Apr 2, 2026

Before you draw up another dose, look at the vial. Really look at it. Because tirzepatide does go bad, and the consequences of using degraded peptide range from wasted money to zero therapeutic effect to, in rare contamination scenarios, genuine health risk. The tricky part is that degradation does not always announce itself with obvious visual cues. Sometimes the solution looks perfectly clear while the active molecule has already lost 40 to 60 percent of its receptor binding affinity due to oxidation at the molecular level.
That is what makes this question so important. Most people think about expiration in terms of a date printed on a label. But tirzepatide is a 39-amino-acid peptide with a C20 fatty acid chain, and it follows the rules of chemistry, not calendars.
Heat, light, mechanical agitation, bacterial contamination, and improper reconstitution can all push a perfectly good vial past the point of usefulness long before any printed date arrives. Understanding the difference between a date and actual degradation is what separates informed researchers from those quietly injecting expensive water.
This guide covers every way tirzepatide can go bad. The visible signs you can catch with your eyes. The invisible degradation you cannot. The storage mistakes that accelerate breakdown. The timelines for every formulation type. And the exact moment when you should stop using a vial and start fresh. SeekPeptides members already know that peptide handling is half the battle, and this article gives you the complete picture so nothing gets wasted and nothing gets used past its prime.
How tirzepatide degrades at the molecular level
Peptides are fragile molecules. They are not like aspirin tablets sitting in a medicine cabinet for years. Tirzepatide is a dual GIP and GLP-1 receptor agonist built from a specific sequence of 39 amino acids, and each of those amino acid residues is vulnerable to chemical modification under the wrong conditions. When researchers talk about tirzepatide going bad, they are describing changes at this molecular level that reduce or eliminate the peptide ability to bind its target receptors and produce therapeutic effects.
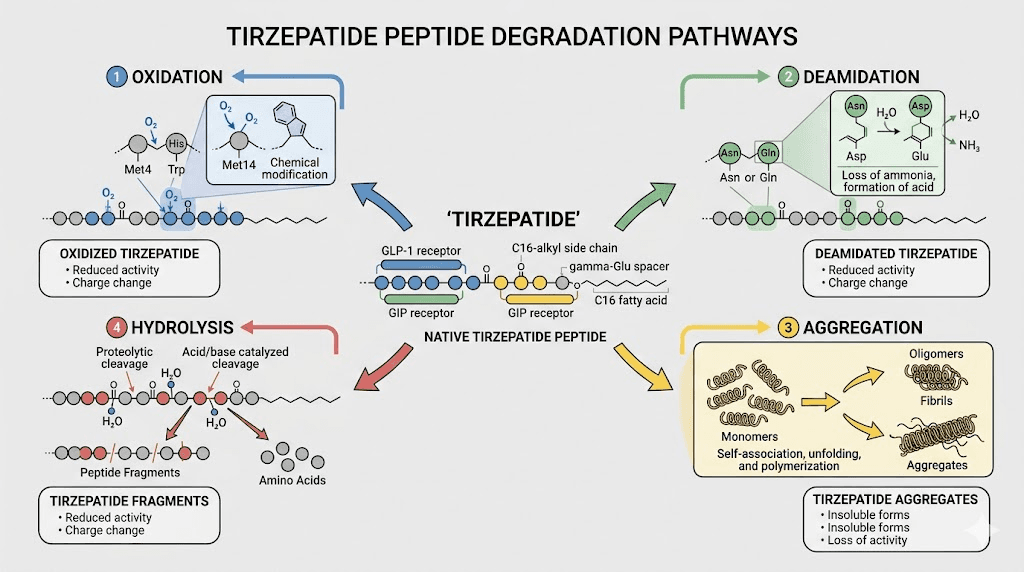
The primary degradation pathways include oxidation, deamidation, aggregation, and hydrolysis. Each attacks the molecule differently.
Oxidation
Methionine residues in the peptide chain are especially vulnerable. Exposure to oxygen, light, or certain metals can oxidize these residues, converting them to methionine sulfoxide. A study on GLP-1 receptor agonist stability found that oxidation of a single methionine residue reduced receptor binding affinity by 40 to 60 percent. That is not a subtle decline. That is the difference between a dose that works and one that barely registers.
The worst part? Oxidation can happen without any visible change to the solution. The vial looks exactly the same. Clear, colorless, perfectly normal. But the molecule inside has been chemically altered in ways that make it significantly less effective. This is why proper storage matters more than most people realize, and why tirzepatide refrigeration is not optional.
Deamidation
Asparagine and glutamine residues can undergo deamidation, where an amide group is converted to a carboxylic acid. This changes the charge and shape of the peptide. Deamidation accelerates at higher temperatures and at pH levels outside the optimal range. The process is slow at refrigerated temperatures but speeds up dramatically at room temperature, roughly doubling in rate for every 10 degree Celsius increase.
Aggregation
This is the one you can sometimes see. When peptide molecules unfold and stick together, they form aggregates. Small aggregates are invisible. Large aggregates appear as cloudiness, visible particles, or a gel-like substance floating in the solution. Aggregated tirzepatide is not only less potent but potentially immunogenic, meaning it could trigger an immune response that the intact molecule would not.
Aggregation is accelerated by temperature fluctuations, mechanical shaking, and freeze-thaw cycles. This is exactly why freezing tirzepatide is such a bad idea. The ice crystals that form during freezing physically damage the peptide structure, and thawing does not reverse that damage.
Hydrolysis
Water itself can break peptide bonds over time, especially at elevated temperatures. The backbone of the peptide chain literally splits apart. Hydrolysis products are smaller fragments that have no therapeutic activity. This process is why even properly stored tirzepatide has a finite shelf life. Eventually, enough bonds break that the remaining intact molecules cannot produce adequate receptor activation.
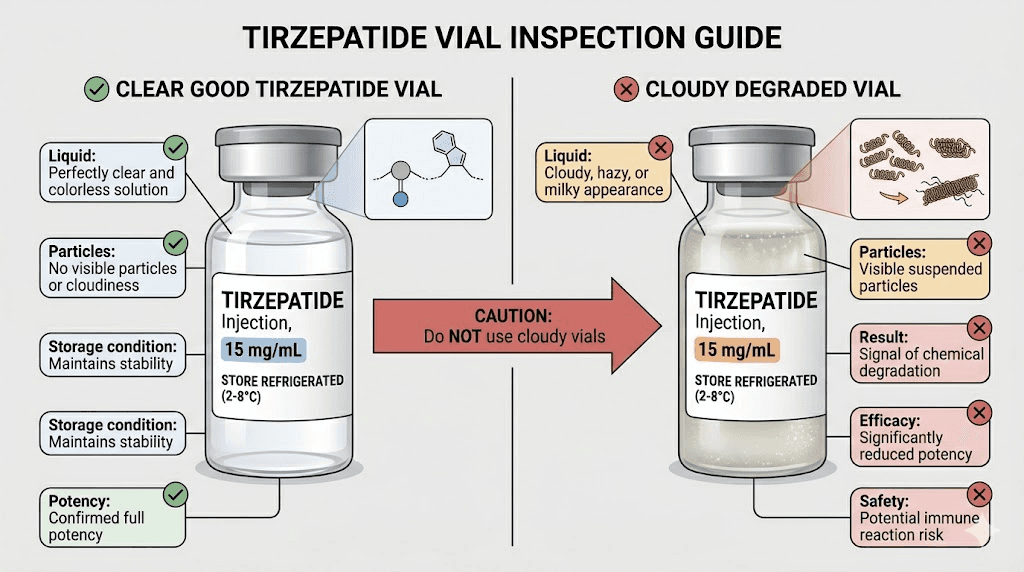
Visible signs that tirzepatide has gone bad
Some degradation is invisible. But some is not. Learning to recognize the visual warning signs can save you from using a compromised vial. Fresh tirzepatide should be a specific color depending on its formulation, and any deviation from that baseline is cause for concern.
Cloudiness or turbidity
Properly reconstituted tirzepatide should be clear. Not slightly hazy. Not mostly clear. Completely clear. If you hold the vial up to a light source and see any cloudiness, haziness, or opalescence, that is protein aggregation. The peptide molecules have unfolded and clumped together. Do not use it.
There is no recovering from aggregation. You cannot shake it back into solution. You cannot warm it up and make it clear again. Once proteins aggregate, the structural damage is permanent. The vial is done.
Visible particles or floaters
Tiny specks, fibers, or floating debris in the solution indicate either contamination or advanced aggregation. Sometimes these are visible only when you hold the vial against a dark background and look carefully. If you see any particles at all, the vial should be discarded.
One important distinction. Immediately after reconstitution, you might see tiny bubbles from the mixing process. Bubbles rise and disappear within minutes. Particles do not. If the specks persist after the vial has sat undisturbed for 10 to 15 minutes, those are not bubbles.
Color changes
Brand-name tirzepatide in pre-filled pens is typically colorless to slightly yellow. Compounded tirzepatide varies depending on additives. Tirzepatide with B12 has a pink or reddish tint from the cyanocobalamin. Standard compounded tirzepatide without additives should be clear and colorless after reconstitution.
Any unexpected yellowing, browning, or darkening suggests oxidative degradation. If your normally colorless solution has developed a yellow tint, that is a chemical change at the molecular level. If your tirzepatide with B12 formulation has changed from pink to brown or dark red, the B12 component may have degraded as well.
Unusual smell
Peptide solutions should not have a strong or unpleasant odor. A foul or unusual smell suggests bacterial contamination. This is rare with proper handling but possible if sterile technique was compromised during reconstitution or if the vial rubber stopper was damaged.
Changes in viscosity
If the solution feels thicker than usual when drawing it into a syringe, or if it looks syrupy compared to how it normally behaves, aggregation or contamination may be occurring. Fresh reconstituted tirzepatide has a viscosity very close to water.
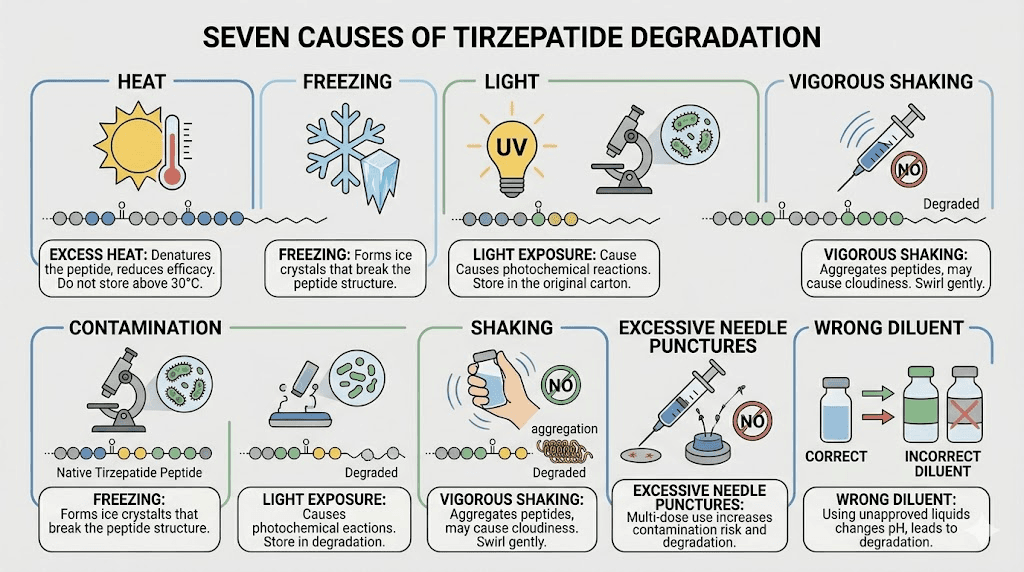
The seven most common causes of tirzepatide going bad
Tirzepatide does not spontaneously degrade for no reason. Something causes it. And in nearly every case, that something is preventable. Here are the seven factors that most commonly push tirzepatide from good to bad, ranked by how frequently they cause problems.
1. Temperature exposure above 30 degrees Celsius
Heat is the single biggest killer of peptide potency. Every chemical reaction that degrades tirzepatide, oxidation, deamidation, hydrolysis, aggregation, accelerates with increasing temperature. The relationship is roughly exponential. A vial left at 40 degrees Celsius degrades roughly four times faster than one kept at 25 degrees, and a vial at 25 degrees degrades roughly four times faster than one at 4 degrees.
This is why leaving tirzepatide out of the fridge is such a concern. At room temperature around 20 to 25 degrees Celsius, the degradation clock moves much faster. And if a vial gets left in a hot car, near a window in direct sunlight, or in a package sitting on a porch in summer heat, the damage can be severe and irreversible within hours.
When tirzepatide gets warm, the damage is cumulative. Even brief exposures add up. A vial that spent 30 minutes at high temperature on three separate occasions has accumulated significant thermal stress, even if it was properly refrigerated between those exposures.
2. Freezing
Just as damaging as heat, but through a completely different mechanism. When water freezes, it forms ice crystals. Those ice crystals physically pierce and tear apart protein structures. The peptide molecules are literally shredded at the microscopic level.
Thawing does not reverse this damage. The fragments cannot reassemble into functional molecules. Frozen tirzepatide should be considered destroyed, regardless of how it looks after thawing. The solution might appear perfectly clear because the fragments are too small to see, but the therapeutic molecule no longer exists in its functional form.
This is why storing vials in the refrigerator door is risky. The door temperature fluctuates every time it opens and closes, and in some refrigerators, the door compartment can dip below freezing near the back wall.
3. Light exposure
Ultraviolet and visible light provide energy that drives photochemical degradation reactions. Tryptophan residues in the peptide chain are particularly susceptible to photo-oxidation. The process generates reactive oxygen species that then attack other parts of the molecule in a cascade effect.
Compounding pharmacies typically ship tirzepatide in amber vials or opaque packaging for this reason. But once the vial is in your refrigerator, it gets exposed to light every time you open the door. This is usually not enough to cause significant degradation over the life of a single vial, but combined with other stress factors, it contributes.
The solution is simple. Store vials in their original box or wrap them in aluminum foil. And never leave a vial sitting on a countertop in daylight.
4. Contamination during reconstitution
Every time a needle pierces the rubber stopper, there is an opportunity for bacteria to enter the vial. Proper reconstitution technique minimizes this risk, but it never eliminates it entirely. Using non-sterile technique, reusing needles, touching the stopper without alcohol swabbing, or reconstituting in an unclean environment all increase contamination risk.
Bacterial contamination is different from chemical degradation. Bacteria metabolize the peptide as a food source, producing waste products that change the solution pH, color, and smell. Contaminated vials can become dangerous, not just ineffective. If you suspect bacterial contamination, discard the vial immediately.
Using bacteriostatic water for reconstitution helps prevent contamination because it contains 0.9% benzyl alcohol, which inhibits bacterial growth. Sterile water lacks this preservative and should ideally be used for single-dose vials only.
5. Mechanical agitation
Shaking a vial of reconstituted tirzepatide is one of the fastest ways to cause aggregation. The mechanical forces at the air-liquid interface denature the peptide molecules, exposing hydrophobic regions that then stick together. This is the same process that happens when you whip egg whites into a foam, the proteins unfold and aggregate.
Always swirl gently. Never shake. When mixing peptides with bacteriostatic water, direct the stream of water down the side of the vial and let the peptide dissolve gradually. If the powder is not fully dissolving, let it sit for a few minutes. It will dissolve on its own. Vigorous shaking to speed up the process will cost you potency.
6. Repeated needle punctures
Each time a needle goes through the rubber stopper, it removes a tiny core of rubber and potentially introduces contaminants. Multi-dose vials are designed for a limited number of punctures. After 20 to 25 punctures, the stopper integrity is compromised, and the sterility barrier may be broken.
This is one reason why vial use timelines matter. Even if the medication has not chemically degraded, a stopper with too many holes is a contamination risk. Using the smallest gauge needle practical for drawing up doses reduces stopper damage.
7. Using the wrong reconstitution solution
The reconstitution solution matters more than most people think. Using regular water, tap water, saline at the wrong concentration, or expired bacteriostatic water can introduce contaminants or create a solution pH that accelerates degradation. Always use fresh, unexpired bacteriostatic water at the correct volume.
How long before tirzepatide goes bad by formulation type
The timeline for tirzepatide going bad depends entirely on which formulation you have and how it is stored. There is no single answer to this question because the starting stability varies dramatically between brand-name pens, compounded liquid vials, and lyophilized powder.
Brand-name tirzepatide pens
The FDA-approved product has undergone extensive stability testing. Unopened pens stored in the refrigerator at 2 to 8 degrees Celsius remain viable until the printed expiration date, which is typically 18 to 24 months from manufacture. Once removed from refrigeration, an unopened pen is good for 21 days at room temperature below 30 degrees Celsius.
Once a pen is first used, the 21-day clock starts regardless of storage method. Even if you put it back in the fridge after each use, you have 21 days from first injection. This is because the injection mechanism introduces a tiny air path that compromises the sealed environment.
Compounded liquid tirzepatide
This is where things get more complicated. Compounded tirzepatide expiration follows different rules. The pharmacy assigns a Beyond-Use Date, typically 28 to 90 days from preparation. This date is shorter than brand-name expiration because compounded products do not undergo the same extensive long-term stability testing.
Once the rubber stopper is first punctured, most pharmacies recommend using the vial within 28 days. Some formulations may have shorter windows. Always check the specific guidance from your compounding pharmacy. Compounded tirzepatide shelf life varies significantly between pharmacies because different formulations and preservative systems produce different stability profiles.
Lyophilized tirzepatide powder
Freeze-dried powder is the most stable form. Lyophilized tirzepatide has no water present, which means most degradation pathways are dramatically slowed. Properly stored lyophilized powder can last months to years, depending on storage temperature.
The critical moment is reconstitution. Once you add bacteriostatic water, the stability clock resets to the reconstituted timeline, typically 28 days refrigerated. Peptides in powder form are always more stable than their reconstituted counterparts.
Timeline comparison table
Formulation | Refrigerated (2-8 C) | Room temp (below 30 C) | After first use |
|---|---|---|---|
Brand-name pen (unopened) | Until printed expiration | 21 days | 21 days regardless |
Brand-name pen (opened) | 21 days | 21 days | 21 days from first injection |
Compounded liquid | 28-90 days (pharmacy BUD) | 21-30 days | 28 days typical |
Lyophilized powder (dry) | Months to years | Months (reduced) | N/A until reconstituted |
Reconstituted from powder | 28 days | Not recommended | 28 days from reconstitution |
These are general guidelines. Always defer to the specific instructions provided by the manufacturer or compounding pharmacy. And remember, these timelines assume proper storage the entire time. A vial that was left out on a hot counter for a day does not get its full refrigerated timeline back just because you put it back in the fridge.
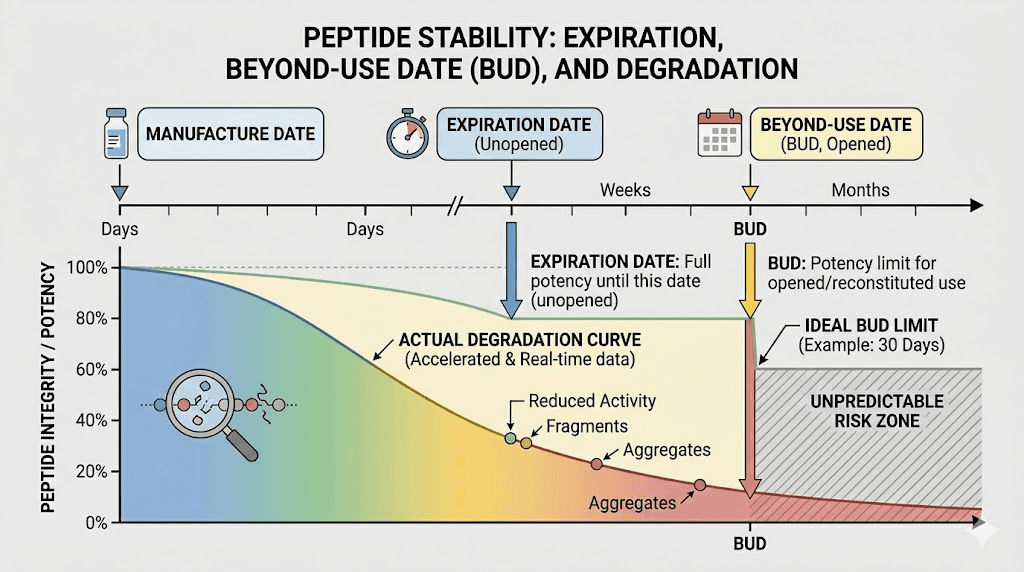
The difference between expiration and degradation
This is a distinction that trips people up constantly. Tirzepatide expiration dates are not the same as degradation. Expiration is a legal and regulatory concept. Degradation is a chemical reality. Understanding the gap between them helps you make better decisions about your medication.
What expiration dates actually mean
An expiration date is the last date at which the manufacturer guarantees the product meets its labeled potency specification, typically 90 to 110 percent of the stated dose. It does not mean the product becomes poison at midnight on that date. It does not mean the product is suddenly ineffective. It means the manufacturer will no longer guarantee its quality beyond that point.
For brand-name products, expiration dates are based on extensive accelerated and real-time stability studies. These are rigorous tests that track degradation products, potency, pH, sterility, and particulate matter over months to years. The assigned date includes a safety margin.
What actually happens past the date
Degradation is a continuous process. A vial at 99 percent potency on the expiration date does not drop to 50 percent the next day. It might be at 95 percent a week later and 90 percent a month later. The decline is gradual. Using expired tirzepatide does not carry a dramatic safety risk in most cases, but it does carry an efficacy risk. You might be getting 80 percent of the intended dose, or 60 percent, or 40 percent. Without analytical testing, you have no way to know.
The real risk is not toxicity. It is unpredictable dosing. If you are on a carefully titrated tirzepatide dosing protocol, using a partially degraded vial means your actual dose is lower than what you calculated. This can stall progress, lead to tirzepatide seeming like it stopped working, or cause confusing inconsistencies in your response.
Beyond-Use Dates for compounded products
Compounded medications use Beyond-Use Dates instead of expiration dates. The BUD is typically much shorter because compounded products have not undergone the same level of stability testing. A compounding pharmacy might assign a 60-day BUD based on limited stability data, even though the product might remain viable for longer.
This does not mean the product is bad on day 61. But it does mean you are in uncharted territory. The pharmacy cannot guarantee anything about the product past that date. For most people, adhering to the BUD is the wisest approach. The cost of a new vial is far less than the cost of using degraded medication for weeks without realizing it.
How storage conditions affect how fast tirzepatide goes bad
The single biggest variable in how quickly tirzepatide degrades is temperature. Everything else, light, contamination, mechanical stress, matters too. But temperature is the master variable that controls the rate of every chemical degradation pathway.
Refrigerated storage (2 to 8 degrees Celsius)
This is the gold standard. At proper refrigeration temperatures, all degradation reactions proceed at their slowest practical rate. Tirzepatide lasts longest in the fridge, which is why every manufacturer and compounding pharmacy lists refrigerated storage as the primary recommendation.
The ideal spot is the middle shelf toward the back of the refrigerator. Not the door. Not the top shelf near a freezer vent. Not the bottom near the crisper where temperatures fluctuate. The middle-back position maintains the most consistent temperature with the least variation from door openings.
Room temperature storage (20 to 25 degrees Celsius)
At room temperature, degradation rates roughly quadruple compared to refrigerated storage. A vial with 28 days of refrigerated life might have only 7 to 10 days at room temperature before reaching the same level of degradation. The 21-day room temperature limit for brand-name pens reflects this accelerated timeline.
Room temperature excursions are inevitable during travel, during injection prep, and during shipping. Brief excursions of 15 to 30 minutes are negligible. But cumulative exposure matters. If you routinely leave the vial out for an hour during each injection session and inject three times per week, that is three hours of room temperature exposure per week, nearly half a day per month of accelerated degradation.
Elevated temperature (above 30 degrees Celsius)
Above 30 degrees, degradation accelerates dramatically. At 40 degrees Celsius, a vial can lose significant potency within hours to days. At 50 degrees, which a package or car interior can easily reach in summer, degradation can be severe within an hour.
This is why shipping and transit are critical moments for tirzepatide stability. Traveling with tirzepatide requires insulated containers and cold packs. A vial that arrives warm after shipping may have already lost a meaningful percentage of its potency, even if it looks perfectly normal.
Temperature cycling
Repeated cycles of warming and cooling are particularly damaging. Each cycle stresses the protein structure. The peptide partially unfolds during warming, then does not always refold correctly during cooling. Over many cycles, the cumulative damage from misfolding and aggregation becomes significant.
This is another reason why the refrigerator door is a bad storage location. Every time you open the fridge, the door temperature rises. When you close it, the temperature drops. A vial stored in the door might experience 10 to 20 temperature cycles per day in a busy household.
Can you tell if tirzepatide has lost potency without visible changes?
Here is the hard truth. No. Not without laboratory testing.
A vial of tirzepatide can lose 30, 40, even 50 percent of its potency through oxidation and deamidation while remaining perfectly clear, colorless, and particle-free. The degradation products are soluble. They are invisible. They do not change the smell or feel of the solution. Only analytical methods like high-performance liquid chromatography can quantify the remaining intact peptide.
This reality is exactly why proper storage is so important. You cannot rely on visual inspection alone to verify potency. You have to prevent degradation proactively through correct handling, because you cannot detect it retroactively through observation.
Indirect signs of potency loss
While you cannot see molecular degradation, you might notice its effects. If your tirzepatide suddenly seems less effective, with reduced appetite suppression, less weight loss progress, or return of hunger between doses, and nothing else in your protocol has changed, degraded medication is one possible explanation.
Of course, there are many other reasons why tirzepatide might seem to stop working, including metabolic adaptation, dietary changes, or reaching a plateau. But if the change coincides with a vial that was exposed to heat, has been open for a long time, or is past its Beyond-Use Date, degradation should be high on your list of suspects.
When to suspect degradation even without visible signs
Consider your vial potentially compromised if any of the following apply:
It was left out of the fridge for more than 4 hours total
It was exposed to temperatures above 30 degrees Celsius
It was accidentally frozen, even briefly
It has been open for more than 28 days
It is past the printed Beyond-Use Date or expiration
It was shipped without cold packs in warm weather
Your results suddenly declined with no other explanation
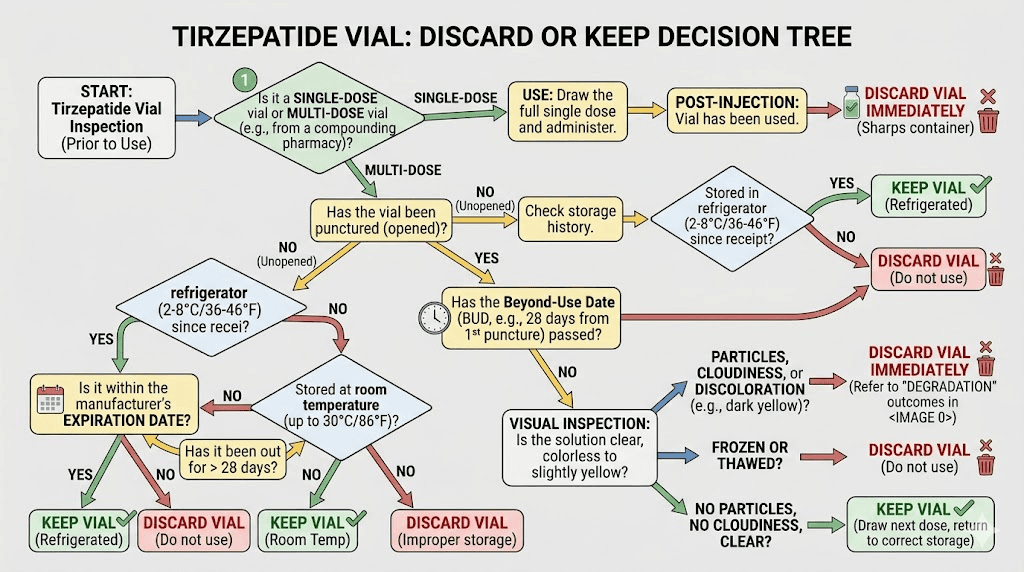
What happens if you use tirzepatide that has gone bad
The consequences of using degraded tirzepatide fall into three categories, and the severity depends on the type and extent of degradation.
Reduced efficacy
This is the most common outcome and the least dangerous. Degraded tirzepatide simply does not work as well. If the peptide has lost 30 percent of its potency through oxidation, you are effectively taking a 30 percent lower dose than intended. For someone on a specific tirzepatide dose in units, this means the calculated dose no longer matches reality.
Reduced efficacy manifests as return of appetite, slower weight loss, or a feeling that the medication has stopped working. It can take weeks to recognize this pattern, during which time you have been using suboptimal medication and potentially stalling your protocol. If you have been tracking your progress and notice a sudden change, compare it against typical tirzepatide timelines to see if your experience has deviated.
Unpredictable dosing
When a vial is partially degraded, each dose contains an unknown amount of active peptide. One draw might contain mostly intact molecules from a well-mixed region. Another might pull from a pocket of more degraded solution. This creates inconsistent dosing from injection to injection, which makes it impossible to properly titrate or adjust your protocol.
Unpredictable dosing is particularly problematic when using a tirzepatide dosage calculator to measure precise amounts. The calculator assumes the stated concentration is accurate. If degradation has reduced the actual concentration, every calculation is off.
Potential immune reactions from aggregated peptide
This is the most concerning scenario, though it is relatively rare. Aggregated proteins can be recognized by the immune system as foreign. This can trigger injection site reactions like redness, swelling, and itching that are more severe than normal. In extreme cases, repeated injection of aggregated protein could theoretically cause the body to develop antibodies against the peptide, which would reduce or eliminate its effectiveness even with fresh, non-degraded doses.
This is why visible cloudiness or particles are absolute discard triggers. The risk is not worth it. A new vial costs money. An immune reaction to aggregated peptide costs much more in terms of both health and long-term treatment viability.
The complete tirzepatide storage guide to prevent going bad
Prevention is always better than detection. Here is the complete protocol for storing tirzepatide to maximize its useful life and prevent degradation.
Primary storage
Refrigerate at 2 to 8 degrees Celsius. Place the vial in the middle of a shelf, away from the back wall (which can sometimes be colder and risk freezing) and away from the door. Keep it in its original box or wrap in aluminum foil to protect from light. Store upright to minimize the surface area exposed to the rubber stopper.
During injection prep
Remove the vial from the refrigerator. Swab the stopper with an alcohol wipe. Draw your dose. Return the vial to the refrigerator immediately. Total time out of the fridge should be under 5 minutes. Do not leave the vial sitting on the counter while you eat breakfast, shower, or do other tasks.
During travel
Traveling with tirzepatide requires an insulated container with a cold pack. The vial should never sit in direct sunlight, in a hot car, or in checked luggage that might be exposed to extreme temperatures in an airplane cargo hold. Use a medical-grade cooler designed for peptide transport.
Reconstitution best practices
When reconstituting tirzepatide, use fresh bacteriostatic water that has not expired. Direct the water stream down the side of the vial, not directly onto the powder. Do not shake. Swirl gently or let it sit for 5 to 10 minutes to dissolve. Once reconstituted, refrigerate immediately and write the date on the vial so you can track the 28-day window.
Multi-dose vial management
Use the smallest needle gauge practical for drawing doses to minimize stopper damage. Always use a new needle for each draw. Proper syringe technique reduces the risk of contamination. If you are getting close to the end of a vial and the remaining amount will not be used within the 28-day window, it is better to discard the remainder and start a new vial than to stretch an old one.
Monitoring checklist
Before every injection, perform a quick visual check. Hold the vial up to a light source. Look for cloudiness, particles, or color changes. Check the date, both the Beyond-Use Date and the date you first punctured the stopper. If anything looks off or the dates have passed, discard and use a fresh vial.
Compounded versus brand-name tirzepatide stability differences
Not all tirzepatide is created equal when it comes to shelf stability. The formulation, manufacturing process, and quality control all affect how quickly the product degrades.
Brand-name advantages
FDA-approved tirzepatide benefits from pharmaceutical-grade manufacturing, extensive stability testing, optimized excipient formulations, and hermetically sealed delivery systems. The pre-filled pen design minimizes exposure to air, light, and contamination. The buffer system is specifically engineered to maintain optimal pH for maximum stability.
These advantages translate into longer shelf life and more predictable degradation profiles. When the label says good until a specific date, you can trust it, assuming proper storage.
Compounded considerations
Compounded tirzepatide from a reputable 503B pharmacy can be high quality, but it faces inherent limitations. The raw peptide may come from different synthesis sources. The buffer system, preservatives, and excipients vary between pharmacies. Quality control testing is less extensive than FDA-mandated testing.
This does not mean compounded tirzepatide is bad. It means the stability profile is less well-characterized. The Beyond-Use Date is an educated estimate rather than a guarantee backed by years of data. Choosing a reputable 503B pharmacy matters enormously because their quality standards directly affect how long your product remains potent.
Lyophilized versus liquid stability
If stability is your primary concern, lyophilized tirzepatide offers significant advantages. Without water present, oxidation, deamidation, and hydrolysis are all dramatically slowed. A properly stored vial of lyophilized tirzepatide can maintain its potency for far longer than a pre-reconstituted liquid formulation.
The tradeoff is convenience. Lyophilized product requires reconstitution before each use period, which adds a step, requires bacteriostatic water, and introduces a contamination opportunity. For people who prioritize stability and do not mind the extra preparation, lyophilized is often the better choice.
Frequently asked questions
Does tirzepatide go bad if left out overnight?
One night at room temperature (approximately 8 to 12 hours at 20 to 25 degrees Celsius) will not ruin the vial, but it does accelerate degradation. The key factor is the total cumulative time the vial has spent out of the fridge. A single overnight exposure reduces the remaining usable life by roughly the equivalent of 2 to 4 days of refrigerated storage. Return it to the fridge immediately and use it within a shorter window than originally planned. If the room was hot (above 30 degrees), err on the side of discarding. Check our guide on how long tirzepatide can be out of the fridge for specific timelines.
Can I still use tirzepatide if it looks cloudy?
No. Cloudiness indicates protein aggregation, which is irreversible. Aggregated tirzepatide has reduced potency and could potentially trigger immune reactions. Discard the vial and use a fresh one.
Does tirzepatide go bad faster after the first needle puncture?
Yes. Each needle puncture introduces a small amount of air and potential contaminants. The first puncture breaks the sealed environment, starting the countdown. Most guidelines recommend using a punctured vial within 28 days, even if the overall Beyond-Use Date extends further.
How can I tell if my tirzepatide lost potency?
You cannot tell by looking at it if the degradation is molecular (oxidation, deamidation). You can only detect visible aggregation (cloudiness, particles) or contamination (color change, smell). If you suspect invisible potency loss, the only definitive answer comes from analytical testing. For practical purposes, adhere strictly to storage guidelines and use-by dates. If your weight loss timeline suddenly stalls with no other explanation, degraded medication is worth investigating.
Is slightly expired tirzepatide safe to use?
Generally, tirzepatide does not become toxic after its expiration date. It gradually loses potency. Using a vial that is a few days past its date carries minimal risk beyond reduced effectiveness. However, using a vial that is weeks or months past its date means the potency loss may be substantial, and you are essentially guessing at your dose. For detailed guidance, see our complete article on what happens if you use expired tirzepatide.
Does the type of syringe affect how fast tirzepatide goes bad?
The syringe itself does not affect stability, but the needle gauge and injection technique matter. Larger gauge needles create bigger holes in the stopper, increasing contamination risk. Using a fresh needle for each draw, the smallest practical gauge, and proper aseptic technique all help maintain vial integrity.
Can I prefill syringes with tirzepatide in advance?
This is not recommended. Peptides can adsorb onto syringe surfaces, reducing the actual dose delivered. Plastic syringes are more problematic than glass for adsorption. Pre-filled syringes also lack the protective environment of a sealed vial, making the peptide more vulnerable to oxidation and contamination. Always draw your dose immediately before injection.
What should reconstituted tirzepatide look like?
Clear and colorless if there are no additives. Slightly pink or reddish if it contains B12. No cloudiness, no particles, no sediment, no unusual color. The consistency should be similar to water. Anything else is a reason to discard. See our tirzepatide color guide for reference photos and detailed descriptions of what each formulation should look like.
What to do if you think your tirzepatide has gone bad
You have done the visual check. Something seems off. Or the dates do not add up. Or your results have suddenly changed. Here is the decision protocol.
Step 1: Visual inspection
Hold the vial up to a bright light source against a white and then a dark background. Look for cloudiness, particles, color changes, or anything that deviates from how the product normally looks. If you see any visible abnormality, go directly to step 4.
Step 2: Check the dates
When was the vial first opened or reconstituted? Is it within the 28-day window? What is the Beyond-Use Date or expiration date? Has that passed? If any date has been exceeded, go to step 4.
Step 3: Review storage history
Was the vial ever left out for an extended period? Was it ever exposed to heat or freezing? Has it been stored in the refrigerator door where temperatures fluctuate? Has the total cumulative time out of the fridge exceeded a few hours? If thermal exposure was significant, go to step 4.
Step 4: Discard and replace
When in doubt, throw it out. The cost of a vial of tirzepatide is real money, yes. But the cost of weeks of suboptimal dosing, with reduced metabolic effects, stalled weight loss results, and wasted time, is far higher. A fresh vial is always the right choice when there is any doubt about the current one.
Step 5: Investigate the cause
Why did the vial go bad? Was it a storage problem you can fix? A shipping issue you need to address with the pharmacy? An accidental room temperature exposure you can prevent in the future? Understanding the cause prevents it from happening again.
SeekPeptides members get access to detailed storage protocols, troubleshooting guides, and community support from researchers who have navigated these exact situations. If you are managing your own peptide protocols, having a reliable knowledge base makes the difference between confident handling and expensive guesswork.
The science of peptide stability testing
Understanding how stability is tested helps you interpret expiration dates and Beyond-Use Dates more intelligently.
Accelerated stability studies
Manufacturers expose the product to elevated temperatures (typically 25 degrees Celsius and 40 degrees Celsius) and measure degradation at regular intervals. Using the Arrhenius equation, they extrapolate how long the product will remain stable at refrigerated temperatures. This approach gives results in months rather than years, which is why new products can receive initial expiration dates before years of real-time data are available.
Real-time stability studies
Products are stored at recommended conditions (2 to 8 degrees Celsius for refrigerated products) and tested periodically over the full intended shelf life. These studies take years to complete but provide the most accurate stability data. Expiration dates may be extended after real-time data confirms longer-than-expected stability.
What they measure
Stability testing tracks multiple parameters. Potency (how much active peptide remains), purity (what percentage of the sample is intact versus degraded), pH (which affects degradation rates and patient safety), sterility (absence of microbial contamination), particulate matter (aggregation), and appearance (color, clarity). A product fails stability testing when any parameter falls outside its specification limit.
Forced degradation studies
Research published in the journal ACS Omega described forced degradation studies on tirzepatide under acidic, basic, oxidative, thermal, and photolytic conditions. These studies deliberately destroy the peptide under extreme conditions to identify what degradation products form. This information is critical for developing stability-indicating analytical methods that can detect degradation in real products. The research showed that tirzepatide is most vulnerable to oxidative stress and basic pH conditions.
Comparing tirzepatide stability to other GLP-1 medications
If you are considering switching between medications, understanding their relative stability can inform your decision. Different GLP-1 receptor agonists have different stability profiles based on their molecular structure and formulation.
Semaglutide shelf life follows a similar pattern to tirzepatide, with refrigerated storage recommended and a limited room temperature window. However, semaglutide is a smaller molecule (31 amino acids versus 39 for tirzepatide) with a different fatty acid modification, which gives it a somewhat different degradation profile. In practical terms, the storage requirements are comparable.
For researchers considering alternatives or switching protocols, understanding how each medication behaves outside ideal storage conditions helps manage the transition. If you are switching from tirzepatide to semaglutide or vice versa, the storage protocols are similar enough that your existing storage setup should work for either.
Comparing semaglutide and tirzepatide side effects is relevant here because some side effects, like injection site reactions, can be worsened by using degraded product. If you are experiencing unusual side effects that seem disproportionate to your dose, degraded medication is one factor worth investigating alongside the dosing protocol itself.
Long-term storage strategies for bulk purchases
Some researchers purchase tirzepatide in larger quantities to save money. Bulk purchasing creates specific storage challenges because you need to maintain potency over a longer timeframe.
Lyophilized powder for long-term storage
If you are buying in bulk, lyophilized powder is far superior to pre-reconstituted liquid. Lyophilized tirzepatide can maintain potency for extended periods when stored properly. Keep unopened vials in the refrigerator in their original packaging. Do not reconstitute until you are ready to begin using a particular vial.
Rotation system
Use a first-in, first-out system. Label each vial with the date you received it. Always use the oldest vial first. This prevents any single vial from sitting in storage longer than necessary.
Temperature monitoring
For serious long-term storage, consider a refrigerator thermometer that logs minimum and maximum temperatures. This lets you verify that your fridge maintained the proper range, which is especially useful if you experience power outages or mechanical issues. A single power outage during a heat wave could compromise every vial in your refrigerator.
Separate storage from food
Ideally, use a dedicated mini-fridge for peptide storage. This reduces temperature fluctuations from frequent door openings, eliminates the risk of food contamination, and lets you set the temperature precisely for peptide stability rather than food safety.
When in doubt, trust the discard protocol
Peptide research requires precision. You measure doses in micrograms. You track progress over weeks and months. Using degraded tirzepatide introduces an uncontrolled variable that undermines all of that careful work.
The cost of a new vial is concrete and immediate. The cost of using a bad vial is diffuse and delayed, weeks of suboptimal results, confusion about whether the protocol is working, potential setbacks that take time to identify and correct. When you frame it that way, discarding a questionable vial is not waste. It is quality control.
SeekPeptides provides comprehensive peptide storage guides, post-reconstitution protocols, and access to a community of researchers who prioritize precision in every aspect of their peptide work. If handling and storage questions keep coming up, having a single trusted resource eliminates the guesswork and protects your investment.
External resources
USP General Chapter on Beyond-Use Dating for Compounded Preparations
ACS Omega: Stability Studies on Tirzepatide Peptide Synthesis
BMC Chemistry: HPLC Stability Analysis of Semaglutide and Tirzepatide
In case I do not see you, good afternoon, good evening, and good night. May your vials stay clear, your storage stay cold, and your protocols stay effective.