Mar 21, 2026

Inside every blood sample, a molecular clock is ticking. GLP-1, the 30-amino-acid gut hormone driving the most significant weight loss revolution in pharmaceutical history, degrades within 90 seconds of entering circulation. Dipeptidyl peptidase-4 cleaves the active peptide almost immediately, converting GLP-1 (7-36) amide into its inactive metabolite GLP-1 (9-36) amide before most collection tubes even reach the centrifuge. This is why measuring GLP-1 accurately is one of the hardest problems in endocrine diagnostics.
And it is exactly why the ELISA, the enzyme-linked immunosorbent assay, has become the gold standard for GLP-1 quantification in both research and clinical settings. But here is the problem most people do not realize. Not all GLP-1 ELISA kits measure the same thing. Some detect only active GLP-1. Others measure total GLP-1, including degraded fragments. The difference between these two approaches can swing your results by 300% or more, and choosing the wrong assay can lead to fundamentally misleading conclusions about how well your GLP-1 therapy is working.
Whether you are a researcher validating peptide protocols, a clinician monitoring patient outcomes on semaglutide or tirzepatide, or someone on GLP-1 therapy who wants to understand the lab work behind the numbers, this guide covers everything. The science behind ELISA methodology.
The critical distinction between active and total assays. Sample collection protocols that prevent degradation artifacts. Normal reference ranges for fasting and postprandial GLP-1. And the practical implications for anyone using GLP-1 receptor agonists for fat loss, metabolic health, or diabetes management.
What is a GLP-1 ELISA and why does it matter
ELISA stands for enzyme-linked immunosorbent assay. It is a plate-based technique designed to detect and quantify specific proteins, peptides, antibodies, and hormones in biological samples. The technology has been the backbone of immunodiagnostics for over four decades, and for GLP-1 measurement specifically, it remains the most widely validated approach in both clinical trials and independent research.
The reason ELISA matters for GLP-1 is simple. You cannot manage what you cannot measure.
GLP-1 receptor agonists like semaglutide, tirzepatide, and retatrutide work by mimicking or amplifying the effects of endogenous GLP-1. But endogenous GLP-1 production does not stop just because you are taking an exogenous agonist. Understanding your baseline GLP-1 levels, your postprandial response, and how these change over time gives you a much clearer picture of what is actually happening metabolically. This is where ELISA comes in.
A GLP-1 ELISA kit provides all the materials needed to quantify GLP-1 concentrations in EDTA plasma, serum, or cell culture supernatant. The typical kit includes a 96-well microplate pre-coated with capture antibodies, detection antibodies conjugated to enzymes, calibration standards at known concentrations, and substrate solutions that produce a measurable color change proportional to the amount of GLP-1 present in the sample.
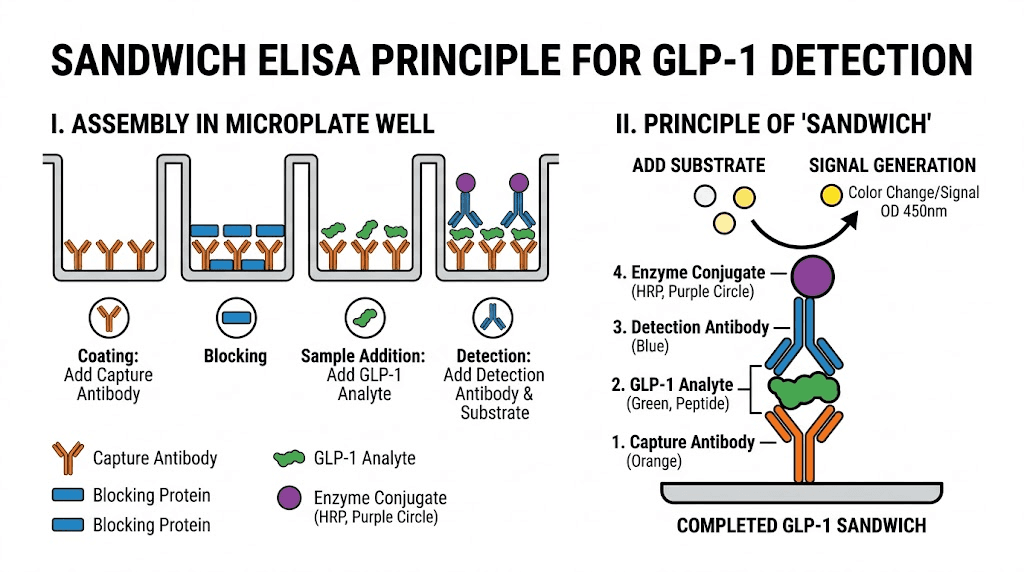
The sandwich ELISA principle explained
Most GLP-1 ELISA kits use what is called a sandwich format. Think of it literally. The GLP-1 molecule gets sandwiched between two antibodies, each recognizing a different part of the peptide.
Here is how it works step by step.
First, the microplate wells come pre-coated with a capture antibody specific to one region of the GLP-1 molecule. When you add your plasma sample to the well, any GLP-1 present binds to these capture antibodies and sticks to the plate. Everything else washes away.
Second, a detection antibody is added. This antibody recognizes a different region of GLP-1 than the capture antibody. It binds to the GLP-1 molecules already attached to the plate, forming the sandwich. This detection antibody is conjugated to biotin.
Third, an avidin-horseradish peroxidase (HRP) conjugate is added. Avidin has an extremely strong affinity for biotin, so it locks onto the detection antibody complex. The HRP enzyme is the signal generator.
Fourth, a chromogenic substrate solution is added. The HRP enzyme catalyzes a reaction that turns the substrate blue. More GLP-1 in the sample means more sandwich complexes, which means more HRP, which means more blue color.
Finally, a stop solution is added that turns the color from blue to yellow. The optical density of each well is measured with a spectrophotometer at 450nm. By comparing the optical density of your unknown samples against calibration standards of known GLP-1 concentration, you calculate the exact GLP-1 level in your sample.
The whole process takes 3-5 hours. Sensitivity ranges from 1-2 pmol/L for high-sensitivity kits to 5-10 pmol/L for standard assays.
Why GLP-1 is particularly difficult to measure
Most hormones are relatively stable in collected blood samples. Testosterone, thyroid hormones, cortisol, these molecules survive the journey from vein to analyzer without significant degradation. GLP-1 does not.
The half-life of active GLP-1 (7-36) amide in circulation is approximately 1.5 to 2 minutes. That is not a typo. Ninety seconds.
The culprit is dipeptidyl peptidase-4 (DPP-4), a ubiquitous enzyme found on endothelial cell surfaces, in plasma, and even on the surface of blood collection tubes. DPP-4 cleaves the first two amino acids from the N-terminus of active GLP-1, converting it from the biologically active (7-36) amide form to the inactive (9-36) amide metabolite.
This happens fast. It happens in vivo. And it continues happening ex vivo in your blood collection tube after the sample is drawn.
This creates a fundamental measurement challenge. If you draw blood, take 10 minutes to process it, and then run an active GLP-1 ELISA, you are not measuring what was actually circulating in the patient. You are measuring whatever survived DPP-4 degradation during those 10 minutes of sample handling. The true active GLP-1 level could have been two, three, or even five times higher than what your assay detects.
This is why sample collection protocol is just as important as the assay itself, a point we will cover in detail later in this guide.
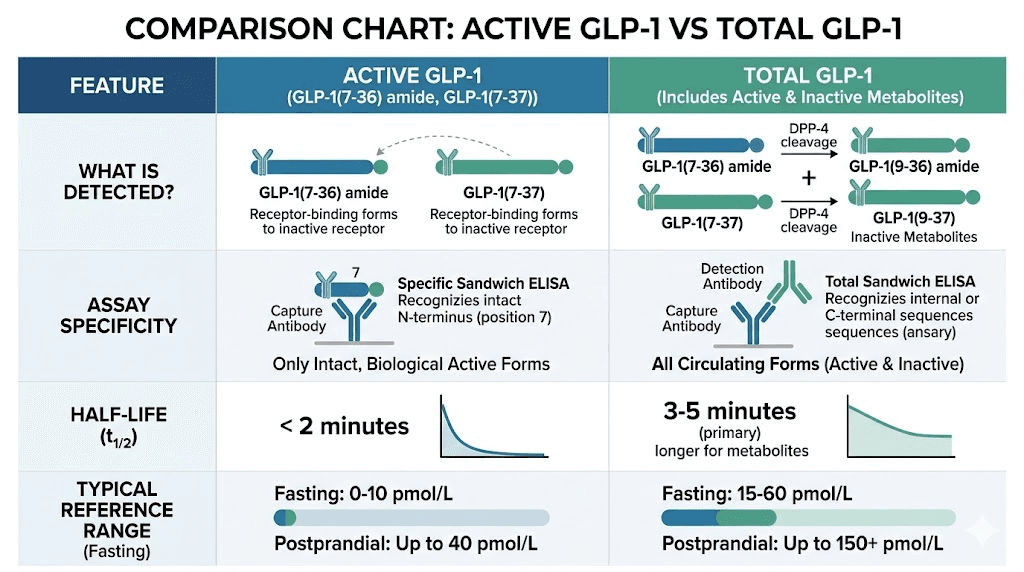
Active GLP-1 versus total GLP-1 ELISA: the critical distinction
This is where most people get confused. And it is where the most consequential mistakes happen.
There are two fundamentally different types of GLP-1 ELISA assays, and they measure very different things.
Active GLP-1 ELISA
Active GLP-1 assays use antibodies that specifically recognize the intact N-terminus of GLP-1 (7-36) amide. Once DPP-4 cleaves those first two amino acids, the resulting (9-36) fragment no longer fits the capture antibody. The assay only detects GLP-1 molecules that retain their full biological activity.
This is important for understanding functional GLP-1 signaling. Active GLP-1 is the form that actually binds to GLP-1 receptors, stimulates insulin secretion, suppresses glucagon release, and triggers the satiety signals that make appetite suppression work.
Reference ranges for active GLP-1:
Fasting: 0-15 pmol/L
Postprandial (30-60 minutes after meal): 15-50 pmol/L (2-3 fold increase)
Active GLP-1 assays are more technically demanding. Because the analyte degrades so rapidly, any delay or error in sample collection will artificially lower your results. This makes active assays more susceptible to pre-analytical variability.
Major commercial active GLP-1 ELISA kits include those from Millipore (EGLP-35K), IBL America, and ALPCO. Research from Bak et al. (2014) found that the Millipore and DRG active GLP-1 ELISAs appeared identical in specificity and were accurate for intact GLP-1 in both buffer and plasma.
Total GLP-1 ELISA
Total GLP-1 assays use antibodies that recognize a region of the GLP-1 molecule that remains intact even after DPP-4 cleavage. This means the assay detects both the active (7-36) amide form AND the inactive (9-36) metabolite.
Since the inactive metabolite is far more abundant in circulation (because active GLP-1 gets degraded so quickly), total GLP-1 levels are typically 3-5 times higher than active GLP-1 levels in the same sample.
Reference ranges for total GLP-1:
Fasting: 3-30 pmol/L
Postprandial: 20-80 pmol/L
Total GLP-1 assays are more forgiving of sample handling issues. Because they detect both active and inactive forms, even if some degradation occurs during processing, the total amount detected remains relatively stable. This makes total assays more reproducible across different collection protocols.
Leading total GLP-1 ELISA kits come from Mercodia, Meso Scale Discovery (MSD), and Millipore. The Mercodia Total GLP-1 ELISA is particularly well-validated and widely used in clinical research.
Which assay should you use
The answer depends on your question.
If you want to know how much biologically functional GLP-1 is circulating at a specific moment, use an active assay. This is most relevant for researchers studying GLP-1 receptor signaling, evaluating DPP-4 inhibitor efficacy, or measuring acute secretory responses to nutrients.
If you want to know how much GLP-1 was secreted overall (regardless of how much survived degradation), use a total assay. This is more relevant for assessing overall L-cell secretory capacity, comparing baseline GLP-1 production between individuals, or monitoring changes in endogenous GLP-1 secretion over time.
For people on GLP-1 receptor agonist therapy, there is an important nuance. Exogenous GLP-1 agonists like semaglutide and tirzepatide are structurally modified to resist DPP-4 degradation. Standard GLP-1 ELISA kits may or may not detect these synthetic agonists depending on where their antibodies bind. If you want to measure the drug itself, you need a specific pharmacokinetic assay, not a standard GLP-1 ELISA. Standard GLP-1 ELISAs are designed to measure your body's own endogenous GLP-1 production.
Sample collection protocol: the make-or-break factor
You can have the most sensitive, most specific, most expensive GLP-1 ELISA kit on the market. None of it matters if you collect your sample wrong.
Pre-analytical error, meaning mistakes that happen before the sample even reaches the assay, accounts for the majority of variability in GLP-1 measurement. A study published in the American Journal of Physiology demonstrated that without proper DPP-4 inhibition during collection, measured active GLP-1 levels can be artificially reduced by 50-80% within minutes.
Here is the correct protocol.
Step 1: prepare collection tubes in advance
Use EDTA-treated plasma collection tubes. Not serum separator tubes. Not heparin tubes. EDTA tubes.
For active GLP-1 measurement, add a DPP-4 inhibitor to the collection tube before drawing blood. Commercial options include diprotin A (Ile-Pro-Ile) at a final concentration of 0.1 mmol/L, or purpose-made DPP-4 inhibitor cocktails available from assay manufacturers like Millipore (catalog number DPP4-010). Some protocols also add aprotinin (500 KIU/mL) as a general protease inhibitor for additional protection.
For total GLP-1 measurement, DPP-4 inhibition is recommended but not strictly required, since the assay detects degraded forms anyway. However, best practice still includes it.
Step 2: draw and process immediately
Draw the blood sample and gently invert the tube 8-10 times to mix with the DPP-4 inhibitor. Do not shake vigorously.
Centrifuge immediately. The target is 1000g for 10 minutes in a refrigerated centrifuge (4 degrees Celsius). Speed matters here. Every minute between draw and centrifugation is a minute of ongoing DPP-4 activity.
Separate the plasma from the cell pellet immediately after centrifugation. Aliquot into pre-labeled cryovials. Do not use glass tubes, as GLP-1 can adhere to glass surfaces.
Step 3: storage conditions
If running the assay within 3 hours, store plasma at 2-8 degrees Celsius. For longer storage, freeze at -70 degrees Celsius or colder. Avoid -20 degree storage, as this temperature is associated with greater peptide degradation over time.
GLP-1 samples can withstand up to three freeze-thaw cycles without significant degradation, though minimizing freeze-thaw is always best practice. If you anticipate needing to run multiple assays, aliquot into small volumes (200-500 microliters) so you only thaw what you need.
Common collection mistakes that destroy accuracy
Letting the sample sit at room temperature before centrifugation is the most common error. Even 15 minutes at room temperature can reduce active GLP-1 by 40-60%.
Using serum tubes instead of EDTA tubes is another frequent mistake. Serum requires clotting time (30+ minutes), during which DPP-4 continues degrading GLP-1 unchecked.
Forgetting DPP-4 inhibitor when measuring active GLP-1 makes the results essentially meaningless. Without inhibition, you are measuring whatever random fraction of GLP-1 survived an uncontrolled degradation process.
Drawing samples into overfilled or underfilled tubes changes the EDTA-to-blood ratio, which can affect both anticoagulation and preservative effectiveness.
Understanding these details matters for anyone getting blood work done while on GLP-1 therapy. If your lab does not follow proper collection protocols, your results may not accurately reflect your actual GLP-1 status.
How GLP-1 ELISA kits compare: manufacturer analysis
Not all GLP-1 ELISA kits are created equal. A landmark validation study by Bak et al. (2014), published in Clinical Chemistry, tested multiple commercially available GLP-1 assays head-to-head and found significant differences in specificity, sensitivity, and cross-reactivity.
Here is how the major kits stack up.
Manufacturer | Type | Sensitivity | Detects | Best for | Rating |
|---|---|---|---|---|---|
Mercodia | Total | 2 pmol/L | All GLP-1 forms (amidated preference) | Clinical research, therapy monitoring | 9/10 |
MSD (Meso Scale) | Total | 0.7 pmol/L | All 6 GLP-1 isoforms | High-sensitivity research | 9/10 |
Millipore Active | Active | 2 pmol/L | GLP-1 (7-36) amide only | Functional signaling studies | 8/10 |
Millipore Total | Total | 1.5 pmol/L | All forms in buffer, amidated in plasma | General research | 7/10 |
ALPCO | Active | 0.5 pmol/L | Amidated GLP-1 only, limited range | Ultra-sensitive detection | 7/10 |
Abcam | Total | 3.9 pg/mL | GLP-1 (7-36) with cross-reactivity | Basic research | 7/10 |
RayBiotech | Total | 12 pg/mL | GLP-1 multiple forms | Budget research | 6/10 |
A few important findings from the Bak study deserve attention. The MSD total GLP-1 kit detected all six GLP-1 isoforms but showed incomplete recovery of non-active forms in plasma versus buffer. The Millipore total GLP-1 kit detected all isoforms in buffer but primarily amidated forms in plasma. The ALPCO kit detected only amidated GLP-1 and had a limited measurement range that might not cover high postprandial values.
The practical takeaway is this. If you are comparing GLP-1 data across different studies, check which assay was used. Absolute values from different manufacturers cannot be directly compared because of differences in standardization, specificity, and calibration. A reading of 25 pmol/L on one kit does not mean the same thing as 25 pmol/L on another.
Normal GLP-1 levels: what the numbers actually mean
Understanding reference ranges for GLP-1 is essential for interpreting ELISA results. But there is a catch that most resources leave out: GLP-1 levels vary dramatically based on fasting state, meal composition, body weight, metabolic health, and even time of day.
Fasting GLP-1 levels
In healthy individuals, fasting active GLP-1 levels typically fall between 0 and 15 pmol/L. Many fasting samples come back at or near the lower limit of detection, which is completely normal. GLP-1 is primarily a postprandial hormone. Your body does not secrete much of it when you have not eaten.
Fasting total GLP-1 is slightly higher, typically 3-30 pmol/L, because the inactive metabolite circulates longer than the active form.
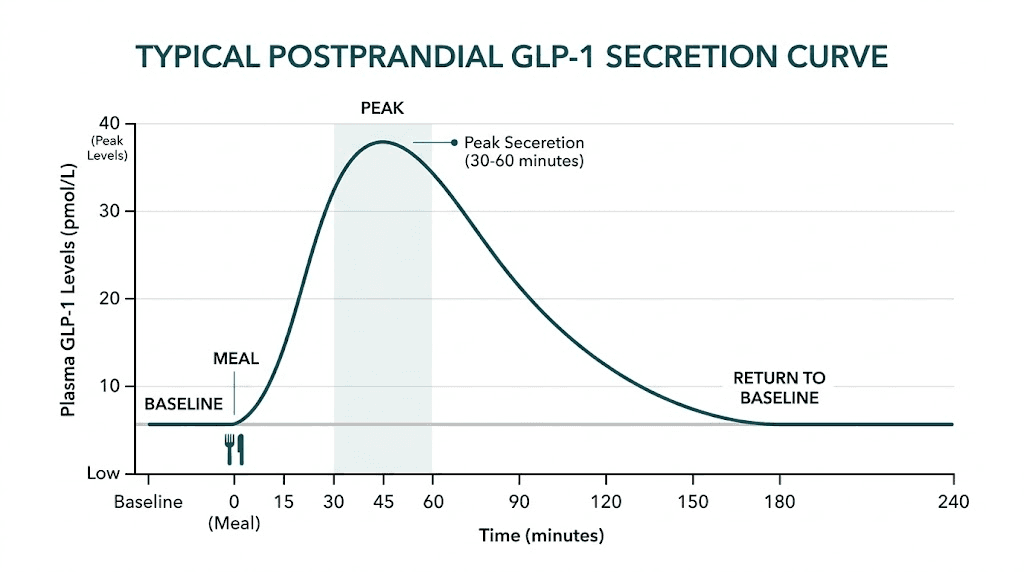
Postprandial GLP-1 levels
After a meal, GLP-1 levels increase 2-3 fold within 10-30 minutes. The peak depends on meal composition.
Protein-rich meals produce the strongest GLP-1 response. Complex carbohydrates trigger a moderate response. Pure fat produces the weakest acute response but a more sustained elevation.
For a standard mixed meal, expect active GLP-1 to peak at 15-50 pmol/L at 30-60 minutes post-meal, then return to baseline by 120-180 minutes. Total GLP-1 peaks at 20-80 pmol/L following the same pattern.
This information is particularly relevant for people managing their diet while on GLP-1 therapy or trying to understand how different foods affect their GLP-1 response.
How metabolic conditions affect GLP-1 levels
Type 2 diabetes is associated with impaired GLP-1 secretion. Fasting and postprandial GLP-1 levels are typically 20-30% lower in people with type 2 diabetes compared to metabolically healthy controls. However, the GLP-1 response to a meal is reduced, not absent, which is why GLP-1 receptor agonists still work in diabetic patients.
Obesity without diabetes shows a more complex pattern. Some studies find reduced GLP-1 secretion, others find normal or even elevated levels. The relationship likely depends on the degree of insulin resistance and the presence of other metabolic dysfunction.
Bariatric surgery, particularly Roux-en-Y gastric bypass, dramatically increases postprandial GLP-1 secretion, often by 5-10 fold. This exaggerated GLP-1 response is considered one of the key mechanisms behind the metabolic improvements seen after surgery.
Pre-diabetes shows a compensatory increase in both fasting and stimulated GLP-1 levels, according to a study published in Frontiers in Endocrinology. The theory is that the body upregulates GLP-1 secretion as an adaptive response to deteriorating glucose tolerance, but this compensation eventually fails as diabetes progresses.
GLP-1 levels during receptor agonist therapy
This is a question that comes up constantly for people on GLP-1 therapy. What happens to my endogenous GLP-1 when I take semaglutide or tirzepatide?
The answer requires nuance. Exogenous GLP-1 receptor agonists do not replace your natural GLP-1 production. They supplement it. Your body continues making and releasing GLP-1 from intestinal L-cells in response to meals. However, receptor agonist therapy can modulate endogenous GLP-1 levels through feedback mechanisms.
Some studies show a modest decrease in postprandial GLP-1 secretion during agonist therapy, possibly because the sustained receptor activation reduces the stimulus for additional endogenous release. Other studies show no significant change. The clinical significance of any such change is minimal, since the therapeutic effect comes primarily from the exogenous agonist.
What matters for ELISA interpretation: if you are measuring GLP-1 levels while on semaglutide or tirzepatide, standard ELISA kits measure your ENDOGENOUS GLP-1 only. They do not detect the drug in your system. Semaglutide and tirzepatide are structurally modified analogs that evade most anti-GLP-1 antibodies used in standard assays.
Practical applications: when and why to test GLP-1 levels
Understanding when GLP-1 ELISA testing is useful (and when it is not) can save you significant money and prevent misinterpretation of results.
When GLP-1 ELISA testing makes sense
Research protocols: If you are conducting peptide research and need to verify that a GLP-1 agonist is producing the expected physiological response, measuring endogenous GLP-1 alongside glucose and insulin provides a comprehensive metabolic picture.
Baseline assessment before therapy: Knowing your pre-treatment endogenous GLP-1 levels can help identify whether you have impaired GLP-1 secretion as part of your metabolic dysfunction. People with very low baseline GLP-1 may respond differently to therapy than those with normal secretion.
Evaluating DPP-4 inhibitor effectiveness: For patients on DPP-4 inhibitors (sitagliptin, saxagliptin, etc.), measuring the ratio of active to total GLP-1 before and after treatment directly demonstrates whether the drug is preventing GLP-1 degradation as intended.
Post-bariatric surgery monitoring: The exaggerated GLP-1 response after gastric bypass surgery can be quantified with ELISA testing, helping guide nutritional management and medication adjustments.
When GLP-1 ELISA testing is NOT useful
Measuring drug levels: As mentioned, standard GLP-1 ELISAs do not reliably detect semaglutide, tirzepatide, or other modified GLP-1 agonists. If you want to know your drug level, you need a specific pharmacokinetic assay.
Routine therapy monitoring: For most people on GLP-1 agonist therapy, clinical outcomes (weight loss, blood glucose, HbA1c) are better indicators of treatment effectiveness than endogenous GLP-1 levels. Measuring GLP-1 via ELISA adds cost without changing management decisions for most patients.
At-home monitoring: GLP-1 ELISA requires specialized equipment (microplate reader, centrifuge, -70 degree freezer) and trained personnel. It is not a point-of-care or at-home test. Anyone searching for GLP-1 monitoring at home should know that home testing focuses on downstream markers like blood glucose, HbA1c, and metabolic panels rather than GLP-1 itself.
Lab tests that actually matter for GLP-1 therapy monitoring
Since direct GLP-1 ELISA is impractical for most people monitoring their therapy, here are the lab tests that provide the most useful information.
Essential monitoring panel
Hemoglobin A1c (HbA1c): Reflects average blood glucose over 2-3 months. This is the single best marker for evaluating metabolic improvement on GLP-1 therapy. Test every 3 months during dose titration, then every 6 months once stable.
Fasting blood glucose: Provides a snapshot of glycemic control. Useful for tracking short-term changes between HbA1c tests. Normal range is 70-100 mg/dL.
Comprehensive metabolic panel (CMP): Monitors liver enzymes (ALT, AST), kidney function (creatinine, BUN, eGFR), and electrolytes. Important because GLP-1 agonists can occasionally affect liver enzymes and kidney markers.
Lipid panel: Total cholesterol, LDL, HDL, and triglycerides. GLP-1 agonists often improve lipid profiles significantly, with tirzepatide showing particularly strong improvements in triglycerides due to its dual GIP/GLP-1 mechanism.
Extended monitoring panel
Fasting insulin: Helps assess insulin resistance. Calculate HOMA-IR (fasting insulin x fasting glucose / 405) to track changes in insulin sensitivity over time.
C-reactive protein (CRP): Inflammatory marker that often decreases with GLP-1 therapy and weight loss. Useful for tracking systemic inflammation.
Thyroid function (TSH): GLP-1 receptor agonists carry a theoretical risk for thyroid C-cell effects. While this has only been demonstrated in rodent studies and not confirmed in humans, monitoring thyroid function is reasonable, especially for individuals with pre-existing thyroid conditions.
Vitamin and mineral levels: B12, iron, vitamin D, and calcium. Reduced food intake on GLP-1 therapy can lead to nutritional deficiencies. B12 monitoring is particularly important since GLP-1 agonists can impair B12 absorption in some individuals.
Monitoring frequency recommendations from major clinical guidelines suggest testing every 2-3 months during the first 6 months of therapy, then every 3-6 months once stable. SeekPeptides members access detailed monitoring protocols tailored to specific GLP-1 agonist regimens, including timing recommendations for each test relative to injection schedules.
The DPP-4 problem: why it complicates everything
If there is one concept you take away from this entire guide, let it be this. DPP-4 is the single biggest source of error in GLP-1 measurement. Understanding how it works, and how to manage it, separates reliable data from noise.
What DPP-4 does
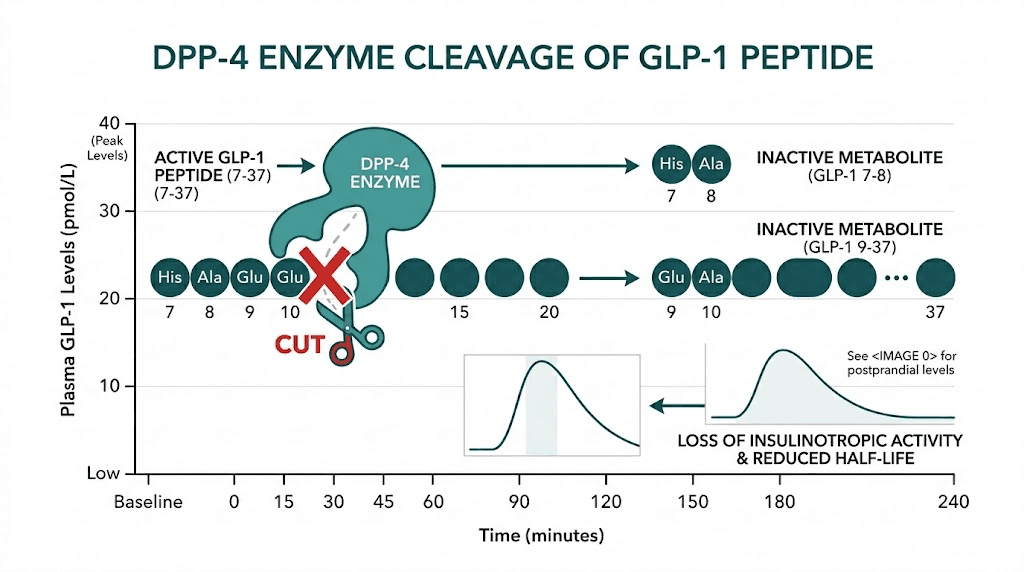
Dipeptidyl peptidase-4 is a serine protease enzyme. It cleaves dipeptides from the N-terminus of proteins that have a proline or alanine residue at the second position. GLP-1 (7-36) amide has alanine at position 8 (the second position), making it a perfect DPP-4 substrate.
The cleavage happens between positions 8 and 9, removing the Histidine-Alanine dipeptide and converting active GLP-1 (7-36) to inactive GLP-1 (9-36). This single cleavage event eliminates the peptide's ability to activate GLP-1 receptors.
DPP-4 is everywhere. It exists as a membrane-bound enzyme on endothelial cells lining blood vessels, as a soluble form circulating in plasma, and even on the surfaces of certain blood cells. The moment GLP-1 enters the bloodstream, DPP-4 starts degrading it.
How DPP-4 affects ELISA accuracy
In vivo, DPP-4 degradation is a normal physiological process. The rapid turnover of GLP-1 is how the body controls incretin signaling. But ex vivo, in your blood collection tube, DPP-4 continues working even though the sample is no longer in the body.
This creates an artifact. The longer between blood draw and sample processing, the lower the measured active GLP-1 will be. A study demonstrated that without DPP-4 inhibition, active GLP-1 levels in collected samples decrease by approximately 10-20% every 5 minutes at room temperature. By 30 minutes, you may have lost more than half of the original active GLP-1 signal.
Total GLP-1 assays are less affected because they detect the degradation products. But even total assays can be impacted if extreme conditions cause further degradation of the (9-36) metabolite by other proteases.
DPP-4 inhibitor solutions for sample collection
The gold standard is to add a DPP-4 inhibitor directly to the collection tube before the blood draw. Commercial options include:
Diprotin A (Ile-Pro-Ile): A tripeptide that competitively inhibits DPP-4. Add to a final concentration of 0.1 mmol/L. Available from most biochemical suppliers.
Sitagliptin: The same DPP-4 inhibitor used therapeutically in diabetes can be added ex vivo. Concentration of 100 micromol/L provides complete inhibition.
DPPIV-010 (Millipore): A purpose-made DPP-4 inhibitor cocktail designed specifically for blood collection. Comes in pre-measured aliquots that you add to standard collection tubes.
Aprotinin: A broad-spectrum protease inhibitor that provides additional protection against non-DPP-4 degradation. Not a DPP-4-specific inhibitor, but useful as part of a cocktail approach.
For anyone also interested in how DPP-4 relates to GLP-1 therapy effectiveness, the same enzyme that complicates measurement is why native GLP-1 has such a short half-life and why modified analogs like semaglutide (which uses an acylated fatty acid chain to bind albumin and resist DPP-4) can last days instead of minutes.
Running a GLP-1 ELISA: step-by-step laboratory protocol
For researchers, lab technicians, or anyone curious about what actually happens when a GLP-1 ELISA is run, here is the complete protocol based on standard sandwich ELISA methodology.
Materials needed
GLP-1 ELISA kit (includes pre-coated microplate, standards, detection antibody, HRP conjugate, substrate, stop solution, wash buffer concentrate), multichannel pipette and tips, microplate reader with 450nm filter, incubator or stable 37 degree Celsius environment, microplate washer (manual or automated), DPP-4 inhibitor (if measuring active GLP-1), and properly collected EDTA-plasma samples.
Day-of protocol
Preparation (30 minutes before starting): Bring all kit components to room temperature. Prepare wash buffer by diluting the concentrate according to kit instructions (typically 1:25 with deionized water). Reconstitute lyophilized standards with the provided diluent and allow to stand for 15 minutes with gentle mixing. Prepare a serial dilution of the top standard to generate a 7-point calibration curve plus a zero standard (diluent only).
Sample preparation: Thaw frozen samples on ice. Vortex gently to mix. Centrifuge briefly (10,000g for 1 minute) to remove any particulates. Dilute samples if needed based on expected concentration range (for most fasting samples, no dilution is needed).
Assay procedure:
Add 100 microliters of standards and samples to designated wells. Run standards in duplicate. Run samples in duplicate if volume allows. Cover the plate and incubate at 37 degrees Celsius for 90 minutes (or as specified by the kit).
Remove the liquid and wash each well 4-5 times with 300 microliters of wash buffer. Ensure complete aspiration between washes. Incomplete washing is the number one cause of high background signal.
Add 100 microliters of biotinylated detection antibody to each well. Cover and incubate at 37 degrees Celsius for 60 minutes.
Wash again 4-5 times.
Add 100 microliters of HRP-avidin conjugate to each well. Cover and incubate at 37 degrees Celsius for 30 minutes.
Wash 5-7 times (more washes at this stage reduce background).
Add 100 microliters of TMB substrate to each well. Incubate in the dark at room temperature for 15-25 minutes. Watch for blue color development. The timing depends on signal strength.
Add 50 microliters of stop solution. The color changes from blue to yellow. Read the plate immediately at 450nm on a microplate reader.
Data analysis: Plot the standard curve (optical density vs. GLP-1 concentration) using a four-parameter logistic (4PL) curve fit. Calculate GLP-1 concentration for each sample from the standard curve. Average duplicate wells. Multiply by any dilution factor used. Report in pmol/L or pg/mL depending on your preference.
Troubleshooting common ELISA problems
High background signal: Usually caused by insufficient washing, contaminated wash buffer, or expired substrate. Increase wash cycles and prepare fresh wash buffer.
Low signal for all samples: Could indicate sample degradation (especially for active GLP-1), incorrect incubation temperature, or expired detection antibody. Verify all reagents are within expiration and samples were properly collected.
High coefficient of variation between duplicates (greater than 15%): Pipetting error, air bubbles in wells, or uneven incubation temperature. Ensure consistent pipetting technique and verify incubator uniformity.
Standard curve does not fit: Check for pipetting errors in standard preparation. The serial dilution is the most error-prone step. A single inaccurate standard concentration will distort the entire curve.
GLP-1 ELISA versus other measurement methods
ELISA is not the only way to measure GLP-1. Understanding the alternatives helps you appreciate why ELISA remains dominant for most applications, while also knowing when other methods might be more appropriate.
Radioimmunoassay (RIA)
RIA was the original method for GLP-1 measurement, developed in the Holst laboratory in Copenhagen. It uses radiolabeled GLP-1 and specific antisera to quantify the peptide. The Copenhagen RIA, particularly the one using antiserum 89390 (C-terminally directed, measuring total GLP-1), is considered the gold standard against which all commercial kits are validated.
Advantages: extremely well-characterized, decades of validation data, highly specific antibodies. Disadvantages: requires radioactive materials and specialized disposal, longer turnaround time, not suitable for high-throughput screening, and increasingly difficult to source reagents.
Mass spectrometry (LC-MS/MS)
Liquid chromatography-tandem mass spectrometry offers absolute structural identification of GLP-1 peptides. Unlike immunoassays, which rely on antibody recognition (and can be fooled by cross-reactive molecules), mass spectrometry physically identifies the molecule based on its mass-to-charge ratio and fragmentation pattern.
Advantages: absolute specificity, can distinguish all GLP-1 isoforms simultaneously, no cross-reactivity concerns. Disadvantages: expensive equipment ($200,000+), requires highly trained operators, lower throughput than ELISA, sensitivity can be limiting for low-concentration samples.
Electrochemiluminescence (ECL)
The Meso Scale Discovery (MSD) platform uses electrochemiluminescence as its detection method instead of the colorimetric approach in traditional ELISA. The MSD V-PLEX GLP-1 assay is considered one of the most sensitive commercial options available.
Advantages: higher sensitivity than standard ELISA (sub-picomolar detection), wider dynamic range, multiplexing capability (measure multiple analytes simultaneously). Disadvantages: requires specific MSD instruments, higher cost per assay, proprietary platform.
Biosensor-based assays
Emerging technologies using surface plasmon resonance (SPR) and other biosensor approaches show promise for real-time, label-free GLP-1 detection. These are primarily research tools at present and not yet validated for clinical use.
Method | Sensitivity | Throughput | Cost/test | Equipment needed | Best for |
|---|---|---|---|---|---|
ELISA | 1-5 pmol/L | High (96-384 wells) | $5-15 | Plate reader | Clinical research, routine |
RIA | 1-2 pmol/L | Medium | $10-25 | Gamma counter | Validation, reference |
LC-MS/MS | 5-20 pmol/L | Low-Medium | $50-100 | Mass spectrometer | Structural ID, pharma |
ECL (MSD) | 0.1-1 pmol/L | High | $15-30 | MSD reader | Ultra-sensitive research |
For most applications, ELISA hits the sweet spot of sensitivity, cost, throughput, and accessibility. It is the method you will encounter in the vast majority of published GLP-1 research.

GLP-1 ELISA in clinical trials: real-world applications
GLP-1 ELISA is not just an academic exercise. It plays a central role in the clinical trials that brought semaglutide, tirzepatide, and next-generation agonists like retatrutide to market.
STEP trials (semaglutide)
The STEP (Semaglutide Treatment Effect in People with obesity) trial program used GLP-1 measurement as a secondary endpoint to understand how exogenous semaglutide affected endogenous incretin physiology. ELISA-based measurement of total GLP-1 helped researchers confirm that semaglutide's therapeutic effect was primarily receptor-mediated rather than dependent on increased endogenous GLP-1 secretion.
SURPASS trials (tirzepatide)
The SURPASS trials for tirzepatide were particularly interesting from an ELISA perspective because tirzepatide is a dual GIP/GLP-1 agonist. Researchers used both GLP-1 and GIP ELISA assays to understand how the dual mechanism affected endogenous secretion of both incretins. Results showed that tirzepatide reduced endogenous GLP-1 secretion modestly while dramatically improving glycemic control, confirming that the therapeutic effect was driven by exogenous receptor activation.
Retatrutide trials
For retatrutide, the triple agonist (GLP-1/GIP/glucagon), ELISA measurement of all three hormones was essential for understanding the complex interplay between the drug and endogenous hormone production. The Phase 2 trial data published in the New England Journal of Medicine included ELISA-derived GLP-1 measurements as part of the comprehensive metabolic assessment.
DPP-4 inhibitor trials
Perhaps the most direct application of GLP-1 ELISA is in DPP-4 inhibitor trials (sitagliptin, saxagliptin, linagliptin). These drugs work by blocking DPP-4, which prevents degradation of endogenous GLP-1. The primary pharmacodynamic endpoint in these trials is the ratio of active to total GLP-1, measured by running both ELISA types on the same samples. A successful DPP-4 inhibitor increases the active/total ratio, meaning more secreted GLP-1 survives to reach its receptors.
Understanding your lab results: practical interpretation guide
If you have had GLP-1 levels measured (or plan to), here is how to interpret the results in context.
What your results might look like
Scenario 1: Healthy individual, fasting
Active GLP-1: 3-8 pmol/L
Total GLP-1: 8-20 pmol/L
Interpretation: Normal baseline. Low fasting GLP-1 is expected and healthy.
Scenario 2: Healthy individual, 30 min post-meal
Active GLP-1: 15-40 pmol/L
Total GLP-1: 30-70 pmol/L
Interpretation: Normal postprandial response. GLP-1 levels should increase 2-3x after eating.
Scenario 3: Type 2 diabetes, fasting
Active GLP-1: 1-5 pmol/L
Total GLP-1: 5-15 pmol/L
Interpretation: Below average but within expected range for diabetes. Impaired GLP-1 secretion is part of the incretin defect in T2D.
Scenario 4: Post-bariatric surgery, post-meal
Active GLP-1: 50-200+ pmol/L
Total GLP-1: 100-400+ pmol/L
Interpretation: Markedly elevated. This exaggerated response is normal and expected after gastric bypass.
Red flags in results
If your active GLP-1 is higher than your total GLP-1 in the same sample, something is wrong. Active should always be less than or equal to total. This usually indicates a technical error, cross-reactivity in the assay, or mixed-up samples.
If your fasting active GLP-1 is greater than 30 pmol/L, verify that you were truly fasting and that the sample was collected correctly. Unusually high fasting GLP-1 could indicate a rare GLP-1-secreting tumor (very rare) or, more likely, a pre-analytical issue.
If your postprandial GLP-1 does not increase at all after a meal, suspect either a sample collection problem or genuinely impaired L-cell function. Repeat the test with strict protocol adherence before drawing conclusions.
The future of GLP-1 measurement
GLP-1 ELISA technology continues to evolve. Several developments are worth watching.
Point-of-care testing
Several companies are developing rapid lateral flow assays for GLP-1 that could eventually enable office-based or even at-home measurement. These would use the same antibody-based detection principles as ELISA but in a simplified format similar to rapid COVID tests. The challenge is achieving sufficient sensitivity, since GLP-1 circulates at picomolar concentrations, far lower than the targets of most rapid tests.
Continuous monitoring
Just as continuous glucose monitors (CGMs) revolutionized diabetes management, researchers are exploring continuous GLP-1 monitoring using implantable biosensors. This technology is still in early development, but the concept of tracking real-time GLP-1 fluctuations throughout the day could transform our understanding of incretin physiology.
Multiplex panels
Next-generation assay platforms allow simultaneous measurement of GLP-1, GIP, glucagon, insulin, C-peptide, and other metabolic hormones from a single small blood sample. The MSD platform already offers this capability, and newer technologies are pushing toward even broader panels with smaller sample requirements.
For people interested in the broader landscape of GLP-1 therapy and monitoring, these developments mean that within the next 5-10 years, comprehensive metabolic profiling could become as routine as a basic metabolic panel is today.
Cost and accessibility of GLP-1 ELISA testing
Understanding the financial side helps set realistic expectations.
Research setting costs
A standard 96-well GLP-1 ELISA kit costs between $400 and $800 depending on the manufacturer and type (active vs. total). This provides enough reagent for approximately 40 samples run in duplicate, plus calibration standards. Per-sample cost works out to roughly $10-20.
Equipment costs include a microplate reader ($5,000-15,000 for a basic model), centrifuge ($2,000-5,000), and consumables (pipette tips, tubes, wash buffer). Most research institutions already have this equipment.
Clinical laboratory costs
Commercial clinical labs that offer GLP-1 testing typically charge $50-150 per test for patients. This is often not covered by insurance unless specifically ordered as part of a diabetes workup or clinical trial. Some direct-to-consumer lab services offer GLP-1 panels at lower prices, though availability varies by region.
GLP-1 monitoring panel alternatives
For practical therapy monitoring, the more cost-effective approach is a standard metabolic blood panel that includes HbA1c, glucose, lipids, liver enzymes, and kidney function. This typically costs $50-100 at direct-to-consumer labs and provides more clinically actionable information for people on GLP-1 therapy than a direct GLP-1 level measurement.
SeekPeptides provides comprehensive monitoring guides for members on GLP-1 protocols, including which tests to order at each stage of therapy, optimal testing frequency based on your specific agonist and dose, and how to interpret results in the context of your overall protocol.
Frequently asked questions
What does a GLP-1 ELISA test actually measure?
A GLP-1 ELISA measures the concentration of glucagon-like peptide-1 in a blood sample. Active GLP-1 ELISAs specifically detect the intact, biologically functional form (7-36 amide), while total GLP-1 ELISAs detect both the active form and its inactive metabolite (9-36 amide). The result is reported in pmol/L or pg/mL.
Can I get a GLP-1 ELISA test done through my regular doctor?
Most standard clinical laboratories do not routinely offer GLP-1 ELISA testing. You would typically need to request it through an endocrinologist, a specialized metabolic clinic, or a direct-to-consumer lab service. Standard metabolic panels and HbA1c provide more practical monitoring for GLP-1 therapy and are widely available.
Will a GLP-1 ELISA show if my semaglutide or tirzepatide is working?
Not directly. Standard GLP-1 ELISA kits measure endogenous (naturally produced) GLP-1, not the synthetic agonist drug in your system. To evaluate whether your semaglutide or tirzepatide is working, clinical outcomes like weight loss, blood glucose, and HbA1c are far more informative.
What is a normal GLP-1 level?
Fasting active GLP-1 ranges from 0-15 pmol/L, while fasting total GLP-1 ranges from 3-30 pmol/L. After a meal, levels increase 2-3 fold, peaking at 30-60 minutes. Reference ranges vary by assay manufacturer, so always compare your results to the specific reference range provided by the lab that ran your test.
Why does DPP-4 matter for GLP-1 testing?
DPP-4 is an enzyme that degrades active GLP-1 within 90 seconds of secretion. It continues degrading GLP-1 in collected blood samples ex vivo. Without adding a DPP-4 inhibitor to the collection tube, active GLP-1 measurements can be artificially reduced by 50-80%, making results unreliable.
How much does a GLP-1 ELISA test cost?
Research kits cost $400-800 per 96-well plate (roughly $10-20 per sample). Clinical laboratory testing costs $50-150 per test for patients. Insurance coverage is limited. For therapy monitoring, a standard metabolic panel ($50-100) provides more actionable information at lower cost.
Can I measure GLP-1 levels at home?
No. GLP-1 ELISA requires specialized laboratory equipment including a microplate reader, centrifuge, and ultra-cold freezer. At-home GLP-1 monitoring focuses on downstream markers like blood glucose (using a glucometer or CGM), weight tracking, and symptom monitoring. Direct GLP-1 measurement at home is not currently feasible.
What is the difference between active and total GLP-1?
Active GLP-1 (7-36 amide) is the biologically functional form that binds GLP-1 receptors and triggers insulin secretion and appetite suppression. Total GLP-1 includes both the active form and the inactive metabolite (9-36 amide) produced by DPP-4 cleavage. Total levels are typically 3-5 times higher than active levels in the same sample because the inactive form circulates longer.
External resources
Bak et al. (2014) - Specificity and sensitivity of commercially available GLP-1 assays (PubMed)
Mercodia Total GLP-1 ELISA - Product specifications and validation data
Sandwich ELISA for GLP-1 metabolite measurement (American Physiological Society)
For researchers serious about optimizing their peptide protocols, SeekPeptides offers the most comprehensive resource available, with evidence-based guides, proven protocols, and a community of thousands who have navigated these exact questions.
In case I do not see you, good afternoon, good evening, and good night. May your assays stay sensitive, your samples stay preserved, and your GLP-1 data stay accurate.