning about gastroparesis, a chronic condition that can persist indefinitely and require lifelong management. The distinction between "you may experience nausea" and "this drug may paralyze your stomach" is significant both medically and legally.
The pre-existing condition argument
A core defense strategy involves attributing plaintiffs' injuries to their underlying health conditions rather than the medications. Diabetes itself can cause gastroparesis through autonomic neuropathy. Hypertension and high cholesterol are NAION risk factors independent of GLP-1 use.
This argument has some scientific merit, which is precisely why the Sodhi JAMA study was so impactful. By focusing on non-diabetic weight loss patients, it eliminated the confounding variable of diabetes-related GI complications and still found dramatically elevated risks. The defense argument works less well against plaintiffs who had no pre-existing conditions that could explain their injuries.
The multifactorial causation argument
Manufacturers also argue that even when injuries occur in GLP-1 users, multiple factors may contribute, making it impossible to attribute the injury solely to their drug. This is a standard defense in pharmaceutical litigation, but it faces challenges when studies show 4-9x risk increases, because such dramatic elevations are difficult to explain away as coincidence or confounding.
What researchers are watching for next
Several upcoming developments will shape the trajectory of GLP-1 litigation and its implications for the research community.
Bellwether trial selection and outcomes
The selection of bellwether cases will determine which specific injuries, which drugs, and which plaintiff profiles go before a jury first. Strong plaintiff verdicts will increase settlement pressure and case values. Defense verdicts will slow the litigation and potentially reduce filing rates. The first trials, whenever they occur, will set the tone for the entire litigation.
New safety data
Research continues to emerge on GLP-1 safety. The February 2026 JAMA Ophthalmology data reporting 14.5 NAION cases per 100,000 person-years in semaglutide users adds to the evidence base. Each new study either strengthens or weakens the litigation. Researchers should monitor publications in JAMA, The Lancet, and specialty journals for updated safety analyses.
Regulatory changes
The FDA and international regulators may issue additional warnings or restrictions based on accumulating data. Any new label changes will be cited in court proceedings and could either support or undermine existing claims. Watch for updates from the FDA, EMA (European Medicines Agency), and WHO.
Compounding pharmacy outcomes
The parallel litigation against compounders will determine the future availability of affordable GLP-1 alternatives. If manufacturers succeed in shutting down compounded versions, it will consolidate the market and affect pricing, access, and researcher options for compounds like semaglutide and tirzepatide.
Practical safety guidance for GLP-1 researchers
Based on the totality of evidence emerging from the litigation, the underlying studies, and regulatory actions, researchers working with GLP-1 compounds should consider the following practical guidelines.
Before starting any GLP-1 protocol
Get a baseline assessment of gastrointestinal function. If you already experience slow digestion, bloating, or gastroparesis symptoms, GLP-1 agonists could significantly worsen these issues. Know your starting point. Consider a baseline eye examination, particularly if you have risk factors for NAION (diabetes, hypertension, sleep apnea, small optic disc). Review your complete medication list for other drugs that slow GI motility, including certain antidepressants, opioids, and anticholinergics. Combining these with GLP-1 agonists could compound the gastroparesis risk.
During a GLP-1 protocol
Follow conservative dosing guidelines. The temptation to escalate quickly exists, but the litigation data suggests that patience with titration may be protective. Monitor for warning signs that go beyond typical adjustment symptoms. Persistent nausea lasting more than a few days, inability to eat normal amounts, feeling of food "sitting" in the stomach, and any vision changes all warrant medical evaluation.
Stay hydrated. Many of the kidney injury claims stem from dehydration caused by GI side effects. If you are experiencing significant nausea or vomiting, aggressive hydration is essential, and persistent vomiting requires medical attention. Track everything. Use tracking tools to document your protocol, doses, timing, side effects, and any changes in how you feel. This documentation helps you identify trends early and provides a clear record if you need medical consultation.
When to seek immediate medical attention
The following symptoms, based on the injuries being litigated, should prompt immediate medical evaluation and not a wait-and-see approach. Severe abdominal pain, especially upper abdominal pain radiating to the back, which could indicate pancreatitis. Inability to keep food or liquids down for more than 24 hours, suggesting possible gastroparesis or obstruction. Any sudden change in vision, particularly painless vision loss in one eye, which could indicate NAION. Severe constipation with abdominal distension and cramping, potentially indicating bowel obstruction. Signs of significant dehydration such as dark urine, dizziness, or rapid heart rate.
For researchers serious about optimizing their peptide protocols safely, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, safety databases, proven protocols, and a community of thousands who have navigated these exact questions. Understanding the risks is the first step. Having access to expert guidance and community experience is what turns that understanding into safer practice.
Frequently asked questions
How many GLP-1 lawsuits have been filed?
As of February 2026, over 3,191 lawsuits have been filed in the federal multidistrict litigation (MDL 3094) in the Eastern District of Pennsylvania. Additional cases are pending in state courts across New Jersey, Texas, Delaware, and Indiana. The total number continues to grow with approximately 200 new cases filed monthly. The litigation targets both Novo Nordisk and Eli Lilly for products including Ozempic, Wegovy, Mounjaro, and related medications.
What side effects are involved in GLP-1 lawsuits?
The primary injuries being litigated include gastroparesis (stomach paralysis), non-arteritic anterior ischemic optic neuropathy (NAION) causing vision loss, bowel obstruction, ileus, pancreatitis, acute kidney injury, and gallbladder disease. Gastroparesis and NAION represent the largest categories of claims, with vision loss cases gaining particular legal momentum due to the permanent nature of the injury.
Have there been any GLP-1 lawsuit settlements yet?
No global settlements have been announced as of February 2026. The litigation is still in the expert discovery and pre-trial phase, with bellwether trial selections expected in mid-2026. Legal analysts project meaningful settlements are unlikely before late 2026 at the earliest. Projected settlement ranges vary from $50,000 for temporary gastroparesis to over $1,000,000 for bilateral vision loss cases.
Who can file a GLP-1 lawsuit?
Individuals who were prescribed a GLP-1 receptor agonist (Ozempic, Wegovy, Mounjaro, Zepbound, Trulicity, Saxenda, Victoza, or Rybelsus) and subsequently developed serious side effects supported by medical documentation may be eligible. Strong claims require objective diagnostic evidence such as gastric emptying studies for gastroparesis or ophthalmological testing for NAION, clear temporal connection between drug use and symptom onset, and documented medical treatment for the condition.
Does the GLP-1 litigation affect compounded semaglutide and tirzepatide?
The patient injury litigation does not directly affect compounded products, but a parallel legal battle does. Eli Lilly and Novo Nordisk have filed separate lawsuits against compounding pharmacies and telehealth platforms, while compounders have filed antitrust counterclaims. The resolution of these cases will significantly impact the availability and affordability of compounded GLP-1 options.
What studies support the GLP-1 lawsuit claims?
Key studies include the Sodhi et al. JAMA study (October 2023) showing 3.67x gastroparesis risk, 9.09x pancreatitis risk, and 4.22x bowel obstruction risk in GLP-1 users. The JAMA Ophthalmology study found 4.28x to 7.64x higher NAION risk in semaglutide users. Additional studies from the Netherlands, Sweden, and the University of Toronto have reinforced these findings across different populations.
Should I stop taking my GLP-1 medication because of these lawsuits?
This guide provides informational context about the litigation, not medical advice. Any decisions about medication changes should be made with your healthcare provider based on your individual circumstances. The litigation data does highlight specific risks worth monitoring, and the studies cited here provide the quantitative context for those discussions. For researchers following GLP-1 protocols, understanding these risks enables more informed decision-making about continuation or discontinuation.
Will the GLP-1 lawsuits lead to these drugs being pulled from the market?
This is unlikely. The drugs have demonstrated significant therapeutic benefits for diabetes management and weight loss. The more probable outcome is updated and expanded warning labels, mandatory patient information requirements, and financial settlements. The litigation is fundamentally about adequate warnings and informed consent, not about whether the drugs should exist. Similar patterns have played out with other pharmaceutical products that remained on the market with updated safety information.
External resources
U.S. District Court, Eastern District of Pennsylvania, MDL 3094
JAMA Ophthalmology: Risk of NAION in Patients Prescribed Semaglutide
In case I do not see you, good afternoon, good evening, and good night. May your research stay informed, your protocols stay safe, and your decisions stay grounded in evidence.